Abstract
Astaxanthin (AST) has a variety of biochemical effects, including anti-inflammatory, antioxidative, and antihypertensive functions. The aim of the present study was to determine whether AST ameliorates blood pressure in salt-induced prehypertensive rats by ROS/MAPK/NF-κB pathways in hypothalamic paraventricular nucleus.
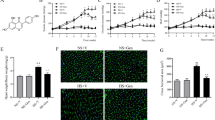
To explore the central effects of AST on the development of blood pressure, prehypertensive rats were induced by a high-salt diet (HS, 8% NaCl) and its control groups were treated with normal-salt diet (NS, 0.3% NaCl). The Dahl salt-sensitive (S) rats with HS diet for 6 weeks received AST or vehicle by gastric perfusion for 6 weeks. Compared to those with NS diet, rats with HS diet exhibited increased mean arterial pressure (MAP) and heart rate (HR). These increases were associated with higher plasma level of norepinephrine (NE), interleukin 1β (IL-1β), and interleukin 6 (IL-6); elevated PVN level of reactive oxygen species (ROS), NOX2, and NOX4, that of IL-1β, IL-6, monocyte chemotactic protein 1 (MCP-1), tyrosine hydroxylase (TH), phosphorylation extracellular-signal-regulated kinase (p-ERK1/2), phosphorylation Jun N-terminal kinases (p-JNK), nuclear factor-kappa B (NF-κB) activity; and lower levels of IL-10, superoxide dismutase (SOD), and catalase (CAT) in the PVN. In addition, our data demonstrated that chronic AST treatment ameliorated these changes in the HS but not NS diet rats. These data suggested that AST could alleviate prehypertensive response in HS-induced prehypertension through ROS/MAPK/NF-κB pathways in the PVN.










Similar content being viewed by others
References
Sueta, D., Koibuchi, N., Hasegawa, Y., Toyama, K., Uekawa, K., Katayama, T., Ma, M. J., Nakagawa, T., Waki, H., Maeda, M., Ogawa, H., & Kim-Mitsuyama, S. (2014). Blood pressure variability, impaired autonomic function and vascular senescence in aged spontaneously hypertensive rats are ameliorated by angiotensin blockade. Atherosclerosis, 236, 101–107.
Xu, M. L., Yu, X. J., Zhao, J. Q., Du, Y., Xia, W. J., Su, Q., Du, M. M., Yang, Q., Qi, J., Li, Y., Zhou, S. W., Zhu, G. Q., Li, H. B., & Kang, Y. M. (2020). Calcitriol ameliorated autonomic dysfunction and hypertension by down-regulating inflammation and oxidative stress in the paraventricular nucleus of SHR. Toxicology and Applied Pharmacology, 394, 114950.
Mittal, B. V., & Singh, A. K. (2010). Hypertension in the developing world: Challenges and opportunities. American Journal of Kidney Diseases, 55, 590–598.
Campbell, N. R. C., & Chen, G. M. (2010). Canadian efforts to prevent and control hypertension. Canadian Journal of Cardiology, 26, 14c–17c.
Grassi, G., & Ram, V. S. (2016). Evidence for a critical role of the sympathetic nervous system in hypertension. Journal of the American Society of Hypertension, 10, 457–466.
Thomas, P., & Dasgupta, I. (2015). The role of the kidney and the sympathetic nervous system in hypertension. Pediatric Nephrology(Berlin, Germany), 30, 549–560.
Marciante, A. B., Wang, L. A., Little, J. T., & Cunningham, J. T. (2020). Caspase lesions of PVN-projecting MnPO neurons block the sustained component of CIH-induced hypertension in adult male rats. American Journal of Physiology-Heart and Circulatory, 318, H34–H48.
Dos Santos, K. M., Moraes, D. J. A., da Silva, M. P., & Antunes, V. R. (2021). Exercise training rescues the electrical activity of liver-projecting DMNV neurones in response to oxytocin in spontaneously hypertensive rats. Journal of Neuroendocrinology, 33, e12977.
Shi, Z., Gan, X. B., Fan, Z. D., Zhang, F., Zhou, Y. B., Gao, X. Y., De, W., & Zhu, G. Q. (2011). Inflammatory cytokines in paraventricular nucleus modulate sympathetic activity and cardiac sympathetic afferent reflex in rats. Acta Physiologica, 203, 289–297.
Kang, Y. M., Ma, Y., Zheng, J. P., Elks, C., Sriramula, S., Yang, Z. M., & Francis, J. (2009). Brain nuclear factor-kappa B activation contributes to neurohumoral excitation in angiotensin II-induced hypertension. Cardiovascular Research, 82, 503–512.
