Abstract
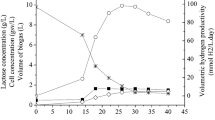
In recent years, there has been a significant deal of interest in the photo-fermentation-based synthesis of biological hydrogen due to its unique advantages, such as high hydrogen yield and the use of renewable raw materials as substrates. This paper assesses biological hydrogen production by photo-fermentation using the purple non-sulfur bacteria Rhodobacter capsulatus and Rhodospirillum rubrum cultivated in Rhodobacter capsulatus V minimal (RCV) medium. Lactose was used as a carbon source at a starting concentration of 10 g/L. An investigation of the use Rhodobacter capsulatus and Rhodospirillum rubrum strains in mixed culture confirmed the viability of the use of a co-culture system in detriment of mono-culture systems. On average, co-cultured hydrogen responses resulted in 1.43- and 1.50-fold increases in hydrogen productivity and yield, respectively, between the two species. The experiments were also conducted in a 1.5-L stirred tank reactor using repeated batch cycles, with or without medium renewal. The medium renewal allowed for the removal of organic acids produced by cellular metabolism and ensured stable and long-term hydrogen production, reaching peak productivity 278.05 mmol H2/L/day and maximum yield 19.67 mol H2/mol lactose (82% of the theoretical yield). For comparative purposes, a subsequent assay was carried out in the same initial conditions without medium renewal. An accumulation of organic acids resulted in lower peak productivity of 104.47 mmol H2/L/day and lower peak hydrogen yield of 11.71 mol H2/mol lactose confirming viability of medium renewal strategy to improve hydrogen production in photo-fermentation.





Similar content being viewed by others
References
Johnston B, Mayo MC, Khare A (2005) Hydrogen: the energy source for the 21st century. Technovation 25:569–585. https://doi.org/10.1016/j.technovation.2003.11.005
Wijayanta AT, Oda T, Purnomo CW et al (2019) Liquid hydrogen, methylcyclohexane, and ammonia as potential hydrogen storage: comparison review. Int J Hydrogen Energy 44:15026–15044. https://doi.org/10.1016/J.IJHYDENE.2019.04.112
Momirlan M, Veziroglu TN (2002) Current status of hydrogen energy. Renew Sustain Energy Rev 6:141–179. https://doi.org/10.1016/S1364-0321(02)00004-7
Sivaramakrishnan R, Shanmugam S, Sekar M et al (2021) Insights on biological hydrogen production routes and potential microorganisms for high hydrogen yield. Fuel 291. https://doi.org/10.1016/J.FUEL.2021.120136
Lo YC, Chen CY, Lee CM, Chang JS (2011) Photo fermentative hydrogen production using dominant components (acetate, lactate, and butyrate) in dark fermentation effluents. Int J Hydrogen Energy 36:14059–14068. https://doi.org/10.1016/J.IJHYDENE.2011.04.148
Srikanth S, Venkata Mohan S, Prathima Devi M et al (2009) Acetate and butyrate as substrates for hydrogen production through photo-fermentation: process optimization and combined performance evaluation. Int J Hydrogen Energy 34:7513–7522. https://doi.org/10.1016/J.IJHYDENE.2009.05.095
Keskin T, Hallenbeck PC (2012) Hydrogen production from sugar industry wastes using single-stage photofermentation. Bioresour Technol 112:131–136. https://doi.org/10.1016/J.BIORTECH.2012.02.077
Lu C, Zhang Z, Zhou X et al (2018) Effect of substrate concentration on hydrogen production by photo-fermentation in the pilot-scale baffled bioreactor. Bioresour Technol 247:1173–1176. https://doi.org/10.1016/J.BIORTECH.2017.07.122
Girelli AM, Astolfi ML, Scuto FR (2020) Agro-industrial wastes as potential carriers for enzyme immobilization: a review. Chemosphere 244:125368. https://doi.org/10.1016/J.CHEMOSPHERE.2019.125368
Kuang Y, Zhao J, Gao Y, Zhang D (2020) Enhanced hydrogen production from food waste dark fermentation by potassium ferrate pretreatment. Environ Sci Pollut Res 27:18145–18156. https://doi.org/10.1007/s11356-020-08207-3
