Abstract
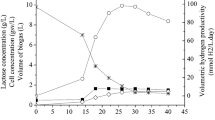
A newly isolated photo non-sulfur (PNS) bacterium was identified as Rhodopseudomonas palustris PB-Z by sequencing of 16S ribosomal DNA (rDNA) genes and phylogenetic analysis. Under vigorous stirring (240 rpm), the hydrogen production performances were greatly improved: The maximum hydrogen production rate and cumulative hydrogen production increased by 188.9 ± 0.07 % and 83.0 ± 0.06 %, respectively, due to the hydrogen bubbles were immediately removed from the culture medium. The effects of different wavelength of light on hydrogen production with stirring were much different from that without stirring. The ranking on the photo-hydrogen production performance was white > yellow > green > blue > red without stirring and white > yellow > blue > red > green under stirring. The best light source for hydrogen production was tungsten filament lamp. The optimum temperature was 35 °C. The maximal hydrogen production rate and cumulative hydrogen production reached 78.7 ± 2.3 ml/l/h and 1728.1 ± 92.7 mol H2/l culture, respectively, under 35 °C, 240 rpm, and illumination of 4000 lux. Pyruvate was one of the main sources of CO2 and has a great impact on the gas composition.








Similar content being viewed by others
References
Das, D. (2009). Advances in biohydrogen production processes: an approach towards commercialization. International Journal of Hydrogen Energy, 34, 7349–7357.
Keskin, T., & Hallenbeck, P. C. (2012). Hydrogen production from sugar industry wastes using single-stage photofermentation. Bioresource Technology, 112, 131–136.
Pott, R. W., Howe, C. J., & Dennis, J. S. (2013). Photofermentation of crude glycerol from biodiesel using Rhodopseudomonas palustris: comparison with organic acids and the identification of inhibitorycompounds. Bioresource Technology, 130, 725–730.
Das, D., & Veziroğlu, T. N. (2001). Hydrogen production by biological processes: a survey of literature. International Journal of Hydrogen Energy, 26, 13–28.
Adessi, A., & De Philippis, R. (2014). Photobioreactor design and illumination systems for H2 production with anoxygenic photosynthetic bacteria: a review. International Journal of Hydrogen Energy, 39, 3127–3141.
Özkan, E., Uyar, B., Özgür, E., Yücel, M., Eroglu, I., & Gündüz, U. (2012). Photofermentative hydrogen production using dark fermentation effluent of sugar beet thick juice in outdoor conditions. International Journal of Hydrogen Energy, 37, 2044–2049.
Chen, C. Y., Yeh, K. L., Lo, Y. C., Wang, H. M., & Chang, J. S. (2010). Engineering strategies for the enhanced photo-H2 production using effluents of dark fermentation processes as substrate. International Journal of Hydrogen Energy, 35, 13356–13364.
Bianchi, L., Mannelli, F., Viti, C., Adessi, A., & De Philippis, R. (2010). Hydrogen-producing purple non-sulfur bacteria isolated from the trophic lake Averno (Naples, Italy). International Journal of Hydrogen Energy, 35, 12216–12223.
Wu, T. Y., Hay, J. X. W., Kong, L. B., Juan, J. C., & Jahim, J. M. (2012). Recent advances in reuse of waste material as substrate to produce biohydrogen by purple non-sulfur (PNS) bacteria. Renewable and Sustainable Energy Reviews, 16, 3117–3122.
Basak, N., & Das, D. (2006). The prospect of purple non-sulfur (PNS) photosynthetic bacteria for hydrogen production: the present state of the art. World Journal of Microbiology and Biotechnology, 23, 31–42.
Ren, H. Y., Liu, B. F., Ding, J., Nan, J., Xie, G. J., Zhao, L., Chen, M. G., & Ren, N. Q. (2012). Enhanced photo-hydrogen production of Rhodopseudomonas faecalis RLD-53 by EDTA addition. International Journal of Hydrogen Energy, 37, 8277–8281.
Laocharoen, S., & Reungsang, A. (2014). Isolation, characterization and optimization of photo-hydrogen production conditions by newly isolated Rhodobacter sphaeroides KKU-PS5. International Journal of Hydrogen Energy, 39, 10870–10882.
Chen, C. Y., & Chang, J. S. (2014). Enhancing phototropic hydrogen production by solid-carrier assisted fermentation and internal optical-fiber illumination. Process Biochemistry, 41, 2041–2049.
Xie, G. J., Liu, B. F., Ding, J., Xing, D. F., Ren, H. Y., Guo, W. Q., & Ren, N. Q. (2012). Enhanced photo-H2 production by Rhodopseudomonas faecalis RLD-53 immobilization on activated carbon fibers. Biomass and Bioenergy, 44, 122–129.
Akroum-Amrouche, D., Abdi, N., Lounici, H., & Mameri, N. (2011). Effect of physico-chemical parameters on biohydrogen production and growth characteristics by batch culture of Rhodobacter sphaeroides CIP 60.6. Applied Energy, 88, 2130–2135.
Dar, M. A., Pawar, K. D., Jadhav, J. P., & Pandit, R. S. (2015). Isolation of cellulolytic bacteria from the gastro-intestinal tract of Achatina fulica (Gastropoda: Pulmonata) and their evaluation for cellulose biodegradation. International Biodeterioration & Biodegradation, 98, 73–80.
Ziane, M., Desriac, N., Le Chevalier, P., Couvert, O., Moussa-Boudjemaa, B., & Leguerinel, I. (2014). Identification, heat resistance and growth potential of mesophilic spore-forming bacteria isolated from Algerian retail packaged couscous. Food Control, 45, 16–21.
