Abstract
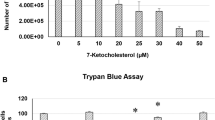
Clinacanthus nutans (Lindau) (C. nutans) has diverse uses in traditional herbal medicine for treating skin rashes, insect and snake bites, lesions caused by herpes simplex virus, diabetes mellitus and gout in Singapore, Malaysia, Indonesia, Thailand and China. We previously showed that C. nutans has the ability to modulate the induction of cytosolic phospholipase A2 (cPLA2) expression in SH-SY5Y cells through the inhibition of histone deacetylases (HDACs). In the current study, we elucidated the effect of C. nutans on the hCMEC/D3 human brain endothelial cell line. Endothelial cells are exposed to high levels of the cholesterol oxidation product, 7-ketocholesterol (7KC), in patients with cardiovascular disease and diabetes, and this process is thought to mediate pathological inflammation. 7KC induced a dose-dependent loss of hCMEC/D3 cell viability, and such damage was significantly inhibited by C. nutans leaf extracts but not stem extracts. 7KC also induced a marked increase in mRNA expression of pro-inflammatory cytokines, IL-1β IL-6, IL-8, TNF-α and cyclooxygenase-2 (COX-2) in brain endothelial cells, and these increases were significantly inhibited by C. nutans leaf but not stem extracts. HPLC analyses showed that leaf extracts have a markedly different chemical profile compared to stem extracts, which might explain their different effects in counteracting 7KC-induced inflammation. Further study is necessary to identify the putative phytochemicals in C. nutans leaves that have anti-inflammatory properties.





Similar content being viewed by others
References
Anderson, A., Campo, A., Fulton, E., Corwin, A., Jerome, W. G., 3rd., & O’Connor, M. S. (2020). 7-Ketocholesterol in disease and aging. Redox Biology, 29, 101380. https://doi.org/10.1016/j.redox.2019.101380
Cao, H., Wang, X., Zhang, B., & Ren, M. (2020). The protective effect of vitexinin septic encephalopathy by reducing leukocyte-endothelial adhesion and inflammatory response. Annals of Palliative Medicine, 9(4), 2079–2089. https://doi.org/10.21037/apm-20-1211
Chalubinski, M., Zemanek, K., Skowron, W., Wojdan, K., Gorzelak, P., & Broncel, M. (2013). The effect of 7-ketocholesterol and 25-hydroxycholesterol on the integrity of the human aortic endothelial and intestinal epithelial barriers. Inflammation Research, 62(12), 1015–1023. https://doi.org/10.1007/s00011-013-0660-x
Chang, M. C., Chen, Y. J., Liou, E. J., Tseng, W. Y., Chan, C. P., Lin, H. J., et al. (2016). 7-Ketocholesterol induces ATM/ATR, Chk1/Chk2, PI3K/Akt signalings, cytotoxicity and IL-8 production in endothelial cells. Oncotarget, 7(46), 74473–74483. https://doi.org/10.18632/oncotarget.12578
Chelyn, J. L., Omar, M. H., Mohd Yousof, N. S., Ranggasamy, R., Wasiman, M. I., & Ismail, Z. (2014). Analysis of flavone C-glycosides in the leaves of Clinacanthus nutans (Burm f) Lindau by HPTLC and HPLC-UV/DAD. Scientific World Journal, 1, 1. https://doi.org/10.1155/2014/724267
De Melo, G. O., Muzitano, M. F., Legora-Machado, A., Almeida, T. A., De Oliveira, D. B., Kaiser, C. R., et al. (2005). C-glycosylflavones from the aerial parts of Eleusine indica inhibit LPS-induced mouse lung inflammation. Planta Medica, 71(4), 362–363. https://doi.org/10.1055/s-2005-864104
Dinarello, C. A. (2002). The IL-1 family and inflammatory diseases. Clinical and Experimental Rheumatology, 20(5 Suppl 27), S1-13.