Rezaeitavabe F, Saadat S, Talebbeydokhti N et al (2020) Enhancing bio-hydrogen production from food waste in single-stage hybrid dark-photo fermentation by addition of two waste materials (exhausted resin and biochar). Biomass Bioenergy 143:105846. https://doi.org/10.1016/J.BIOMBIOE.2020.105846
Basak N, Das D (2006) The prospect of purple non-sulfur (PNS) photosynthetic bacteria for hydrogen production: the present state of the art. World J Microbiol Biotechnol 23:31–42. https://doi.org/10.1007/S11274-006-9190-9
Wu TY, Hay JXW, Kong LB et al (2012) Recent advances in reuse of waste material as substrate to produce biohydrogen by purple non-sulfur (PNS) bacteria. Renew Sustain Energy Rev 16:3117–3122. https://doi.org/10.1016/J.RSER.2012.02.002
Zagrodnik R, Laniecki M (2015) The role of pH control on biohydrogen production by single stage hybrid dark- and photo-fermentation. Bioresour Technol 194:187–195. https://doi.org/10.1016/J.BIORTECH.2015.07.028
Moreira FS, Rodrigues MS, Sousa LM et al (2022) Single-stage repeated batch cycles using co-culture of Enterobacter cloacae and purple non-sulfur bacteria for hydrogen production. Energy 239:122465. https://doi.org/10.1016/J.ENERGY.2021.122465
Machado RG, Moreira FS, Batista FRX et al (2018) Repeated batch cycles as an alternative for hydrogen production by co-culture photofermentation. Energy 153:861–869. https://doi.org/10.1016/J.ENERGY.2018.04.101
Xiao J, Hay W, Wu TY et al (2013) Biohydrogen production through photo fermentation or dark fermentation using waste as a substrate: overview, economics, and future prospects of hydrogen usage. Biofuels, Bioprod Bioref 7:334–352. https://doi.org/10.1002/bbb.1403
Das D, Veziroglu TN (2008) Advances in biological hydrogen production processes. Int J Hydrogen Energy 33:6046–6057. https://doi.org/10.1016/J.IJHYDENE.2008.07.098
Chen Y, Yin Y, Wang J (2021) Influence of butyrate on fermentative hydrogen production and microbial community analysis. Int J Hydrogen Energy 46:26825–26833. https://doi.org/10.1016/J.IJHYDENE.2021.05.185
Noblecourt A, Christophe G, Larroche C et al (2017) High hydrogen production rate in a submerged membrane anaerobic bioreactor. Int J Hydrogen Energy 42:24656–24666. https://doi.org/10.1016/J.IJHYDENE.2017.08.037
Lopez-Hidalgo AM, Smoliński A, Sanchez A (2022) A meta-analysis of research trends on hydrogen production via dark fermentation. Int J Hydrogen Energy 47:13300–13339. https://doi.org/10.1016/J.IJHYDENE.2022.02.106
Roe AJ, O’Byrne C, McLaggan D, Booth IR (2002) Inhibition of Escherichia coli growth by acetic acid: a problem with methionine biosynthesis and homocysteine toxicity. Microbiology (N Y) 148:2215–2222. https://doi.org/10.1099/00221287-148-7-2215/CITE/REFWORKS
Noguer MC, Escudié R, Bernet N, Eric T (2022) Populational and metabolic shifts induced by acetate, butyrate and lactate in dark fermentation. Int J Hydrogen Energy 47:28385–28398. https://doi.org/10.1016/J.IJHYDENE.2022.06.163
Bundhoo MAZ, Mohee R (2016) Inhibition of dark fermentative bio-hydrogen production: a review. Int J Hydrogen Energy 41:6713–6733. https://doi.org/10.1016/J.IJHYDENE.2016.03.057
Mıynat ME, Argun H (2020) Prevention of substrate and product inhibitions by using a dilution strategy during dark fermentative hydrogen production from molasses. Int J Hydrogen Energy 45:34695–34706. https://doi.org/10.1016/J.IJHYDENE.2020.03.118
Chen Y, Yin Y, Wang J (2021) Recent advance in inhibition of dark fermentative hydrogen production. Int J Hydrogen Energy 46:5053–5073. https://doi.org/10.1016/J.IJHYDENE.2020.11.096
Weaver PF, Wall JD, Gest H (1975) Characterization of Rhodopseudomonas capsulata. Springer-Verlag
Moreira FS, Machado RG, Romão BB et al (2017) Improvement of hydrogen production by biological route using repeated batch cycles. Process Biochem 58:60–68. https://doi.org/10.1016/J.PROCBIO.2017.04.013
