Abstract
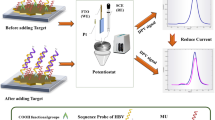
In this work, we report the construction of a novel electrochemical device for molecular diagnosis of hepatitis B virus in the blood plasma of infected patients, using graphite electrodes functionalized with poly(4-aminophenol) and sensitized with a specific DNA probe. The recognition of genomic DNA was evaluated by electrochemical techniques (DPV and EIS) and scanning electron microscopy. The genosensor was efficient in detecting genomic DNA with a linear range from 1.176 to 4.825 μg mL−1 and detection limit of 35.69 ng mL−1 (4.63 IU ml−1 or 25.93 copies.ml−1), which is better than the 10.00 IU ml−1 limit of reference method, real-time PCR, used in point of care. EIS analysis shows that the genosensor resistance increased exponentially with the concentration of the genomic DNA target. This novel platform has advantages to its applicability in real samples, such as good sensitivity, selectivity, low sample volume, and fast assay time (36 min), thus interesting for application in the diagnosis of hepatitis B virus in blood plasma. Also, the ease of synthesis of the low-cost polymer by electrosynthesis directly on the electrode surface allows the translation of the platform to portable devices.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
World Health O. (2021). Global progress report on HIV, viral Hepatitis and sexually transmitted infections, 2021: accountability for the global health sector strategies 2016-2021: actions for impact. Web annex 2: data methods; Worlds Health Organization. https://apps.who.int/iris/handle/10665/342813
Nguyen, M. H., Wong, G., Gane, E., Kao, J. H., & Dusheiko, G. (2020). Hepatitis B virus: Advances in prevention, diagnosis, and therapy. Clinical Microbiology Reviews 33(2), e00046–19. https://doi.org/10.1128/CMR.00046-19
Yang, L., Du, F., Chen, G., Yasmeen, A., & Tang, Z. (2014). A novel colorimetric PCR-based biosensor for detection and quantification of hepatitis B virus. Analytical Chimica Acta, 840, 75–81. https://doi.org/10.1016/j.aca.2014.05.032
Oliveira, D. A., Silva, J. V., Flauzino, J. M. R., Castro, A. C. H., Moço, A. C. R., Soares, M. M. C. N., Madurro, J. M., & Brito-Madurro, A. G. (2018). Application of nanomaterials for the electrical and optical detection of the hepatitis B virus. Analytical Biochemistry, 549, 157–163. https://doi.org/10.1016/j.ab.2018.03.023
Xiao, G., Zhu, F., Wang, M., Zhang, H., Ye, D., Yang, J., Jiang, L., Liu, C., Yan, L., & Qin, R. (2016). Diagnostic accuracy of APRI and FIB-4 for predicting hepatitis B virus-related liver fibrosis accompanied with hepatocellular carcinoma. Digestive and Liver Disease, 48(10), 1220–1226. https://doi.org/10.1016/j.dld.2016.06.001
Mao, X., Liu, S., Yang, C., Liu, F., Wang, K., & Chen, G. (2016). Colorimetric detection of hepatitis B virus (HBV) DNA based on DNA-templated copper nanoclusters. Analytica Chimica Acta, 909, 101–108. https://doi.org/10.1016/j.aca.2016.01.009
Mashhadizadeh, M. H., & Talemi, R. P. (2016). Synergistic effect of magnetite and gold nanoparticles onto the response of a label-free impedimetric hepatitis B virus DNA biosensor. Materials Science and Engineering C, 59, 773–781. https://doi.org/10.1016/j.msec.2015.10.082
Liu C. J., Williams K. E., Orr H. T. & Akkin T. (2017). Visualizing and mapping the cerebellum with serial optical coherence scanner. Neurophotonics 4(1), 011006. https://doi.org/10.1117/1.NPh.4.1.011006
Rodrigues, L. P., Ferreira, D. C., Ferreira, L. F., Cuadros-Orellana, S., Oliveira, G. C., Brito-Madurro, A. G., & Madurro, J. M. (2015). Electropolymerization of hydroxyphenylacetic acid isomers and the development of a bioelectrode for the diagnosis of bacterial meningitis. Journal of Applied Electrochemistry, 45(12), 1277–1287. https://doi.org/10.1007/s10800-015-0892-2