Ghelli, A., Porcelli, A. M., Zanna, C., & Rugolo, M. (2002). 7-Ketocholesterol and staurosporine induce opposite changes in intracellular pH, associated with distinct types of cell death in ECV304 cells. Archives of Biochemistry and Biophysics, 402(2), 208–217. https://doi.org/10.1016/S0003-9861(02)00085-1
Hans, C. P., Zerfaoui, M., Naura, A. S., Catling, A., & Boulares, A. H. (2008). Differential effects of PARP inhibition on vascular cell survival and ACAT-1 expression favouring atherosclerotic plaque stability. Cardiovascular Research, 78(3), 429–439. https://doi.org/10.1093/cvr/cvn018
Kamarudin, M. N. A., Sarker, M. M. R., Kadir, H. A., & Ming, L. C. (2017). Ethnopharmacological uses, phytochemistry, biological activities, and therapeutic applications of Clinacanthus nutans (Burm. f.) Lindau: A comprehensive review. Journal of Ethnopharmacology, 206, 245–266. https://doi.org/10.1016/j.jep.2017.05.007
Khoo, L. W., Audrey Kow, S., Lee, M. T., Tan, C. P., Shaari, K., Tham, C. L., et al. (2018). A comprehensive review on phytochemistry and pharmacological activities of Clinacanthus nutans (Burm.f.) Lindau. Evidence Based Complementary Alternative Medicine. https://doi.org/10.1155/2018/9276260
Koka, S., Xia, M., Chen, Y., Bhat, O. M., Yuan, X., Boini, K. M., et al. (2017). Endothelial NLRP3 inflammasome activation and arterial neointima formation associated with acid sphingomyelinase during hypercholesterolemia. Redox Biol, 13, 336–344. https://doi.org/10.1016/j.redox.2017.06.004
Lam, K. Y., Ling, A. P., Koh, R. Y., Wong, Y. P., & Say, Y. H. (2016). A Review on Medicinal Properties of Orientin. Advances in Pharmacological Sciences, 2016, 4104595. https://doi.org/10.1155/2016/4104595
Larrayoz, I. M., Huang, J. D., Lee, J. W., Pascual, I., & Rodriguez, I. R. (2010). 7-Ketocholesterol-induced inflammation: Involvement of multiple kinase signaling pathways via NFkappaB but independently of reactive oxygen species formation. Investigative Ophthalmology & Visual Science, 51(10), 4942–4955. https://doi.org/10.1167/iovs.09-4854
Le, C. F., Kailaivasan, T. H., Chow, S. C., Abdullah, Z., Ling, S. K., & Fang, C. M. (2017). Phytosterols isolated from Clinacanthus nutans induce immunosuppressive activity in murine cells. International Immunopharmacology, 44, 203–210. https://doi.org/10.1016/j.intimp.2017.01.013
Lee, C. Y., Seet, R. C., Huang, S. H., Long, L. H., & Halliwell, B. (2009). Different patterns of oxidized lipid products in plasma and urine of dengue fever, stroke, and Parkinson’s disease patients: Cautions in the use of biomarkers of oxidative stress. Antioxidants & Redox Signaling, 11(3), 407–420. https://doi.org/10.1089/ARS.2008.2179
Li, C., Cai, C., Zheng, X., Sun, J., & Ye, L. (2020). Orientin suppresses oxidized low-density lipoproteins induced inflammation and oxidative stress of macrophages in atherosclerosis. Bioscience, Biotechnology, and Biochemistry, 84(4), 774–779. https://doi.org/10.1080/09168451.2019.1702871
Lin, C. M., Huang, S. T., Liang, Y. C., Lin, M. S., Shih, C. M., Chang, Y. C., et al. (2005). Isovitexin suppresses lipopolysaccharide-mediated inducible nitric oxide synthase through inhibition of NF-kappa B in mouse macrophages. Planta Medica, 71(8), 748–753. https://doi.org/10.1055/s-2005-871287
Lv, H., Yu, Z., Zheng, Y., Wang, L., Qin, X., Cheng, G., et al. (2016). Isovitexin exerts anti-inflammatory and anti-oxidant activities on lipopolysaccharide-induced acute lung injury by inhibiting MAPK and NF-kappaB and activating HO-1/Nrf2 pathways. International Journal of Biological Sciences, 12(1), 72–86. https://doi.org/10.7150/ijbs.13188
Murakami, H., Tamasawa, N., Matsui, J., Yasujima, M., & Suda, T. (2000). Plasma oxysterols and tocopherol in patients with diabetes mellitus and hyperlipidemia. Lipids, 35(3), 333–338. https://doi.org/10.1007/s11745-000-0530-1
Qiu, J., Yuan, H., Chen, S., Zhou, Y., Song, D., & Chen, R. (2016). TNFalpha up-regulates COX-2 in chronic progressive nephropathy through nuclear accumulation of RelB and NF-kappaB2. Archives of Physiology and Biochemistry, 122(2), 88–93. https://doi.org/10.3109/13813455.2016.1141961
Rosa-Fernandes, L., Maselli, L. M. F., Maeda, N. Y., Palmisano, G., & Bydlowski, S. P. (2017). Outside-in, inside-out: Proteomic analysis of endothelial stress mediated by 7-ketocholesterol. Chemistry and Physics of Lipids, 207(Pt B), 231–238. https://doi.org/10.1016/j.chemphyslip.2017.06.008
Saokaew, S., Sugimoto, T., Kamae, I., Pratoomsoot, C., & Chaiyakunapruk, N. (2015). Healthcare databases in Thailand and Japan: Potential sources for health technology assessment research. PLoS ONE, 10(11), e0141993. https://doi.org/10.1371/journal.pone.0141993
Song, J., Wang, D., Chen, H., Huang, X., Zhong, Y., Jiang, N., et al. (2017). Association of plasma 7-ketocholesterol with cardiovascular outcomes and total mortality in patients with coronary artery disease. Circulation Research, 120(10), 1622–1631. https://doi.org/10.1161/CIRCRESAHA.117.311049
Tan, C. S., Ho, C. F., Heng, S. S., Wu, J. S., Tan, B. K., Ng, Y. K., et al. (2016). Clinacanthus nutans extracts modulate epigenetic link to cytosolic phospholipase A2 expression in SH-SY5Y cells and primary cortical neurons. Neuromolecular Medicine, 18(3), 441–452. https://doi.org/10.1007/s12017-016-8404-z
Teshima, K.-I., Kaneko, T., & Ohtani, K. (1997). C-glycosyl flavones from Clinacanthus nutans. Natural Medicines, 51(6), 557.