Silva FTM, Bessa LP, Vieira LM et al (2019) Dark fermentation effluent as substrate for hydrogen production from Rhodobacter capsulatus highlighting the performance of different fermentation systems. 3 Biotech 9(4):1–9. https://doi.org/10.1007/S13205-019-1676-X
Matsumoto M, Nishimura Y (2007) Hydrogen production by fermentation using acetic acid and lactic acid. J Biosci Bioeng 103:236–241. https://doi.org/10.1263/JBB.103.236
Akroum-Amrouche D, Abdi N, Lounici H, Mameri N (2011) Effect of physico-chemical parameters on biohydrogen production and growth characteristics by batch culture of Rhodobacter sphaeroides CIP 60.6. Appl Energy 88:2130–2135. https://doi.org/10.1016/J.APENERGY.2010.12.044
Levin DB, Pitt L, Love M (2004) Biohydrogen production: prospects and limitations to practical application. Int J Hydrogen Energy 29:173–185. https://doi.org/10.1016/S0360-3199(03)00094-6
Silva FTM, Moreira LR, de Souza FJ et al (2016) Replacement of sugars to hydrogen production by Rhodobacter capsulatus using dark fermentation effluent as substrate. Bioresour Technol 200:72–80. https://doi.org/10.1016/J.BIORTECH.2015.10.002
Deseure J, Obeid J, Willison JC, Magnin JP (2021) Reliable determination of the growth and hydrogen production parameters of the photosynthetic bacterium Rhodobacter capsulatus in fed batch culture using a combination of the Gompertz function and the Luedeking-Piret model. Heliyon 7. https://doi.org/10.1016/J.HELIYON.2021.E07394
Elkahlout K, Sagir E, Alipour S et al (2019) Long-term stable hydrogen production from acetate using immobilized Rhodobacter capsulatus in a panel photobioreactor. Int J Hydrogen Energy 44:18801–18810. https://doi.org/10.1016/J.IJHYDENE.2018.10.133
Uyar B, Muazzez, Rgan G, et al. Hydrogen production by hup 2 mutant and wild-type strains of Rhodobacter capsulatus from dark fermentation effluent of sugar beet thick juice in batch and continuous photobioreactors. https://doi.org/10.1007/s00449-015-1435-2
Dadak A, Aghbashlo M, Tabatabaei M et al (2016) Exergy-based sustainability assessment of continuous photobiological hydrogen production using anaerobic bacterium Rhodospirillum rubrum. J Clean Prod 139:157–166. https://doi.org/10.1016/J.JCLEPRO.2016.08.020
Akhlaghi N, Najafpour-Darzi G (2020) A comprehensive review on biological hydrogen production. Int J Hydrogen Energy 45:22492–22512. https://doi.org/10.1016/J.IJHYDENE.2020.06.182
Uyar B, Gürgan M, Özgür E, Gündüz U, Yücel M, Eroglu I (2015) Hydrogen production by hup 2 mutant and wild-type strains of Rhodobacter capsulatus from dark fermentation effluent of sugar beet thick juice in batch and continuous photobioreactors. Bioprocess Biosyst Eng 38:1935–1942. https://doi.org/10.1007/s00449-015-1435-2
Hu C, Choy Y, Giannis A (2010) Evaluation of lighting systems, carbon sources, and bacteria cultures on photofermentative hydrogen production. Appl Biochem Biotechnol 185:257–269. https://doi.org/10.1007/s12010-017-2655-5
Ruknongsaeng P, Reungsang A, Moonamart S, Danvirutai P (2005) Influence of nitrogen, acetate and propionate on hydrogen production from pineapple waste extract by Rhodospirillum rubrum. J Water Environ Technol 3:93–117. https://doi.org/10.2965/JWET.2005.93
Kondo T, Arakawa M, Wakayama T, Miyake J (2002) Hydrogen production by combining two types of photosynthetic bacteria with different characteristics. Int J Hydrogen Energy 27:1303–1308. https://doi.org/10.1016/S0360-3199(02)00122-2
Argun H, Kargi F, Kapdan IK (2008) Light fermentation of dark fermentation effluent for bio-hydrogen production by different Rhodobacter species at different initial volatile fatty acid (VFA) concentrations. Int J Hydrogen Energy 33:7405–7412. https://doi.org/10.1016/J.IJHYDENE.2008.09.059
Myung S, Rollin J, You C et al (2014) In vitro metabolic engineering of hydrogen production at theoretical yield from sucrose. Metab Eng 24:70–77. https://doi.org/10.1016/J.YMBEN.2014.05.006
Rai PK, Singh SP (2016) Integrated dark- and photo-fermentation: recent advances and provisions for improvement. Int J Hydrogen Energy 41:19957–19971. https://doi.org/10.1016/J.IJHYDENE.2016.08.084