Cardinale, J. P., Sriramula, S., Mariappan, N., Agarwal, D., & Francis, J. (2012). Angiotensin II-induced hypertension is modulated by nuclear factor-kappa B in the paraventricular nucleus. Hypertension, 59, 113-U282.
McCartney, D. M., Byrne, D. G., & Turner, M. J. (2015). Dietary contributors to hypertension in adults reviewed. Irish Journal of Medical Science, 184, 81–90.
Zou, Z., Wang, M., Wang, Z., Aluko, R. E., & He, R. (2020). Antihypertensive and antioxidant activities of enzymatic wheat bran protein hydrolysates. Jounal of Food Biochemistry, 44, e13090.
Ambati, R. R., Phang, S. M., Ravi, S., & Aswathanarayana, R. G. (2014). Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications-a review. Marine Drugs, 12, 128–152.
Galasso, C., Orefice, I., Pellone, P., Cirino, P., Miele, R., Ianora, A., Brunet, C. & Sansone, C. (2018). On the neuroprotective role of astaxanthin: new perspectives? Marine drugs, 16.
Kumar, A., Dhaliwal, N., Dhaliwal, J., Dharavath, R. N., & Chopra, K. (2020). Astaxanthin attenuates oxidative stress and inflammatory responses in complete Freund-adjuvant-induced arthritis in rats. Pharmacological Reports, 72, 104–114.
Monroy-Ruiz, J., Sevilla, M. A., Carron, R., & Montero, M. J. (2011). Astaxanthin-enriched-diet reduces blood pressure and improves cardiovascular parameters in spontaneously hypertensive rats. Pharmacological Research, 63, 44–50.
Stewart, J. S., Lignell, A., Pettersson, A., Elfving, E., & Soni, M. G. (2008). Safety assessment of astaxanthin-rich microalgae biomass: Acute and subchronic toxicity studies in rats. Food and Chemical Toxicology, 46, 3030–3036.
Hatabu, T., Harada, T., Takao, Y., Thi, D. H., Yamasato, A., Horiuchi, T., Mochizuki, A., & Kondo, Y. (2020). Daily meal supplemented with astaxanthin-enriched yolk has mitigative effects against hypertension in spontaneously hypertensive rats. Biological &/and Pharmaceutical Bulletin, 43, 404–408.
Zhou, L. P., Gao, M., Xiao, Z. M., Zhang, J., Li, X. M., & Wang, A. M. (2015). Protective effect of astaxanthin against multiple organ injury in a rat model of sepsis. Journal of Surgical Research, 195, 559–567.
Zhang, X. S., Zhang, X., Wu, Q., Li, W., Wang, C. X., Xie, G. B., Zhou, X. M., Shi, J. X., & Zhou, M. L. (2014). Astaxanthin offers neuroprotection and reduces neuroinflammation in experimental subarachnoid hemorrhage. Journal of Surgical Research, 192, 206–213.
Yu, X. J., Zhao, Y. N., Hou, Y. K., Li, H. B., Xia, W. J., Gao, H. L., Liu, K. L., Su, Q., Yang, H. Y., Liang, B., Chen, W. S., Cui, W., Li, Y., Zhu, G. Q., Yang, Z. M., & Kang, Y. M. (2019). Chronic intracerebroventricular infusion of metformin inhibits salt-sensitive hypertension via attenuation of oxidative stress and neurohormonal excitation in rat paraventricular nucleus. Neuroscience Bulletin, 35, 57–66.
Palkovits, M. (1973). Isolated removal of hypothalamic or other brain nuclei of the rat. Brain Research, 59, 449–450.
Chen, Y. M., Yu, X. J., Liu, K. L., Gao, H. L., Li, Y., Sun, T. Z., Shi, X. L., Li, H. B., Zhu, G. Q., Qi, J., & Kang, Y. M. (2020). Inhibition of hypothalamic inhibitor kappa b kinase beta/nuclear transcription factor kappa b pathway attenuates metabolism and cardiac dysfunction in type 2 diabetic rats. Neuroendocrinology, 110, 899–913.
Sueta, D., Koibuchi, N., Hasegawa, Y., Toyama, K., Uekawa, K., Katayama, T., Ma, M., Nakagawa, T., Waki, H., Maeda, M., Ogawa, H., & Kim-Mitsuyama, S. (2014). Blood pressure variability, impaired autonomic function and vascular senescence in aged spontaneously hypertensive rats are ameliorated by angiotensin blockade. Atherosclerosis, 236, 101–107.
Li, H. B., Li, X., Huo, C. J., Su, Q., Guo, J., Yuan, Z. Y., Zhu, G. Q., Shi, X. L., Liu, J. J., & Kang, Y. M. (2016). TLR4/MyD88/NF-kappa B signaling and PPAR-gamma within the paraventricular nucleus are involved in the effects of telmisartan in hypertension. Toxicology and Applied Pharmacology, 305, 93–102.
