Abstract
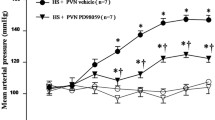
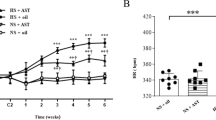
Hypertension caused by a high-salt (HS) diet is one of the major causes of cardiovascular diseases. Underlining pathology includes oxidative stress and inflammation in the hypothalamic paraventricular nucleus (PVN). This study investigates genistein’s (Gen) role in HS-induced hypertension and the underlying molecular mechanism. We placed male Wistar rats on HS (8% NaCl) or normal salt diet (0.3% NaCl). Then, we injected bilateral PVN in rats with Gen, vehicle, or nicotinamide (NAM) for 4 weeks. Tail cuff was used weekly to assess the systolic pressure, diastolic pressure, and mean arterial pressure (MAP). Cardiac hypertrophy was analyzed by heart weight/body weight ratio and wheat germ agglutinin staining. ELISA kits, Western blot, or dihydroethidium staining determined the levels of inflammatory cytokines and oxidative stress markers. Western blot measured protein levels of Sirt1, Ac-FOXO1, Nrf2, NQO-1, HO-1, and gp91phox. Our result showed that PVN infusion of Gen significantly reduced the increase of systolic pressure, diastolic pressure, and MAP induced by an HS diet. Additionally, there was a decrease in cardiac hypertrophy and the levels of inflammatory cytokines in PVN and plasma. Meanwhile, PVN infusion of Gen notably inhibited the levels of oxidized glutathione and superoxide dismutase and improved the glutathione level and total antioxidant capacities and superoxide dismutase activities. It also decreased the level of reactive oxygen species and gp91phox expression in PVN. Furthermore, Gen infusion markedly increases the Sirt1, Nrf2, HO-1, and NQO-1 levels and decreases the Ac-FOXO1 level. However, PVN infusion of NAM could significantly block these changes induced by Gen in HS diet rats. Our results demonstrated that PVN infusion of Gen could inhibit the progression of hypertension induced by an HS diet by activating the Sirt1/Nrf2 pathway.









Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Tiwana, J., & Yang, E. (2017). Clinical implications of the 2017 ACC/AHA hypertension guidelines. European Heart Journal, 40(2019), 2106–2109.
Mills, K. T., Stefanescu, A., & He, J. (2020). The global epidemiology of hypertension. Nature Reviews Nephrology, 16, 223–237.
Oparil, S., Acelajado, M. C., Bakris, G. L., Berlowitz, D. R., Cifkova, R., Dominiczak, A. F., et al. (2018). Hypertension. Nature Reviews Disease Primers, 4, 18014.
Rucker, A. J., Rudemiller, N. P., & CrowleySalt, S. D. (2018). Hypertension, and immunity. Annual Review of Physiology, 80, 283–307.
Su, Q., Liu, J. J., Cui, W., Shi, X. L., Guo, J., Li, H. B., et al. (2016). Alpha lipoic acid supplementation attenuates reactive oxygen species in hypothalamic paraventricular nucleus and sympathoexcitation in high salt-induced hypertension. Toxicology Letters, 241, 152–158.
Sun, W., Wang, X., Hou, C., Yang, L., Li, H., Guo, J., et al. (2017). Oleuropein improves mitochondrial function to attenuate oxidative stress by activating the Nrf2 pathway in the hypothalamic paraventricular nucleus of spontaneously hypertensive rats. Neuropharmacology, 113, 556–566.
Yu, X.-J., Zhang, D.-M., Jia, L.-L., Qi, J., Song, X.-A., Tan, H., et al. (2015). Inhibition of NF-κB activity in the hypothalamic paraventricular nucleus attenuates hypertension and cardiac hypertrophy by modulating cytokines and attenuating oxidative stress. Toxicology and Applied Pharmacology, 284, 315–322.
Song, X. A., Jia, L. L., Cui, W., Zhang, M., Chen, W., Yuan, Z. Y., et al. (2014). Inhibition of TNF-alpha in hypothalamic paraventricular nucleus attenuates hypertension and cardiac hypertrophy by inhibiting neurohormonal excitation in spontaneously hypertensive rats. Toxicology and Applied Pharmacology, 281, 101–108.