Tsai, H. D., Wu, J. S., Kao, M. H., Chen, J. J., Sun, G. Y., Ong, W. Y., et al. (2016). Clinacanthus nutans protects cortical neurons against hypoxia-induced toxicity by downregulating HDAC1/6. Neuromolecular Medicine, 18(3), 274–282. https://doi.org/10.1007/s12017-016-8401-2
Wang, M., Long, W., Li, D., Wang, D., Zhong, Y., Mu, D., et al. (2017). Plasma 7-ketocholesterol levels and the risk of incident cardiovascular events. Heart, 103(22), 1788–1794. https://doi.org/10.1136/heartjnl-2016-310914
Wang, W., Cheng, H., Gu, X., & Yin, X. (2019). The natural flavonoid glycoside vitexin displays preclinical antitumor activity by suppressing NF-kappaB signaling in nasopharyngeal carcinoma. Onco Targets and Therapy, 12, 4461–4468. https://doi.org/10.2147/OTT.S210077
Weksler, B., Romero, I. A., & Couraud, P. O. (2013). The hCMEC/D3 cell line as a model of the human blood brain barrier. Fluids Barriers CNS, 10(1), 16. https://doi.org/10.1186/2045-8118-10-16
Weksler, B. B., Subileau, E. A., Perriere, N., Charneau, P., Holloway, K., Leveque, M., et al. (2005). Blood-brain barrier-specific properties of a human adult brain endothelial cell line. The FASEB Journal, 19(13), 1872–1874. https://doi.org/10.1096/fj.04-3458fje
Wu, J. S., Kao, M. H., Tsai, H. D., Cheung, W. M., Chen, J. J., Ong, W. Y., et al. (2018). Clinacanthus nutans mitigates neuronal apoptosis and ischemic brain damage through augmenting the C/EBPbeta-driven PPAR-gamma transcription. Molecular Neurobiology, 55(7), 5425–5438. https://doi.org/10.1007/s12035-017-0776-z
Xiao, Q., Qu, Z., Zhao, Y., Yang, L., & Gao, P. (2017). Orientin ameliorates LPS-induced inflammatory responses through the inhibitory of the NF-kappaB pathway and NLRP3 inflammasome. Evidence-Based Complementary and Alternative Medicine, 2017, 2495496. https://doi.org/10.1155/2017/2495496
Yang, C., Xie, L., Gu, Q., Qiu, Q., Wu, X., & Yin, L. (2019). 7-Ketocholesterol disturbs RPE cells phagocytosis of the outer segment of photoreceptor and induces inflammation through ERK signaling pathway. Experimental Eye Research, 189, 107849. https://doi.org/10.1016/j.exer.2019.107849
Zhou, K., Wu, J., Chen, J., Zhou, Y., Chen, X., Wu, Q., et al. (2019). Schaftoside ameliorates oxygen glucose deprivation-induced inflammation associated with the TLR4/Myd88/Drp1-related mitochondrial fission in BV2 microglia cells. Journal of Pharmacological Sciences, 139(1), 15–22. https://doi.org/10.1016/j.jphs.2018.10.012
Acknowledgement
This work was supported by grants from the Ministry of Education, Singapore (R-181-000-183-114, WYO) and the National University Health System, Singapore (NUHSRO/2019/051/T1/Seed-Mar/04, WYO).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kuo, X., Herr, D.R. & Ong, WY. Anti-inflammatory and Cytoprotective Effect of Clinacanthus nutans Leaf But Not Stem Extracts on 7-Ketocholesterol Induced Brain Endothelial Cell Injury. Neuromol Med 23, 176–183 (2021). https://doi.org/10.1007/s12017-020-08621-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-020-08621-3