Jasim, A., Ullah, M. W., Shi, Z., Lin, X., & Yang, G. (2017). Fabrication of bacterial cellulose/polyaniline/single-walled carbon nanotubes membrane for potential application as biosensor. Carbohydrate Polymers, 163, 62–69. https://doi.org/10.1016/j.carbpol.2017.01.056
Deng, H., Li, Z., Bian, Z., Yang, F., Liu, S., Fan, Z., Wang, Y., & Tang, G. (2017). Influence of measurement uncertainty on the world health organization recommended regulation for mainstream cigarette smoke constituents. Regulatory Toxicology and Pharmacology, 86, 231–240. https://doi.org/10.1016/j.yrtph.2017.03.010
Lu, Q., Wang, H., Liu, Y., Hou, Y., Li, H., & Zhang, Y. (2017). Graphitic carbon nitride nanodots: As reductant for the synthesis of silver nanoparticles and its biothiols biosensing application. Biosensors and Bioelectronics, 89, 411–416. https://doi.org/10.1016/j.bios.2016.05.064
Presnova, G., Presnov, D., Krupenin, V., Grigorenko, V., Trifonov, A., Andreeva, I., Ignatenko, O., Egorov, A., & Rubtsova, M. (2017). Biosensor based on a silicon nanowire field-effect transistor functionalized by gold nanoparticles for the highly sensitive determination of prostate specific antigen. Biosensors and Bioelectronics, 88, 283–289. https://doi.org/10.1016/j.bios.2016.08.054
Rezaei, B., Shams-Ghahfarokhi, L., Havakeshian, E., & Ensafi, A. A. (2016). An electrochemical biosensor based on nanoporous stainless steel modified by gold and palladium nanoparticles for simultaneous determination of levodopa and uric acid. Talanta, 158, 42–50. https://doi.org/10.1016/j.talanta.2016.04.061
Skotadis, E., Voutyras, K., Chatzipetrou, M., Tsekenis, G., Patsiouras, L., Madianos, L., Chatzandroulis, S., Zergioti, I., & Tsoukalas, D. (2016). Label-free DNA biosensor based on resistance change of platinum nanoparticles assemblies. Biosensors and Bioelectronics, 81, 388–394. https://doi.org/10.1016/j.bios.2016.03.028
Heli, H., Sattarahmady, N., Hatam, G. R., Reisi, F., & Vais, R. D. (2016). An electrochemical genosensor for Leishmania major detection based on dual effect of immobilization and electrocatalysis of cobalt-zinc ferrite quantum dots. Talanta, 156, 172–179. https://doi.org/10.1016/j.talanta.2016.04.065
Rahi, A., Sattarahmady, N., & Heli, H. (2016). An ultrasensitive electrochemical genosensor for Brucella based on palladium nanoparticles. Analytical Biochemistry, 510, 11–17. https://doi.org/10.1016/j.ab.2016.07.012
Castro, A. C. H., Alves, L. M., Siquieroli, A. C. S., Madurro, J. M., & Brito-Madurro, A. G. (2020). Label-free electrochemical immunosensor for detection of oncomarker CA125 in serum. Microchemical Journal, 155, 104746. https://doi.org/10.1016/j.microc.2020.104746
Castro, A. C. H., Kochi, L. T., Moço, A. C. R., Coimbra, R. S., Oliveira, G. C., Cuadros-Orellana, S., Madurro, J. M., & Brito-Madurro, A. G. (2018). A new genosensor for meningococcal meningitis diagnosis using biological samples. Journal of Solid State Electrochemistry, 22(8), 2339–2346. https://doi.org/10.1007/s10008-018-3940-0
Balvedi, R. P. A., Castro, A. C. H., Madurro, J. M., & Brito-Madurro, A. G. (2014). Detection of a specific biomarker for Epstein-Barr virus using a polymer-based genosensor. International Journal of Molecular Sciences, 15(5), 9051–9066. https://doi.org/10.3390/ijms15059051
Silva, T. D. S., Castro, A. C. H., Rodovalho, V. R., Madurro, J. M., & Brito-Madurro, A. G. (2016). Development of electrochemical genosensor for MYCN oncogene detection using rhodamine B as electroactive label. Journal of Solid State Electrochemistry, 20(9), 2411–2418. https://doi.org/10.1007/s10008-016-3326-0
Singhal, C., Ingle, A., Chakraborty, D., Pn, A. K., Pundir, C. S., & Narang, J. (2017). Impedimetric genosensor for detection of hepatitis C virus (HCV1) DNA using viral probe on methylene blue doped silica nanoparticles. International Journal of Biological Macromolecules, 98, 84–93. https://doi.org/10.1016/j.ijbiomac.2017.01.093