Li, H. B., Yang, T., Richards, E. M., Pepine, C. J., & Raizada, M. K. (2020). Maternal treatment with captopril persistently alters gut-brain communication and attenuates hypertension of male offspring. Hypertension, 75, 1315–1324.
Sharma, R. K., Oliveira, A. C., Kim, S., Rigatto, K., Zubcevic, J., Rathinasabapathy, A., Kumar, A., Lebowitz, J. J., Khoshbouei, H., Lobaton, G., Aquino, V., Richards, E. M., Katovich, M. J., Shenoy, V., & Raizada, M. K. (2018). Involvement of neuroinflammation in the pathogenesis of monocrotaline-induced pulmonary hypertension. Hypertension, 71, 1156–1163.
Li, Y., Yu, X. J., Xiao, T., Chi, H. L., Zhu, G. Q., & Kang, Y. M. (2021). Nrf1 knock-down in the hypothalamic paraventricular nucleus alleviates hypertension through intervention of superoxide production-removal balance and mitochondrial function. Cardiovascular Toxicology, 21, 472–489.
Xia, W. J., Xu, M. L., Yu, X. J., Du, M. M., Li, X. H., Yang, T., Li, L., Li, Y., Kang, K. B., Su, Q., Xu, J. X., Shi, X. L., Wang, X. M., Li, H. B., & Kang, Y. M. (2021). Antihypertensive effects of exercise involve reshaping of gut microbiota and improvement of gut-brain axis in spontaneously hypertensive rat. Gut Microbes, 13, 1–24.
Kuo, M.H., Lee, H.F., Tu, Y.F., Lin, L.H., Cheng, Y.Y., & Lee, H.T. (2019). Astaxanthin ameliorates ischemic-hypoxic-induced neurotrophin receptor p75 upregulation in the endothelial cells of neonatal mouse brains. International Journal of Molecular Sciences, 20.
Grimmig, B., Kim, S. H., Nash, K., Bickford, P. C., & Douglas Shytle, R. (2017). Neuroprotective mechanisms of astaxanthin: A potential therapeutic role in preserving cognitive function in age and neurodegeneration. Geroscience, 39, 19–32.
Allison, M. A., Aragaki, A. K., Ray, R. M., Margolis, K. L., Beresford, S. A., Kuller, L., Jo O’Sullivan, M., Wassertheil-Smoller, S., & Van Horn, L. (2016). A randomized trial of a low-fat diet intervention on blood pressure and hypertension: Tertiary analysis of the whi dietary modification trial. American Journal of Hypertension, 29, 959–968.
Tian, H., Kang, Y. M., Gao, H. L., Shi, X. L., Fu, L. Y., Li, Y., Jia, X. Y., Liu, K. L., Qi, J., Li, H. O., Chen, Y. I., Chen, W. S., Cui, W., Zhu, G. Q., & Yu, X. J. (2019). Chronic infusion of berberine into the hypothalamic paraventricular nucleus attenuates hypertension and sympathoexcitation via the ROS/Erk1/2/iNOS pathway. Phytomedicine, 52, 216–224.
Su, Q., Qin, D. N., Wang, F. X., Ren, J., Li, H. B., Zhang, M., Yang, Q., Miao, Y. W., Yu, X. J., Qi, J., Zhu, Z. M., Zhu, G. Q., & Kang, Y. M. (2014). Inhibition of reactive oxygen species in hypothalamic paraventricular nucleus attenuates the renin-angiotensin system and proinflammatory cytokines in hypertension. Toxicol Appl Pharm, 276, 115–120.
Tan, X., Jiao, P. L., Wang, Y. K., Wu, Z. T., Zeng, X. R., Li, M. L., & Wang, W. Z. (2017). The phosphoinositide-3 kinase signaling is involved in neuroinflammation in hypertensive rats. CNS Neuroscience & Therapeutics, 23, 350–359.
Yu, X. J., Zhang, D. M., Jia, L. L., Qi, J., Song, X. A., Tan, H., Cui, W., Chen, W., Zhu, G. Q., Qin, D. N., & Kang, Y. M. (2015). Inhibition of NF-kappaB activity in the hypothalamic paraventricular nucleus attenuates hypertension and cardiac hypertrophy by modulating cytokines and attenuating oxidative stress. Toxicology and Applied Pharmacology, 284, 315–322.
Visioli, F., & Artaria, C. (2017). Astaxanthin in cardiovascular health and disease: Mechanisms of action, therapeutic merits, and knowledge gaps. Food & Function, 8, 39–63.
