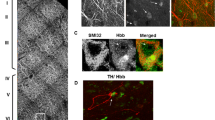
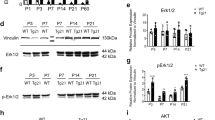
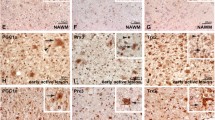
Abstract
Multiple sclerosis (MS) is a neuro-inflammatory and demyelinating disease. Downregulation of neuronal mitochondrial gene expression and activity have been reported in several studies of MS. We have previously shown that hemoglobin-β (Hbb) signals to the nucleus of neurons and upregulates H3K4me3, a histone mark involved in regulating cellular metabolism and differentiation. The present study was undertaken to evaluate the effect of erythropoietin (EPO) on the upregulation of hemoglobin and mitochondrial-associated neuroprotection. We found that administering EPO (5000 IU/kg intraperitoneally) to mice upregulated brain Hbb expression, levels of H3K4me3, expression of mitochondrial complex III, complex V, and mitochondrial respiration. We also found that the neuronal mitochondrial metabolite N-acetylaspartate (NAA), a marker of neuronal mitochondrial activity, was increased with EPO treatment. Further, we measured the effects of EPO on preventing mitochondrial deficits in the cuprizone toxic demyelinating mouse model of MS. We found that EPO prevented cuprizone-mediated decreases in Hbb, complex III, and NAA. Our data suggest that EPO mediated regulation of Hbb supports neuronal energetics and may provide neuroprotection in MS and other neurodegenerative diseases where a dysfunction of mitochondria contributes to disease.







Similar content being viewed by others
References
Bjartmar C, Kidd G, Mörk S, Rudick R, Trapp BD (2000) Neurological disability correlates with spinal cord axonal loss and reduced N-acetylaspartate in chronic multiple sclerosis patients. Ann Neurol 48:893–901
De Stefano N, Narayanan S, Francis GS, Arnaoutelis R, Tartaglia MC, Antel JP, Matthews PM, Arnold DL (2001) Evidence of axonal damage in the early stages of multiple sclerosis and its relevance to disability. Arch Neurol 58:65–70
England JD, Gamboni F, Levinson SR, Finger TE (1990) Changed distribution of sodium channels along demyelinated axons. Proc Natl Acad Sci U S A 87:6777–6780
Stys PK (2005) General mechanisms of axonal damage and its prevention. J Neurol Sci 233:3–13
Waxman SG (2006) Axonal conduction and injury in multiple sclerosis: the role of sodium channels. Nat Rev Neurosci 7:932–941
Confavreux C, Vukusic S (2006) Natural history of multiple sclerosis: a unifying concept. Brain 129:606–616
Dutta R, McDonough J, Yin X, Peterson J, Chang A, Torres T, Gudz T, Macklin WB et al (2006) Mitochondrial dysfunction as a cause of axonal degeneration in multiple sclerosis patients. Ann Neurol 59:478–489
Pandit A, Vadnal J, Houston S, Freeman E, McDonough J (2009) Impaired regulation of electron transport chain subunit genes by nuclear respiratory factor 2 in multiple sclerosis. J Neurol Sci 279:14–20
Campbell GR, Ziabreva I, Reeve AK, Krishnan KJ, Reynolds R, Howell O, Lassmann H, Turnbull DM et al (2011) Mitochondrial DNA deletions and neurodegeneration in multiple sclerosis. Ann Neurol 69:481–492
Broadwater L, Pandit A, Azzam S, Clements R, Vadnal J, Sulak M, Yong VW, Freeman EJ et al (2011) Analysis of the mitochondrial proteome in multiple sclerosis cortex. Biochim Biophys Acta 1812:630–641
Witte ME, Nijland PG, Drexhage JA, Gerritsen W, Geerts D, van Het Hof B, Reijerkerk A, de Vries HE et al (2013) Reduced expression of PGC-1partly underlies mitochondrial changes and correlates with neuronal loss in multiple sclerosis cortex. Acta Neuropathol 125:231–243