Oliveira, D. A., Silva, J. V., Flauzino, J. M. R., Sousa, H. S., Castro, A. C. H., Moço, A. C. R., Soares, M. M. C. N., Madurro, J. M., & Brito-Madurro, A. G. (2019). Carbon nanomaterial as platform for electrochemical genosensor: A system for the diagnosis of the hepatitis C in real sample. Journal of Electroanalytical Chemistry, 844, 6–13. https://doi.org/10.1016/j.jelechem.2019.04.045
Moço, A. C. R., Guedes, P. H. G., Flauzino, J. M. R., Silva, H. S., Vieira, J. G., Castro, A. C. H., Gomes, E. V. R., Tolentino, F. M., Madurro, J. M., & Brito-Madurro, A. G. (2019). Electrochemical detection of Zika virus in biological samples: A step for diagnosis point-of-care. Electroanalysis, 31(8), 1580–1587. https://doi.org/10.1002/elan.201900068
Afonso, A. S., Goulart, L. R., Goulart, I. M. B., Machado, A. E. H., Madurro, J. M., & Brito-Madurro, A. G. (2010). A promising bioelectrode based on gene of Mycobacterium leprae immobilized onto poly(4-aminophenol). Journal of Applied Polymer Science, 118(5), 2921–2928. https://doi.org/10.1002/app.32595
Zhao, F., Bai, Y., Cao, L., Han, G., Fang, C., Wei, S., & Chen, Z. (2020). New electrochemical DNA sensor based on nanoflowers of Cu3(PO4)2-BSA-GO for hepatitis B virus DNA detection. Journal of Electroanalytical Chemistry, 867, 114184. https://doi.org/10.1016/j.jelechem.2020.114184
Lok, A. S. F., Akarca, U. & Greene, S. (1994). Mutations in the pre-core region of hepatitis B virus serve to enhance the stability of the secondary structure of the pre-genome encapsidation signal. Proceedings of the National Academy of Sciences U.S.A., 91(9), 4077–4081. https://doi.org/10.1073/pnas.91.9.4077
Vieira, S. N., Ferreira, L. F., Franco, D. L., Afonso, A. S., Gonçalves, R. A., Brito-Madurro, A. G., & Madurro, J. M. (2006). Electrochemical modification of graphite electrodes with poly(4-aminophenol). Macromolecular Symposia, 245–246(1), 236–242. https://doi.org/10.1002/masy.200651333
Waring, M. J. (1974). Stabililzation of two-standard ribohomopolymer helices and destabilization of a three-stranded helix by ethidium bromide. Biochemical Journal, 143(2), 483–486. https://doi.org/10.1042/bj1430483
Pastré, D., Piétrement, O., Zozime, A., & Cam, E. (2005). Study of the DNA/ethidium bromide interactions on mica surface by atomic force microscope: Influence of the surface friction. Biopolymers, 77(1), 53–62. https://doi.org/10.1002/bip.20185
European Association For The Study Of The Liver. (2012). EASL clinical practice guidelines: Management of chronic hepatitis B virus infection. Journal of Hepatology, 57(1), 167–185. https://doi.org/10.1016/j.jhep.2012.02.010
Keeffe, E. B., Dieterich, D. T., Han, S. H., Jacobson, I. M., Martin, P., Schiff, E. R., Tobias, H., & Wright, T. L. (2006). A treatment algorithm for the management of chronic hepatitis B virus infection in the United States: An update. Clinical Gastroenterology and Hepatology, 4(8), 936–962. https://doi.org/10.1016/j.cgh.2006.05.016
Caliendo, A. M., Valsamakis, A., Bremer, J. W., Ferreira-Gonzalez, A., Granger, S., Sabatini, L., Tsongalis, G. J., Wang, Y. F., Yen-Lieberman, B., Young, S., & Lurain, N. S. (2011). Multilaboratory evaluation of real-time PCR tests for hepatitis B virus DNA quantification. Journal of Clinical Microbiology, 49(8), 2854–2858. https://doi.org/10.1128/jcm.00471-11
Ismail, A. M., Sivakumar, J., Anantharam, R., Dayalan, S., Samuel, P., Fletcher, G. J., Gnanamony, M., & Abraham, P. (2011). Performance characteristics and comparison of Abbott and Artus real-time systems for hepatitis B virus DNA quantification. Journal of Clinical Microbiology, 49(9), 3215–3221. https://doi.org/10.1128/jcm.00915-11
Drexler, J. F., Reber, U., Wuttkopf, A., Eis-Hübinger, A. M., & Drosten, C. (2012). Performance of the novel Qiagen artus QS-RGQ viral load assays compared to that of the Abbott RealTime system with genetically diversified HIV and hepatitis C Virus plasma specimens. Journal of Clinical Microbiology, 50(6), 2114–2117. https://doi.org/10.1128/jcm.05874-11