Stefanadi, E., Tousoulis, D., Androulakis, E. S., Papageorgiou, N., Charakida, M., Siasos, G., Tsioufis, C., & Stefanadis, C. (2010). Inflammatory markers in essential hypertension: Potential clinical implications. Current Vascular Pharmacology, 8, 509–516.
Wang, M.L., Kang, Y.M., Li, X.G., Su, Q., Li, H.B., Liu, K.L., Fu, L.Y., Saahene, R.O., Li, Y., Tan, H., Yu, X.J. (2018). Central blockade of NLRP3 reduces blood pressure via regulating inflammation microenvironment and neurohormonal excitation in salt-induced prehypertensive rats. Journal of Neuroinflammation, 15.
Wang, M. L., Yu, X. J., Li, X. G., Pang, D. Z., Su, Q., Saahene, R. O., Li, H. B., Mao, X. Y., Liu, K. L., Fu, L. Y., Li, Y., Zhu, G. Q., & Kang, Y. M. (2018). Blockade of TLR4 within the paraventricular nucleus attenuates blood pressure by regulating ros and inflammatory cytokines in prehypertensive rats. American Journal of Hypertension, 31, 1013–1023.
Suzuki, Y., Ohgami, K., Shiratori, K., Jin, X. H., Ilieva, I., Koyama, Y., Yazawa, K., Yoshida, K., Kase, S., & Ohno, S. (2006). Suppressive effects of astaxanthin against rat endotoxin-induced uveitis by inhibiting the NF-kappaB signaling pathway. Experimental Eye Research, 82, 275–281.
Speranza, L., Pesce, M., Patruno, A., Franceschelli, S., de Lutiis, M. A., Grilli, A., & Felaco, M. (2012). Astaxanthin treatment reduced oxidative induced pro-inflammatory cytokines secretion in U937: SHP-1 as a novel biological target. Marine Drugs, 10, 890–899.
Li, J., Wang, F., Xia, Y., Dai, W., Chen, K., Li, S., Liu, T., Zheng, Y., Wang, J., Lu, W., Zhou, Y., Yin, Q., Lu, J., Zhou, Y., & Guo, C. (2015). Astaxanthin pretreatment attenuates hepatic ischemia reperfusion-induced apoptosis and autophagy via the ROS/MAPK pathway in mice. Marine Drugs, 13, 3368–3387.
Yang, X., Guo, A.L., Pang, Y.P., Cheng, X.J., Xu, T., Li, X.R., Liu, J., Zhang, Y.Y., & Liu, Y. (2019). Astaxanthin attenuates environmental tobacco smoke-induced cognitive deficits: a critical role of p38 MAPK. Marine Drugs, 17.
Gao, H. L., Yu, X. J., Liu, K. L., Shi, X. L., Qi, J., Chen, Y. M., Zhang, Y., Bai, J., Yi, Q. Y., Feng, Z. P., Chen, W. S., Cui, W., Liu, J. J., Zhu, G. Q., & Kang, Y. M. (2017). PVN blockade of p44/42 mapk pathway attenuates salt-induced hypertension through modulating neurotransmitters and attenuating oxidative stress. Scientific Reports, 7, 43038.
Zhao, Y. N., Li, J. M., Chen, C. X., Zhang, P., & Li, S. X. (2015). Hypertension-mediated enhancement of JNK activation in association with endoplasmic reticulum stress in rat model hippocampus with cerebral ischemia-reperfusion. Genetics and Molecular Research : GMR, 14, 10980–10990.
Acknowledgements
This work was supported by National Natural Science Foundation of China (Nos. 81770426, 82070439, 82070440), the Fundamental Research Funds for the Central Universities (Nos. PY3A044, xjh012019059), China Postdoctoral Science Foundation (Nos. 2019M663750, 2020M683440), and Natural Science Basic Research Program of Shaanxi (Nos. 2021JQ-068, 2020JM-079, 2019JQ-605).
Author information
Authors and Affiliations
Contributions
YK, HG, and XY designed the study. HG, WC, LY, and KL performed all experiments. HG and NJ also performed the data analysis and drafted the manuscript. YZ, YL, DZ, JY, and DZ participated in data analysis. YL, DZ, HT, and NZ critically revised the manuscript. All authors reviewed the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Handling Editor: Kurt J. Varner.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, HL., Yu, XJ., Zhang, Y. et al. Astaxanthin Ameliorates Blood Pressure in Salt-Induced Prehypertensive Rats Through ROS/MAPK/NF-κB Pathways in the Hypothalamic Paraventricular Nucleus. Cardiovasc Toxicol 21, 1045–1057 (2021). https://doi.org/10.1007/s12012-021-09695-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-021-09695-6