Ge Y, Gonen O, Inglese M, Babb JS, Markowitz CE, Grossman RI (2004) Neuronal cell injury precedes brain atrophy in multiple sclerosis. Neurology 62:624–627
Cader S, Johansen-Berg H, Wylezinska M, Palace J, Behrens TE, Smith S, Matthews PM (2007) Discordant white matter N-acetylasparate and diffusion MRI measures suggest that chronic metabolic dysfunction contributes to axonal pathology in multiple sclerosis. NeuroImage 36:19–27
Xuan Y, Yan G, Wu R, Huang Q, Li X, Xu H (2015) The cuprizone-induced changes in (1)H-MRS metabolites and oxidative parameters in C57BL/6 mouse brain: Effects of quetiapine. Neurochem Int 90:185–192
Praet J, Guglielmetti C, Berneman Z, Van der Linden A, Ponsaerts P (2014) Cellular and molecular neuropathology of the cuprizone mouse model: Clinical relevance for multiple sclerosis. Neurosci Biobehav Rev 47:485–505
Hagemeyer N, Boretius S, Ott C, Von Streitberg A, Welpinghus H, Sperling S et al (2012) Erythropoietin attenuates neurological and histological consequences of toxic demyelination in mice. Mol Med 18:628–635
Masuda S, Okano M, Yamagishi K, Nagao M, Ueda M, Sasaki R (1994) A novel site of erythropoietin production. Oxygen-dependent production in cultured rat astrocytes. J Biol Chem 269:19488–19493
Knabe W, Sirén AL, Ehrenreich H, Kuhn HJ (2005) Expression patterns of erythropoietin and its receptor in the developing spinal cord and dorsal root ganglia. Anat Embryol (Berl) 210:209–219
Knabe W, Knerlich F, Washausen S, Kietzmann T, Sirén AL, Brunnett G et al (2004) Expression patterns of erythropoietin and its receptor in the developing midbrain. Anat Embryol (Berl) 207:503–512
Juul SE, Anderson DK, Li Y, Christensen RD (1998) Erythropoietin and erythropoietin receptor in the developing human central nervous system. Pediatr Res 43:40–49
Hassouna I, Ott C, Wüstefeld L, Offen N, Neher RA, Mitkovski M, Winkler D, Sperling S et al (2016) Revisiting adult neurogenesis and the role of erythropoietin for neuronal and oligodendroglial differentiation in the hippocampus. Mol Psychiatry 21:1752–1767
Sargin D, Friedrichs H, El-Kordi A, Ehrenreich H (2010) Erythropoietin as neuroprotective and neuroregenerative treatment strategy: comprehensive overview of 12 years of preclinical and clinical research. Best Pract Res Clin Anaesthesiol 24:573–594
Siren AL, Fasshauer T, Bartels C, Ehrenreich H (2009) Therapeutic potential of erythropoietin and its structural or functional variants in the nervous system. Neurotherapeutics 6:108–127
Kumral A, Ozer E, Yilmaz O, Akhisaroglu M, Gokmen N, Duman N, Ulukus C, Genc S et al (2003) Neuroprotective effect of erythropoietin on hypoxic-ischemic brain injury in neonatal rats. Biol Neonate 83:224–228
Chattopadhyay A, Choudhury TD, Bandyopadhyay D, Datta AG (2000) Protective effect of erythropoietin on the oxidative damage of erythrocyte membrane by hydroxyl radical. Biochem Pharmacol 59:419–425
Bernaudin M, Marti HH, Roussel S, Divoux D, Nouvelot A, MacKenzie ET, Petit E (1999) A potential role for erythropoietin in focal permanent cerebral ischemia in mice. J Cereb Blood Flow Metab 19:643–651
Schelshorn DW, Schneider A, Kuschinsky W, Weber D, Krüger C, Dittgen T, Bürgers HF, Sabouri F et al (2009) Expression of hemoglobin in rodent neurons. J Cereb Blood Flow Metab 29:585–595