Han, H., Wang, C., Ma, Z., & Su, Z. (2006). A facile method to produce highly monodispersed nanospheres of cystine aggregates. Nanotechnology, 17(20), 5163–5166. https://doi.org/10.1088/0957-4484/17/20/021
Riedel, T., Surman, F., Hageneder, S., Pop-Georgievski, O., Noehammer, C., Hofner, M., Brynda, E., Rodriguez-Emmenegger, C., & Dostálek, J. (2016). Hepatitis B plasmonic biosensor for the analysis of clinical serum samples. Biosensors and Bioelectronics, 85, 272–279. https://doi.org/10.1016/j.bios.2016.05.014
Wang, X., Li, Y., Wang, H., Fu, Q., Peng, J., Wang, Y., Du, J., Zhou, Y., & Zhan, L. (2010). Gold nanorod-based localized surface plasmon resonance biosensor for sensitive detection of hepatitis B virus in buffer, blood serum and plasma. Biosensors and Bioelectronics, 26(2), 404–410. https://doi.org/10.1016/j.bios.2010.07.121
Amiri, A. R., & Macgregor, R. B. (2011). The effect of hydrostatic pressure on the thermal stability of DNA hairpins. Biophysical Chemistry, 156(1), 88–95. https://doi.org/10.1016/j.bpc.2011.02.001
Quinchia, J., Echeverri, D., Cruz-Pacheco, A. F., Maldonado, M. E., & Orozco, J. (2020). Electrochemical biosensors for determination of colorectal tumor biomarkers. Micromachines, 11(4), 411. https://doi.org/10.3390/mi11040411
Wang, Y., Zhao, G., Zhang, Y., Pang, X., Cao, W., Du, B., & Wei, Q. (2018). Sandwich-type electrochemical immunosensor for CEA detection based on Ag/MoS2@Fe3O4 and an analogous ELISA method with total internal reflection microscopy. Sensors and Actuators B: Chemical, 266, 561–569. https://doi.org/10.1016/j.snb.2018.03.178
Chen, Y. J., Peng, Y. R., Lin, H. Y., Hsueh, T.-Y., Lai, C. S. & Hua, M. Y. (2021). Preparation and characterization of Au/NiPc/Anti-p53/BSA electrode for application as a p53 antigen sensor. Chemosensors 9(1), 17. https://doi.org/10.3390/chemosensors9010017
Popov, A., Brasiunas, B., Kausaite-Minkstimiene, A. & Ramanaviciene, A. (2021). Metal nanoparticle and quantum dot tags for signal amplification in electrochemical immunosensors for biomarker detection. Chemosensors, 9(4), 85. https://doi.org/10.3390/chemosensors9040085
Castro, A. C. H., França, E. G., de Paula, L. F., Soares, M. M. C. N., Goulart, L. R., Madurro, J. M., & Brito-Madurro, A. G. (2014). Preparation of genosensor for detection of specific DNA sequence of the hepatitis B virus. Applied Surface Science, 314, 273–279. https://doi.org/10.1016/j.apsusc.2014.06.084
Ferreira, L. F., Souza, L. M., Franco, D. L., Castro, A. C. H., Oliveira, A. A., Boodts, J. F. C., Brito-Madurro, A. G., & Madurro, J. M. (2011). Formation of novel polymeric films derived from 4-hydroxybenzoic acid. Materials Chemistry and Physics, 129(1–2), 46–52. https://doi.org/10.1016/j.matchemphys.2011.03.053
Funding
The authors are grateful for the financial support from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Contributions
Ana C. H. Castro: conceptualization, methodology, formal analysis, investigation, writing. Leandro T. Kochi: methodology, investigation, conceptualization, writing. José M. R. Flauzino: methodology, investigation, validation, formal analysis. Márcia M. C. N. Soares: human serum sample supply, methodology. Valéria A. Alves: methodology, formal analysis. Luís A. da Silva: methodology, formal analysis. Joao M. Madurro: investigation, conceptualization, supervision, writing – reviewing and editing, funding acquisition. Ana G. Brito Madurro: investigation, conceptualization, supervision, writing – reviewing and editing, funding acquisition.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Castro, A.C.H., Kochi, L.T., Flauzino, J.M.R. et al. Electrochemical Biosensor for Sensitive Detection of Hepatitis B in Human Plasma. Appl Biochem Biotechnol 194, 2604–2619 (2022). https://doi.org/10.1007/s12010-022-03829-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-03829-4