Abstract
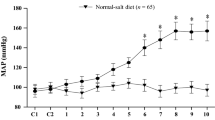
Angiotensin II (ANG II)-induced inflammatory and oxidative stress responses contribute to the pathogenesis of hypertension. In this study, we determined whether renin–angiotensin system (RAS) activation in the hypothalamic paraventricular nucleus (PVN) contributes to the ANG II-induced hypertensive response via interaction with neurotransmitters in the PVN. Rats underwent subcutaneous infusion of ANG II or saline for 4 weeks. These rats were treated for 4 weeks through bilateral PVN infusion with either vehicle or losartan (LOS), an angiotensin II type 1 receptor (AT1-R) antagonist, via osmotic minipump. ANG II infusion resulted in higher levels of glutamate, norepinephrine (NE), AT1-R and pro-inflammatory cytokines (PIC), and lower level of gamma-aminobutyric acid (GABA) in the PVN. Rats receiving ANG II also had higher levels of mean arterial pressure, plasma PIC, NE and aldosterone than control animals. PVN treatment with LOS attenuated these ANG II-induced hypertensive responses. In conclusion, these findings suggest that the RAS activation in the PVN contributes to the ANG II-induced hypertensive response via interaction with PIC and neurotransmitters (glutamate, NE and GABA) in the PVN.






Similar content being viewed by others
References
Zhu, Z., Zhu, S., Zhu, J., van der Giet, M., & Tepel, M. (2004). Effect of sodium on vasoconstriction and angiotensin II type 1 receptor mRNA expression in cold-induced hypertensive rats. Clinical and Experimental Hypertension, 26, 475–483.
Zhu, G. Q., Gao, L., Patel, K. P., Zucker, I. H., & Wang, W. (2004). ANG II in the paraventricular nucleus potentiates the cardiac sympathetic afferent reflex in rats with heart failure. Journal of Applied Physiology, 97, 1746–1754.
Veerasingham, S. J., & Raizada, M. K. (2003). Brain renin-angiotensin system dysfunction in hypertension: recent advances and perspectives. British Journal of Pharmacology, 139, 191–202.
Simpson, J. B. (1981). The circumventricular organs and the central actions of angiotensin. Neuroendocrinology, 32, 248–256.
Zhu, G. Q., Gao, L., Patel, K. P., Zucker, I. H., & Wang, W. (2004). ANG II in the paraventricular nucleus potentiates the cardiac sympathetic afferent reflex in rats with heart failure. Journal of Applied Physiology, 97, 1746–1754.
Sriramula, S., Cardinale, J. P., Lazartigues, E., & Francis, J. (2011). ACE2 overexpression in the paraventricular nucleus attenuates angiotensin II-induced hypertension. Cardiovascular Research, 92, 401–408.
Granger, J. P. (2006). An emerging role for inflammatory cytokines in hypertension. American Journal of Physiology-Heart and Circulatory Physiology, 290, H923–H924.
Phillips, M. I., & Kagiyama, S. (2002). Angiotensin II as a pro-inflammatory mediator. Current Opinion in Investigational Drugs, 3, 569–577.
Zhang, Z. H., Wei, S. G., Francis, J., & Felder, R. B. (2003). Cardiovascular and renal sympathetic activation by blood-borne TNF in rat: the role of central prostaglandins. American Journal of Physiol-Regulatory Integrative and Comparative Physiology, 284, R916–R927.
Kang, Y. M., Gao, F., Li, H. H., Cardinale, J. P., Elks, C., et al. (2011). NF-κB in the paraventricular nucleus modulates neurotransmitters and contributes to sympathoexcitation in heart failure. Basic Research in Cardiology, 106, 1087–1097.
Kang, Y. M., Zhang, A. Q., Zhao, X. F., Cardinale, J. P., Elks, C., et al. (2011). Paraventricular nucleus corticotrophin releasing hormone contributes to sympathoexcitation via interaction with neurotransmitters in heart failure. Basic Research in Cardiology, 106, 473–483.
