Abstract
Purpose
Determine durable effects of the 3-month Better Exercise Adherence after Treatment for Cancer (BEAT Cancer) physical activity (PA) behavior change intervention 12 months post-baseline (i.e., 9 months after intervention completion).
Methods
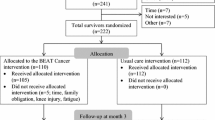
This 2-arm multicenter trial randomized 222 post-primary treatment breast cancer survivors to BEAT Cancer (individualized exercise and group education) vs. usual care (written materials). Assessments occurred at baseline, 3, 6, and 12 months, with the 12 months assessment reported here. Measures included PA (accelerometer, self-report), cardiorespiratory fitness, muscle strength, body mass index, Functional Assessment of Cancer Therapy (FACT), SF-36, fatigue, depression, anxiety, satisfaction with life, Pittsburgh Sleep Quality Index (PSQI), lower extremity joint dysfunction, and perceived memory.
Results
Adjusted linear mixed-model analyses demonstrated statistically significant month 12 between-group differences favoring BEAT Cancer for weekly minutes of moderate-to-vigorous self-report PA (mean between-group difference (M) = 44; 95% confidence interval (CI) = 12 to 76; p = .001), fitness (M = 1.5 ml/kg/min; CI = 0.4 to 2.6; p = .01), FACT-General (M = 3.5; CI = 0.7 to 6.3; p = .014), FACT-Breast (M = 3.6; CI = 0.1 to 7.1; p = .044), social well-being (M = 1.3; CI = 0.1 to 2.5; p = .037), functional well-being (M = 1.2; CI = 0.2 to 2.3; p = .023), SF-36 vitality (M = 6.1; CI = 1.4 to 10.8; p = .011), fatigue (M = − 0.7; CI = − 1.1 to − 0.2; p = .004), satisfaction with life (M = 1.9; CI = 0.3 to 3.5; p = .019), sleep duration (M = − 0.2; CI = − 0.4 to − 0.03, p = .028), and memory (M = 1.1; CI = 0.2 to 2.1; p = .024).
Conclusions
A 3-month PA intervention resulted in statistically significant and clinically important benefits compared to usual care at 12 months.
Implications for Cancer Survivors
Three months of individualized and group PA counseling causes benefits detectable 9 months later.
Trial registration
ClinicalTrials.gov NCT00929617 (https://clinicaltrials.gov/ct2/show/NCT00929617; registered June 29, 2009).




Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon request.
References
American Cancer Society. Breast cancer facts & figures 2019–2020. Atlanta: American Cancer Society Inc; 2019.
American Cancer Society. Cancer facts & figures 2021. Atlanta: American Cancer Society; 2021.
Bradshaw PT, Stevens J, Khankari N, Teitelbaum SL, Neugut AI, Gammon MD. Cardiovascular disease mortality among breast cancer survivors. Epidemiology. 2016;27:6–13.
Patnaik JL, Byers T, DiGuiseppi C, Dabelea D, Denberg TD. Cardiovascular disease competes with breast cancer as the leading cause of death for older females diagnosed with breast cancer: a retrospective cohort study. Breast Cancer Res. 2011;13:R64.
Donin N, Filson C, Drakaki A, Tan HJ, Castillo A, Kwan L, et al. Risk of second primary malignancies among cancer survivors in the United States, 1992 through 2008. Cancer. 2016;122:3075–86.
Von Ah DM, Russell KM, Carpenter J, Monahan PO, Zhao QQ, Tallman E, et al. Health-related quality of life of African American breast cancer survivors compared with healthy African American Women. Cancer Nurs. 2012;35:337–46.
Abrahams HJG, Smits L, Lugt M, Roos WK, Kamm Y, Heins MJ, et al. Severe fatigue after treatment of ductal carcinoma in situ: a comparison with age-matched breast cancer survivors and healthy controls. Breast. 2017;31:76–81.
Campbell KL, Winters-Stone KM, Wiskemann J, May AM, Schwartz AL, Courneya KS, et al. Exercise guidelines for cancer survivors: consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc. 2019;51:2375–90.
Yan AF, Wang Y, Ng AV. Physical activity and annual medical outlay in U.S. colorectal, breast and prostate cancer survivors. Prev Med Rep. 2018;9:118–23.
Arem H, Mama SK, Duan X, Rowland JH, Bellizzi KM, Ehlers DK. Prevalence of healthy behaviors among cancer survivors in the United States: how far have we come? Cancer Epidemiol Biomarkers Prev. 2020;29:1179–87.
Grimmett C, Corbett T, Brunet J, Shepherd J, Pinto BM, May CR, et al. Systematic review and meta-analysis of maintenance of physical activity behaviour change in cancer survivors. Int J Behav Nutr Phys Act. 2019;16:37.
Fjeldsoe B, Neuhaus M, Winkler E, Eakin E. Systematic review of maintenance of behavior change following physical activity and dietary interventions. Health Psychol. 2011;30:99–109.
Hartman SJ, Chen R, Tam RM, Narayan HK, Natarajan L, Liu L. Fitbit use and activity levels from intervention to 2 years after: secondary analysis of a randomized controlled trial. JMIR Mhealth Uhealth. 2022;10:e37086.