Su, Q., Qin, D. N., Wang, F. X., Ren, J., Li, H. B., Zhang, M., et al. (2014). Inhibition of reactive oxygen species in hypothalamic paraventricular nucleus attenuates the renin-angiotensin system and proinflammatory cytokines in hypertension. Toxicology and Applied Pharmacology, 276, 115–120.
Cheng, X. W., Narisawa, M., Jin, X., Murohara, T., & Kuzuya, M. (2018). Sirtuin 1 as a potential therapeutic target in pulmonary artery hypertension. Journal of Hypertension, 36, 1032–1035.
Nie, Q., Zhang, J., He, B., Wang, F., Sun, M., Wang, C., et al. (2020). A novel mechanism of protection against isoproterenol-induced cardiac inflammation via regulation of the SIRT1/NRF2 signaling pathway with a natural SIRT1 agonist. European Journal of Pharmacology, 886, 173398.
Sun, X., Wang, P., Yao, L. P., Wang, W., Gao, Y. M., Zhang, J., et al. (2018). Paeonol alleviated acute alcohol-induced liver injury via SIRT1/Nrf2/NF-kappaB signaling pathway. Environmental Toxicology and Pharmacology, 60, 110–117.
Yu, X. J., Xin, G. R., Liu, K. L., Liu, X. J., Fu, L. Y., Qi, J., et al. (2021). Paraventricular nucleus infusion of oligomeric proantho cyanidins improves renovascular hypertension. Frontiers in Neuroscience, 15, 642015.
Ahmed, S. M., Luo, L., Namani, A., Wang, X. J., & Tang, X. (1863). Nrf2 signaling pathway: Pivotal roles in inflammation. Biochimica et Biophysica Acta, Molecular Basis of Disease, 2017, 585–597.
Saha, S., Buttari, B., Panieri, E., Profumo, E., & Saso, L. (2020). An overview of Nrf2 signaling pathway and its role in inflammation. Molecules, 25, 5474.
Thangavel, P., Puga-Olguin, A., Rodriguez-Landa, J. F., & Zepeda, R. C. (2019). Genistein as potential therapeutic candidate for menopausal symptoms and other related diseases. Molecules, 24, 3892.
Ganai, A. A., & Farooqi, H. (2015). Bioactivity of genistein: A review of in vitro and in vivo studies. Biomedicine & Pharmacotherapy, 76, 30–38.
Matori, H., Umar, S., Nadadur, R. D., Sharma, S., Partow-Navid, R., Afkhami, M., et al. (2012). Genistein, a soy phytoestrogen, reverses severe pulmonary hypertension and prevents right heart failure in rats. Hypertension, 60, 425–430.
Gupta, S. K., Dongare, S., Mathur, R., Mohanty, I. R., Srivastava, S., Mathur, S., et al. (2015). Genistein ameliorates cardiac inflammation and oxidative stress in streptozotocin-induced diabetic cardiomyopathy in rats. Molecular and Cellular Biochemistry, 408, 63–72.
Devi, K. P., Shanmuganathan, B., Manayi, A., Nabavi, S. F., & Nabavi, S. M. (2017). Molecular and therapeutic targets of genistein in Alzheimer’s disease. Molecular Neurobiology, 54, 7028–7041.
Mukund, V., Mukund, D., Sharma, V., Mannarapu, M., & Alam, A. (2017). Genistein: Its role in metabolic diseases and cancer. Critical Reviews in Oncology Hematology, 119, 13–22.
Kang, Y. M., He, R. L., Yang, L. M., Qin, D. N., Guggilam, A., Elks, C., et al. (2009). Brain tumour necrosis factor-alpha modulates neurotransmitters in hypothalamic paraventricular nucleus in heart failure. Cardiovascular Research, 83, 737–746.
Qi, J., Zhang, D.-M., Suo, Y.-P., Song, X.-A., Yu, X.-J., Elks, C., et al. (2012). Renin-angiotensin system modulates neurotransmitters in the paraventricular nucleus and contributes to angiotensin II-induced hypertensive response. Cardiovascular Toxicology, 13, 48–54.
Wang, L. P., Yuan, D., Zheng, J., Wu, X. C., Wang, J. T., Liu, X., et al. (2019). Chikusetsu saponin IVa attenuates isoprenaline-induced myocardial fibrosis in mice through activation autophagy mediated by AMPK/mTOR/ULK1 signaling. Phytomedicine, 58, 152764.