Sriramula, S., Cardinale, J. P., Lazartigues, E., & Francis, J. (2011). ACE2 overexpression in the paraventricular nucleus attenuates angiotensin II-induced hypertension. Cardiovascular Research, 92, 401–408.
Cardinale, J. P., Sriramula, S., Mariappan, N., Agarwal, D., & Francis, J. (2012). Angiotensin II–induced hypertension is modulated by nuclear factor-B in the paraventricular nucleus. Hypertension, 59, 113–121.
Francis, J., MohanKumar, S. M., & MohanKumar, P. S. (2000). Correlations of norepinephrine release in the paraventricular nucleus with plasma corticosterone and leptin after systemic lipopolysaccharide: blockade by soluble IL-1 receptor. Brain Research, 867, 180–187.
Cowling, R. T., Gurantz, D., Peng, J., Dillmann, W. H., & Greenberg, B. H. (2002). Transcription factor NF-kappa B is necessary for up-regulation of type 1 angiotensin II receptor mRNA in rat cardiac fibroblasts treated with tumor necrosis factor-alpha or interleukin-1beta. Journal of Biological Chemistry, 277, 5719–5724.
Kang, Y. M., He, R. L., Yang, L. M., Qin, D. N., Guggilam, A., et al. (2009). Brain tumour necrosis factor-alpha modulates neurotransmitters in hypothalamic paraventricular nucleus in heart failure. Cardiovascular Research, 83, 737–746.
MohanKumar, S. M., MohanKumar, P. S., & Quadri, S. K. (1998). Specificity of interleukin-1beta-induced changes in monoamine concentrations in hypothalamic nuclei: blockade by interleukin-1 receptor antagonist. Brain Research Bulletin, 47, 29–34.
Yang, L. M., Hu, B., Xia, Y. H., Zhang, B. L., & Zhao, H. (2008). Lateral habenula lesions improve the behavioral response in depressed rats via increasing the serotonin level in dorsal raphe nucleus. Behavioural Brain Research, 188, 84–90.
Barber, M., Kasturi, B. S., Austin, M. E., Patel, K. P., MohanKumar, S. M., et al. (2003). Diabetes-induced neuroendocrine changes in rats: role of brain monoamines, insulin and leptin. Brain Research, 964, 128–135.
Guggilam, A., Haque, M., Kerut, E. K., McIlwain, E., Lucchesi, P., et al. (2007). TNF-alpha blockade decreases oxidative stress in the paraventricular nucleus and attenuates sympathoexcitation in heart failure rats. American Journal of Physiology—Heart Circulation Physiology, 293, H599–H609.
Guggilam, A., Patel, K. P., Haque, M., Ebenezer, P. J., Kapusta, D. R., et al. (2008). Cytokine blockade attenuates sympathoexcitation in heart failure: cross-talk between nNOS, AT-1R and cytokines in the hypothalamic paraventricular nucleus. European Journal of Heart Failure, 10, 625–634.
Kang, Y. M., Zhang, Z. H., Johnson, R. F., Yu, Y., Beltz, T., et al. (2006). Novel effect of mineralocorticoid receptor antagonism to reduce proinflammatory cytokines and hypothalamic activation in rats with ischemia-induced heart failure. Circulation Research, 99, 758–766.
Sriramula, S., Haque, M., Majid, D. S., & Francis, J. (2008). Involvement of tumor necrosis factor-alpha in angiotensin II-mediated effects on salt appetite, hypertension, and cardiac hypertrophy. Hypertension, 51, 1345–1351.
Kang, Y. M., Ma, Y., Zheng, J. P., Elks, C., Sriramula, S., et al. (2009). Brain nuclear factor-kappa B activation contributes to neurohumoral excitation in angiotensin II-induced hypertension. Cardiovascular Research, 82, 503–512.
Gorbea-Oppliger, V. J., & Fink, G. D. (1995). Cerebroventricular injection of angiotensin II antagonist: effects on blood pressure responses to central and systemic angiotensin II. Journal of Pharmacology and Experimental Therapeutics, 273, 611–616.