Mama SK, Lopez-Olivo MA, Bhuiyan N, Leach HJ. Effectiveness of physical activity interventions among rural cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev. 2021;30:2143–53.
Batalik L, Winnige P, Dosbaba F, Vlazna D, Janikova A. Home-based aerobic and resistance exercise interventions in cancer patients and survivors: a systematic review. Cancers (Basel). 2021;13:1915.
Abdin S, Lavallee JF, Faulkner J, Husted M. A systematic review of the effectiveness of physical activity interventions in adults with breast cancer by physical activity type and mode of participation. Psychooncology. 2019;28:1381–93.
Rogers LQ, McAuley E, Anton PM, Courneya KS, Vicari S, Hopkins-Price P, et al. Better exercise adherence after treatment for cancer (BEAT Cancer) study: rationale, design, and methods. Contemp Clin Trials. 2012;33:124–37.
Rogers LQ, Courneya KS, Anton PM, Hopkins-Price P, Verhulst S, Vicari SK, et al. Effects of the BEAT Cancer physical activity behavior change intervention on physical activity, aerobic fitness, and quality of life in breast cancer survivors: a multicenter randomized controlled trial. Breast Cancer Res Treat. 2015;149:109–19.
Rogers LQ, Courneya KS, Anton PM, Verhulst S, Vicari SK, Robbs RS, et al. Effects of a multicomponent physical activity behavior change intervention on fatigue, anxiety, and depressive symptomatology in breast cancer survivors: randomized trial. Psychooncology. 2017;26:1901–6.
Rogers LQ, Courneya KS, Carter SJ, Anton PM, Verhulst S, Vicari SK, et al. Effects of a multicomponent physical activity behavior change intervention on breast cancer survivor health status outcomes in a randomized controlled trial. Breast Cancer Res Treat. 2016;159:283–91.
Ehlers DK, Rogers LQ, Courneya KS, Robbs RS, McAuley E. Effects of BEAT Cancer randomized physical activity trial on subjective memory impairments in breast cancer survivors. Psychooncology. 2018;27:687–90.
Rogers LQ, Courneya KS, Oster RA, Anton PM, Robbs RS, Forero A, et al. Physical activity and sleep quality in breast cancer survivors: a randomized trial. Med Sci Sports Exerc. 2017;49:2009–15.
Freedson PS, Melanson E, Sirard J. Calibration of the Computer Science and Applications. Inc accelerometer Med Sci Sports Exerc. 1998;30:777–81.
Sirard JR, Melanson EL, Li L, Freedson PS. Field evaluation of the Computer Science and Applications, Inc. physical activity monitor. Med Sci Sports Exerc. 2000;32:695–700.
Godin G, Jobin J, Bouillon J. Assessment of leisure time exercise behavior by self-report: a concurrent validity study. Can J Public Health. 1986;77:359–62.
Piercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, et al. The physical activity guidelines for Americans. JAMA. 2018;320:2020–8.
Wasserman K, Hansen JE, Sue DY, Stringer WW, Whipp BJ. Principles of exercise testing and interpretation: including pathophysiology and clinical applications. Med Sci Sports Exerc. 2005;37:1249.
Brady MJ, Cella DF, Mo F, Bonomi AE, Tulsky DS, Lloyd SR, et al. Reliability and validity of the Functional Assessment of Cancer Therapy-Breast quality-of-life instrument. J Clin Oncol. 1997;15:974–86.
Ware JE, Kosinski MA, Dewey JE. How to score version 2 of the SF-36® health survey. Lincoln, RI: Quality Metric Incorporated; 2000.
Hann DM, Jacobsen PB, Azzarello LM, Martin SC, Curran SL, Fields KK, et al. Measurement of fatigue in cancer patients: development and validation of the Fatigue Symptom Inventory. Qual Life Res. 1998;7:301–10.
Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67:361–70.
Diener E, Emmons RA, Larsen RJ, Griffin S. The satisfaction with life scale. J Pers Assess. 1985;49:71–5.
Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213.
Tryon WW. Nocturnal activity and sleep assessment. Clin Psychol Rev. 1996;16:197–213.
Rogers JC, Irrang JJ. Measures of adult lower extremity function. Arthritis Rheum. 2003;49:S67–84.
Zelinski EM, Gilewski MJ. A 10-item Rasch modeled memory self-efficacy scale. Aging Ment Health. 2004;8:293–306.
Cohen J. Statistical power analysis for the behavioral sciences. Hillsdale, N.J.: L. Erlbaum Associates; 1988.
Li T, Wei S, Shi Y, Pang S, Qin Q, Yin J, et al. The dose-response effect of physical activity on cancer mortality: findings from 71 prospective cohort studies. Br J Sports Med. 2016;50:339–45.
Peddle-McIntyre CJ, Cavalheri V, Boyle T, McVeigh JA, Jeffery E, Lynch BM, et al. A review of accelerometer-based activity monitoring in cancer survivorship research. Med Sci Sports Exerc. 2018;50:1790–801.