Nakada, E., Asada, Y., Arai, T., & Miyake, J. (1995). Light penetration into cell suspensions of photosynthetic bacteria and relation to hydrogen production. Journal of Fermentation and Bioengineering, 80, 53–57.
Guo, C. L., Zhu, X., Liao, Q., Wang, Y. Z., Chen, R., & Lee, D. J. (2011). Enhancement of photo-hydrogen production in a biofilm photobioreactor using optical fiber with additional rough surface. Bioresource Technology, 102, 8507–8513.
Li, H. Z. (1999). Bubbles in non-Newtonian fluids: formation, interactions and coalescence. Chemical Engineering Science, 54, 2247–2254.
Li, H. Z., Mouline, Y., Choplin, L., & Midoux, N. (1997). Chaotic bubble coalescence in non-Newtonian fluids. International Journal of Multiphase Flow, 23, 713–723.
Kulkarni, A. A., & Joshi, J. B. (2005). Bubble formation and bubble rise velocity in gas-liquid systems: a review. Industrial and Engineering Chemistry Research, 44, 5873–5931.
Clark, I. C., Zhang, R. H., & Upadhyaya, S. K. (2012). The effect of low pressure and mixing on biological hydrogen production via anaerobic fermentation. International Journal of Hydrogen Energy, 37, 11504–11513.
Ma, C., Wang, X., Guo, L., Wu, X., & Yang, H. (2012). Enhanced photo-fermentative hydrogen production by Rhodobacter capsulatus with pigment content manipulation. Bioresource Technology, 118, 490–495.
Eltsova, Z., Vasilieva, L., & Tsygankov, A. (2010). Hydrogen production by recombinant strains of Rhodobacter sphaeroides using a modified photosynthetic apparatus. Applied Biochemistry and Microbiology, 46, 487–491.
Tian, X., Liao, Q., Liu, W., Wang, Y. Z., Zhu, X., Li, J., & Wang, H. (2009). Photo-hydrogen production rate of a PVA-boric acid gel granule containing immobilized photosynthetic bacteria cells. International Journal of Hydrogen Energy, 34, 4708–4717.
Sevinç, P., Gündüz, U., Eroglu, I., & Yücel, M. (2012). Kinetic analysis of photosynthetic growth, hydrogen production and dual substrate utilization by Rhodobacter capsulatus. International Journal of Hydrogen Energy, 37, 16430–16436.
Androga, D. D., Sevinç, P., Koku, H., Yücel, M., Gündüz, U., & Eroglu, I. (2012). Optimization of temperature and light intensity for improved photofermentative hydrogen production using Rhodobacter capsulatus DSM 1710. International Journal of Hydrogen Energy, 39, 2472–2480.
Basak, N., Jana, A. K., & Das, D. (2012). Optimization of molecular hydrogen production by Rhodobacter sphaeroides O.U.001 in the annular photobioreactor using response surface methodology. International Journal of Hydrogen Energy, 39, 11889–11901.
Cai, J., & Wang, G. (2012). Hydrogen production by a marine photosynthetic bacterium, Rhodovulum sulfidophilum P5, isolated from a shrimp pond. International Journal of Hydrogen Energy, 37, 15070–15080.
He, D., Bultel, Y., Magnin, J. P., & Willison, J. C. (2006). Kinetic analysis of photosynthetic growth and photohydrogen production of two strains of Rhodobacter Capsulatus. Enzyme and Microbial Technology, 38, 253–259.
Wang, Y. Z., Liao, Q., Zhu, X., Tian, X., & Zhang, C. (2010). Characteristics of hydrogen production and substrate consumption of Rhodopseudomonas palustris CQK 01 in an immobilized-cell photobioreactor. Bioresource Technology, 101, 4034–4041.
Wang, Y. Z., Liao, Q., Zhu, X., Li, J., & Lee, D. J. (2011). Effect of culture conditions on the kinetics of hydrogen production by photosynthetic bacteria in batch culture. International Journal of Hydrogen Energy, 36, 14004–14013.
Koku, H., Gündüz, U., Eroğlu, Yücel, M., & Türker, L. (2002). Aspects of the metabolism of hydrogen production by Rhodobacter sphaeroides. International Journal of Hydrogen Energy, 27, 1315–1329.
Yang, H., Zhang, J., Wang, X., Feng, J., Yan, W., & Guo, L. (2015). Coexpression of Mo- and Fe-nitrogenase in Rhodobacter capsulatus enhanced its photosynthetic hydrogen production. International Journal of Hydrogen Energy, 40, 927–934.
Tian, X., Liao, Q., Zhu, X., Wang, Y., Zhang, P., Li, J., & Wang, H. (2010). Characteristics of a biofilm photobioreactor as applied to photo-hydrogen production. Bioresource Technology, 101, 977–983.
Patel, S. K. S., Kumar, P., & Kalia, V. C. (2012). Enhancing biological hydrogen production through complementary microbial metabolisms. International Journal of Hydrogen Energy, 37, 10590–10603.
Kim, M. S., Baek, J. S., & Lee, J. K. (2006). Comparison of H2 accumulation by Rhodobacter sphaeroides KD131 and its uptake hydrogenase and PHB synthetase deficient mutant. International Journal of Hydrogen Energy, 31, 121–127.
Patel, S. K. S., & Kalia, V. C. (2013). Integrative biological hydrogen production: an overview. Indian Journal of Microbiology, 53, 3–10.
Acknowledgments
This work was supported by the International Cooperation Projects of Shanxi Province (No. 2013081020).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, M., Lv, Y. & Liu, Y. A New Hydrogen-Producing Strain and Its Characterization of Hydrogen Production. Appl Biochem Biotechnol 177, 1676–1689 (2015). https://doi.org/10.1007/s12010-015-1845-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1845-2