Wang X, Fang Y, Wang Y et al (2018) Single-stage photo-fermentative hydrogen production from hydrolyzed straw biomass using Rhodobacter sphaeroides. Int J Hydrogen Energy 43:13810–13820. https://doi.org/10.1016/J.IJHYDENE.2018.01.057
Abo-Hashesh M, Desaunay N, Hallenbeck PC (2013) High yield single stage conversion of glucose to hydrogen by photofermentation with continuous cultures of Rhodobacter capsulatus JP91. Bioresour Technol 128:513–517. https://doi.org/10.1016/J.BIORTECH.2012.10.091
Eroglu E, Melis A (2011) Photobiological hydrogen production: recent advances and state of the art. Bioresour Technol 102:8403–8413. https://doi.org/10.1016/J.BIORTECH.2011.03.026
Boran E, Özgür E, van der Burg J et al (2010) Biological hydrogen production by Rhodobacter capsulatus in solar tubular photo bioreactor. J Clean Prod 18:S29–S35. https://doi.org/10.1016/J.JCLEPRO.2010.03.018
Kapdan IK, Kargi F, Oztekin R, Argun H (2009) Bio-hydrogen production from acid hydrolyzed wheat starch by photo-fermentation using different Rhodobacter sp. Int J Hydrogen Energy 34:2201–2207. https://doi.org/10.1016/J.IJHYDENE.2009.01.017
Keskin T, Abo-Hashesh M, Hallenbeck PC (2011) Photofermentative hydrogen production from wastes. Bioresour Technol 102:8557–8568. https://doi.org/10.1016/J.BIORTECH.2011.04.004
Davila-Vazquez G, de León-Rodríguez A, Alatriste-Mondragón F, Razo-Flores E (2011) The buffer composition impacts the hydrogen production and the microbial community composition in non-axenic cultures. Biomass Bioenergy 35:3174–3181. https://doi.org/10.1016/J.BIOMBIOE.2011.04.046
Castelló E, Nunes Ferraz-Junior AD, Andreani C et al (2020) Stability problems in the hydrogen production by dark fermentation: possible causes and solutions. Renew Sustain Energy Rev 119:109602. https://doi.org/10.1016/J.RSER.2019.109602
Vavilin VA, Rytow SV, Lokshina LY (1995) Modelling hydrogen partial pressure change as a result of competition between the butyric and propionic groups of acidogenic bacteria. Bioresour Technol 54:171–177. https://doi.org/10.1016/0960-8524(95)00127-1
van Ginkel S, Logan BE (2005) Inhibition of biohydrogen production by undissociated acetic and butyric acids. Environ Sci Technol 39:9351–9356. https://doi.org/10.1021/ES0510515
Srikanth S, Venkata Mohan S (2014) Regulating feedback inhibition caused by the accumulated acid intermediates during acidogenic hydrogen production through feed replacement. Int J Hydrogen Energy 39:10028–10040. https://doi.org/10.1016/J.IJHYDENE.2014.04.152
Romão BB, Silva FTM, Costa HC de B et al (2019) Alternative techniques to improve hydrogen production by dark fermentation. 3 Biotech 9:1–8. https://doi.org/10.1007/S13205-018-1538-Y/FIGURES/4
Sekoai PT, Ghimire A, Ezeokoli OT et al (2021) Valorization of volatile fatty acids from the dark fermentation waste streams-a promising pathway for a biorefinery concept. Renew Sustain Energy Rev 143:110971. https://doi.org/10.1016/J.RSER.2021.110971
Chalima A, Hatzidaki A, Karnaouri A, Topakas E (2019) Integration of a dark fermentation effluent in a microalgal-based biorefinery for the production of high-added value omega-3 fatty acids. Appl Energy 241:130–138. https://doi.org/10.1016/J.APENERGY.2019.03.058
Funding
The authors gratefully acknowledge Brazilian Agencies National Council for Scientific and Technological Development (CNPq), Coordination for the Improvement of Higher Education Personnel (CAPES), Research Supporting Foundation of Minas Gerais (FAPEMIG) and Vale S. A. for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ferreira, G.M.T., Moreira, F.S., Cardoso, V.L. et al. Enhancement of Photo-Fermentative Hydrogen Production with Co-culture of Rhodobacter capsulatus and Rhodospirillum rubrum by Using Medium Renewal Strategy. Bioenerg. Res. 16, 1816–1828 (2023). https://doi.org/10.1007/s12155-022-10550-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-022-10550-x