Guida JL, Agurs-Collins T, Ahles TA, Campisi J, Dale W, Demark-Wahnefried W, et al. Strategies to prevent or remediate cancer and treatment-related aging. J Natl Cancer Inst. 2021;113:112–22.
U.S. Department of Health and Human Services. Physical activity and health: a report of the surgeon general. Washington D.C. 1996. https://profiles.nlm.nih.gov/101584932X106.
Yost KJ, Eton DT. Combining distribution- and anchor-based approaches to determine minimally important differences: the FACIT experience. Eval Health Prof. 2005;28:172–91.
Bjorner JB, Wallenstein GV, Martin MC, Lin P, Blaisdell-Gross B, Tak Piech C, et al. Interpreting score differences in the SF-36 Vitality scale: using clinical conditions and functional outcomes to define the minimally important difference. Curr Med Res Opin. 2007;23:731–9.
Servaes P, Verhagen C, Bleijenberg G. Fatigue in cancer patients during and after treatment: prevalence, correlates and interventions. Eur J Cancer. 2002;38:27–43.
Donovan KA, Jacobsen PB, Small BJ, Munster PN, Andrykowski MA. Identifying clinically meaningful fatigue with the Fatigue Symptom Inventory. J Pain Symptom Manage. 2008;36:480–7.
Hollen PJ, Msaouel P, Gralla RJ. Determining issues of importance for the evaluation of quality of life and patient-reported outcomes in breast cancer: results of a survey of 1072 patients. Breast Cancer Res Treat. 2015;151:679–86.
Kim SH, Son BH, Hwang SY, Han W, Yang JH, Lee S, et al. Fatigue and depression in disease-free breast cancer survivors: prevalence, correlates, and association with quality of life. J Pain Symptom Manage. 2008;35:644–55.
Jones JM, Olson K, Catton P, Catton CN, Fleshner NE, Krzyzanowska MK, et al. Cancer-related fatigue and associated disability in post-treatment cancer survivors. J Cancer Surviv. 2016;10:51–61.
Olson EA, Mullen SP, Rogers LQ, Courneya KS, Verhulst S, McAuley E. Meeting physical activity guidelines in rural breast cancer survivors. Am J Health Behav. 2014;38:890–9.
McAuley E, White SM, Rogers LQ, Motl RW, Courneya KS. Physical activity and fatigue in breast cancer and multiple sclerosis: psychosocial mechanisms. Psychosom Med. 2010;72:88–96.
Rogers LQ, McAuley E, Courneya KS, Verhulst SJ. Correlates of physical activity self-efficacy among breast cancer survivors. Am J Health Behav. 2008;32:594–603.
Xu S, Thompson W, Ancoli-Israel S, Liu L, Palmer B, Natarajan L. Cognition, quality-of-life, and symptom clusters in breast cancer: using Bayesian networks to elucidate complex relationships. Psychooncology. 2018;27:802–9.
Rogers LQ, Goncalves L, Martin MY, Pisu M, Smith TL, Hessong D, et al. Beyond efficacy: a qualitative organizational perspective on key implementation science constructs important to physical activity intervention translation to rural community cancer care sites. J Cancer Surviv. 2019;13:537–46.
Gomersall SR, Skinner TL, Winkler E, Healy GN, Eakin E, Fjeldsoe B. Feasibility, acceptability and efficacy of a text message-enhanced clinical exercise rehabilitation intervention for increasing ‘whole-of-day’ activity in people living with and beyond cancer. BMC Public Health. 2019;19:542.
Jankowski CM, Ory MG, Friedman DB, Dwyer A, Birken SA, Risendal B. Searching for maintenance in exercise interventions for cancer survivors. J Cancer Surviv. 2014;8:697–706.
Acknowledgements
We thank the study participants, staff, and trainees who contributed their time and effort to helping make this project possible. We also thank Lingling Wang for assisting with the data presentation for this manuscript. Results have been presented clearly, honestly, and without fabrication, falsification, or inappropriate data manipulation.
Funding
This work was supported by the National Institutes of Health (R01CA136859 and P30DK056336).
Author information
Authors and Affiliations
Contributions
LQR, KSC, PMA, and EM made substantial contributions to the conception and design of the work. RO, PMA, SP, and DE made substantial contributions to the data acquisition. LQR, KSC, and RO made substantial contributions to the data analysis. All authors made substantial contributions to data interpretation. LQR wrote the initial manuscript draft with the remaining coauthors providing substantial contributions to subsequent drafts. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All study procedures involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its amendments or comparable ethical standards. The proposal was approved by the University of Alabama at Birmingham, the University of Illinois at Urbana-Champaign, and Southern Illinois University School of Medicine Institutional Review Boards for Human Use (protocol numbers F121114008, 09707, and 08–022, respectively). Informed consent was obtained from all participants included in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rogers, L.Q., Courneya, K.S., Oster, R.A. et al. Physical activity intervention benefits persist months post-intervention: randomized trial in breast cancer survivors. J Cancer Surviv 17, 1834–1846 (2023). https://doi.org/10.1007/s11764-022-01329-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-022-01329-2