Brown N, Alkhayer K, Clements R, Singhal N, Gregory R, Azzam S, Li S, Freeman E et al (2016) Neuronal hemoglobin expression and its relevance to multiple sclerosis neuropathology. J Mol Neurosci 59:1–17
Schnaitman C, Greenawalt JW (1968) Enzymatic properties of the inner and outer membranes of rat liver mitochondria. J Cell Biol 38:158–175
Li S, Clements R, Sulak M, Gregory R, Freeman E, McDonough J (2013) Decreased NAA in gray matter is correlated with decreased availability of acetate in white matter in postmortem multiple sclerosis cortex. Neurochem Res 38:2385–2396
Schmued L, Bowyer J, Cozart M, Heard D, Binienda Z, Paule M (2008) Introducing Black-Gold II, a highly soluble gold phosphate complex with several unique advantages for the histochemical localization of myelin. Brain Res 1229:210–217
Cervellini I, Ghezzi P, Mengozzi M (2013) Therapeutic efficacy of erythropoietin in experimental autoimmune encephalomyelitis in mice, a model of multiple sclerosis. Methods Mol Biol 982:163–173
Singhal NK, Li S, Arning E, Alkhayer K, Clements R, Sarcyk Z, Dassanayake RS, Brasch NE et al (2015) Changes in methionine metabolism and histone H3 trimethylation are linked to mitochondrial defects in multiple sclerosis. J Neurosci 35:15170–15186
Sättler MB, Merkler D, Maier K, Stadelmann C, Ehrenreich H, Bähr M et al (2004) Neuroprotective effects and intracellular signaling pathways of erythropoietin in a rat model of multiple sclerosis. Cell Death Differ 11(Suppl 2):S181–S192
Digicaylioglu M, Lipton SA (2001) Erythropoietin-mediated neuroprotection involves cross-talk between Jak2 and NF-kappaB signalling cascades. Nature 412:641–647
Horng LY, Hsu PL, Chen LW, Tseng WZ, Hsu KT, Wu CL, Wu RT (2015) Activating mitochondrial function and haemoglobin expression with EH-201, an inducer of erythropoietin in neuronal cells, reverses memory impairment. Br J Pharmacol 172:4741–4756
Biagioli M, Pinto M, Cesselli D, Zaninello M, Lazarevic D, Roncaglia P et al (2009) Unexpected expression of α- and β-globin in mesencephalic dopaminergic neurons and glial cells. Proc Natl Acad Sci U S A 106:15454–15459
Clark JB (1998) N-acetyl aspartate: a marker for neuronal loss or mitochondrial dysfunction. Dev Neurosci 20:271–276
Signoretti S, Marmarou A, Tavazzi B, Lazzarino G, Beaumont A, Vagnozzi R (2001) N-Acetylaspartate reduction as a measure of injury severity and mitochondrial dysfunction following diffuse traumatic brain injury. J Neurotrauma 18:977–991
Richter F, Meurers BH, Zhu C, Medvedeva VP, Chesselet MF (2009) Neurons express hemoglobin alpha- and beta-chains in rat and human brains. J Comp Neurol 515:538–547
Ferrer I, Gómez A, Carmona M, Huesa G, Porta S, Riera-Codina M et al (2011) Neuronal hemoglobin is reduced in Alzheimer’s disease, argyrophilic grain disease, Parkinson’s disease, and dementia with Lewy bodies. J Alzheimers Dis 23:537–550
Freed J, Chakrabarti L (2016) Defining a role for hemoglobin in Parkinson’s disease. NPJ Parkinsons Dis 2:16021
Acknowledgements
This research was funded by the College of Arts and Sciences at Kent State University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Singhal, N.K., Alkhayer, K., Shelestak, J. et al. Erythropoietin Upregulates Brain Hemoglobin Expression and Supports Neuronal Mitochondrial Activity. Mol Neurobiol 55, 8051–8058 (2018). https://doi.org/10.1007/s12035-018-0971-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-0971-6