Li, H. B., Qin, D. N., Ma, L., Miao, Y. W., Zhang, D. M., Lu, Y., et al. (2014). Chronic infusion of lisinopril into hypothalamic paraventricular nucleus modulates cytokines and attenuates oxidative stress in rostral ventrolateral medulla in hypertension. Toxicology and Applied Pharmacology, 279, 141–149.
Sun, H. J., Chen, D., Han, Y., Zhou, Y. B., Wang, J. J., Chen, Q., et al. (2016). Relaxin in paraventricular nucleus contributes to sympathetic overdrive and hypertension via PI3K-Akt pathway. Neuropharmacology, 103, 247–256.
Qi, J., Zhao, X. F., Yu, X. J., Yi, Q. Y., Shi, X. L., Tan, H., et al. (2016). Targeting interleukin-1 beta to suppress sympathoexcitation in hypothalamic paraventricular nucleus in Dahl salt-sensitive hypertensive rats. Cardiovascular Toxicology, 16, 298–306.
Incir, S., Bolayirli, I. M., Inan, O., Aydin, M. S., Bilgin, I. A., Sayan, I., et al. (2016). The effects of genistein supplementation on fructose induced insulin resistance, oxidative stress and inflammation. Life Sciences, 158, 57–62.
Gholampour, F., Mohammadi, Z., Karimi, Z., & Owji, S. M. (2020). Protective effect of genistein in a rat model of ischemic acute kidney injury. Gene, 753, 144789.
Hodgson, J. M., Puddey, I. P., Beilin, L. J., Mori, T. A., Burke, V., Croft, K. D., & Rogers, P. B. (1999). Effects of isoflavonoids on blood pressure in subjects with high-normal ambulatory blood pressure levels: A randomized controlled trial. American Journal of Hypertension, 12, 47–53.
Irace, C., Marini, H., Bitto, A., Altavilla, D., Polito, F., Adamo, E. B., et al. (2013). Genistein and endothelial function in postmenopausal women with metabolic syndrome. European Journal of Clinical Investigation, 43, 1025–1031.
Xu, J., Jackson, C. W., Khoury, N., Escobar, I., & Perez-Pinzon, M. A. (2018). Brain SIRT1 mediates metabolic homeostasis and neuroprotection. Frontiers in Endocrinology (Lausanne), 9, 702.
Schmidlin, C. J., Dodson, M. B., Madhavan, L., & Zhang, D. D. (2019). Redox regulation by NRF2 in aging and disease. Free Radical Biology & Medicine, 134, 702–707.
Li, L., Luo, W., Qian, Y., Zhu, W., Qian, J., Li, J., et al. (2019). Luteolin protects against diabetic cardiomyopathy by inhibiting NF-kappaB-mediated inflammation and activating the Nrf2-mediated antioxidant responses. Phytomedicine, 59, 152774.
Ruiz, S., Pergola, P. E., Zager, R. A., & Vaziri, N. D. (2013). Targeting the transcription factor Nrf2 to ameliorate oxidative stress and inflammation in chronic kidney disease. Kidney International, 83, 1029–1041.
Acknowledgements
This work was supported by grants Shaanxi Key Research and Development Program (No.2021SF-203), the National Natural Science Foundation of China (No.81800373), and the Natural Science Research Program of Shaanxi Province (Nos. 2020JM-079, 2020GCZX-13 and 2019JQ-263).
Author information
Authors and Affiliations
Contributions
LGN, NS, and YMK designed the study. LGN, NS, KLL, QS, and JQ performed all experiments and drafted the manuscript. YLF and QS also performed the data analysis and drafted the manuscript. GRX participated in data analysis. NS, KLL, and JQ critically revised the manuscript. LGN and YMK edited the revised manuscript language. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that there are no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Handling Editor: Y. James Kang
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Niu, LG., Sun, N., Liu, KL. et al. Genistein Alleviates Oxidative Stress and Inflammation in the Hypothalamic Paraventricular Nucleus by Activating the Sirt1/Nrf2 Pathway in High Salt-Induced Hypertension. Cardiovasc Toxicol 22, 898–909 (2022). https://doi.org/10.1007/s12012-022-09765-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-022-09765-3