Ruiz-Ortega, M., Ruperez, M., Lorenzo, O., Esteban, V., Blanco, J., et al. (2002). Angiotensin II regulates the synthesis of proinflammatory cytokines and chemokines in the kidney. Kidney International, 82, S12–S22.
Arenas, I. A., Xu, Y., Lopez-Jaramillo, P., & Davidge, S. T. (2004). Angiotensin II-induced MMP-2 release from endothelial cells is mediated by TNF alpha. American Journal of Physiology-Cell Physiology, 286, C779–C784.
Sasamura, H., Nakazato, Y., Hayashida, T., Kitamura, Y., Hayashi, M., et al. (1997). Regulation of vascular type 1 angiotensin receptors by cytokines. Hypertension, 30, 35–41.
Shi, Z., Gan, X. B., Fan, Z. D., Zhang, F., Zhou, Y. B., et al. (2011). Inflammatory cytokines in paraventricular nucleus modulate sympathetic activity and cardiac sympathetic afferent reflex in rats. Acta Physiology (Oxford), 203, 289–297.
Kang, Y. M., Ma, Y., Elks, C., Zheng, J. P., Yang, Z. M., et al. (2008). Cross-talk between cytokines and renin–angiotensin in hypothalamic paraventricular nucleus in heart failure: role of nuclear factor-κB. Cardiovascular Research, 79, 671–678.
Wardener, H. E. (2001). The hypothalamus and hypertension. Physiology Review, 81, 1599–1658.
Fan, Z. D., Zhang, L., Shi, Z., Gan, X. B., Gao, X. Y., et al. (2012). Artificial microRNA interference targeting AT1a receptors in paraventricular nucleus attenuates hypertension in rats. Gene Therapy, 19, 810–817.
Cottone, S., Vadalà, A., Vella, M. C., Nardi, E., Mulé, G., et al. (1998). Changes of plasma endothelin and growth factor levels, and of left ventricular mass, after chronic AT1-receptor blockade in human hypertension. American Journal of Hypertension, 11, 548–553.
Francis, J., Zhang, Z. H., Weiss, R. M., & Felder, R. B. (2004). Neural regulation of the proinflammatory cytokine response to acute myocardial infarction. American Journal of Physiology—Heart Circulation Physiology, 287, H791–H797.
Kenney, M. J., Weiss, M. L., & Haywood, J. R. (2003). The paraventricular nucleus: an important component of the central neurocircuitry regulating sympathetic nerve out flow. Acta Physiologica Scandinavica, 177, 7–15.
Swanson, L. W., & Sawchenko, P. E. (1998). Paraventricular nucleus: a site for the integration of neuroendocrine and autonomic mechanisms. Neuroendocrinology, 31, 410–417.
Li, Y. F., Jackson, K. L., Stern, J. E., Rabeler, B., & Patel, K. P. (2006). Interaction between glutamate and GABA systems in the integration of sympathetic outflow by the paraventricular nucleus of the hypothalamus. American Journal of Physiology—Heart Circulation Physiology, 291, H2847–H2856.
Acknowledgments
This study was supported by National Basic Research Program of China (No. 2012CB517805), National Natural Science Foundation of China (Nos. 81170248, 81070199), Research Fund for the Doctoral Program of Higher Education of China (No. 20101417110002), Shanxi Natural Science Foundation (No. 2010011052-1) and Fundamental Research Funds for the Central Universities of China (No. 08142001).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors Jie Qi, Dong-Mei Zhang, Yu-Ping Suo and Xin-Ai Song contributed equally to this study.
Rights and permissions
About this article
Cite this article
Qi, J., Zhang, DM., Suo, YP. et al. Renin–Angiotensin System Modulates Neurotransmitters in the Paraventricular Nucleus and Contributes to Angiotensin II-Induced Hypertensive Response. Cardiovasc Toxicol 13, 48–54 (2013). https://doi.org/10.1007/s12012-012-9184-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-012-9184-9