Abstract
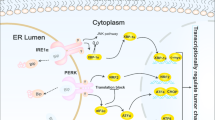
Unfolded protein response (UPR) pathway is a promising target for cancer treatment because of its over-activation in different cancers and its role in tumorigenesis and chemotherapeutic drug resistance. Endoplasmic Reticulum Metallo Protease 1 (ERMP1) is overexpressed in cancers such as colorectal cancer. The ERMP1 role in UPR activation was previously reported in breast cancer. We aimed to investigate the ERMP1 role in the UPR activation in colorectal cancer. In this regard, ERMP1 gene was silenced in colorectal cancer HCT116 cell line using specific small hairpin RNA (shRNA). Then, UPR associated protein markers including inositol requiring enzyme 1 (IRE1α), activating transcription factor 6 (ATF-6), eukaryotic initiation factor 2α (eIF2α) and phosphorylated eIF2α (P- eIF2α) were evaluated using western blot. We found that ERMP1 gene expression and all of the above UPR associated protein markers were significantly decreased after ERMP1 gene silencing. Therefore, it seems that ERMP1 plays an important role in UPR activation. Since the overexpression of ERMP1 as a potential oncogene can highly activate the UPR pathway in colorectal cancer, it can be considered as a promising target for colorectal cancer treatment. However, further investigations are required to confirm these findings.




Similar content being viewed by others
Data availability
We make sure that all data and materials support our published claims and comply with field standards.
Abbreviations
- ATF-6:
-
Activating transcription factor 6
- BiP:
-
Binding immunoglobulin protein
- CSC:
-
Cancer stem cell
- CRC:
-
Colorectal cancer
- ER:
-
Endoplasmic reticulum
- ERMP1:
-
Endoplasmic reticulum metallo protease 1
- eIF2α:
-
Eukaryotic initiation factor 2α
- Fxna:
-
Felix-ina
- GRP78:
-
Glucose-regulated protein 78
- HRP:
-
Horseradish peroxidase
- IRE1α:
-
Inositol requiring enzyme 1
- PBS:
-
Phosphate-buffered saline
- P- eIF2α:
-
Phosphorylated eIF2α
- PERK:
-
Protein kinase RNA-like ER kinase
- ROS:
-
Reactive oxygen species
- SDS-PAGE:
-
Sodium dodecyl sulfate-polyacrylamide
- shRNA:
-
Specific small hairpin RNA
- UPR:
-
Unfolded protein response
- XBP1:
-
X-box-binding protein-1
References
Ahmed D, Eide PW, Eilertsen IA, Danielsen SA, Eknæs M, Hektoen M, Lind GE, Lothe RA (2013) Epigenetic and genetic features of 24 colon cancer cell lines. Oncogenesis 2(9):e71–e71. https://doi.org/10.1038/oncsis.2013.35
Akbari-Birgani S, Paranjothy T, Zuse A, Janikowski T, Cieślar-Pobuda A, Likus W, Urasińska E, Schweizer F, Ghavami S, Klonisch T, Łos MJ (2016) Cancer stem cells, cancer-initiating cells and methods for their detection. Drug Discov Today 21(5):836–842. https://doi.org/10.1016/j.drudis.2016.03.004
Bernards R (2008) Cancer: entangled pathways. Nature 455(7212):479. https://doi.org/10.1038/455479a
Bhat TA, Chaudhary AK, Kumar S, O’Malley J, Inigo JR, Kumar R, Yadav N, Chandra D (2017) Endoplasmic reticulum-mediated unfolded protein response and mitochondrial apoptosis in cancer. Biochim Biophys Acta Rev Cancer 1867(1):58–66. https://doi.org/10.1016/j.bbcan.2016.12.002
Blais JD, Addison CHL, Edge R, Falls T, Zhao H, Wary K, Koumenis C, Harding HP, Ron D, Holcik M, Bell JC (2006) Perk-dependent translational regulation promotes tumor cell adaptation and angiogenesis in response to hypoxic stress. Mol Cell Biol 26(24):9517–9532. https://doi.org/10.1128/MCB.01145-06
Coleman OI, Lobner EM, Bierwirth S, Sorbie A, Waldschmitt N, Rath E, Berger E, Lagkouvardos I, Clavel T, KD MC, Weber A, Heikenwalder M, Janssen KP, Haller D (2018) Activated ATF6 induces intestinal dysbiosis and innate immune response to promote colorectal tumorigenesis. Gastroenterology 155(5):1539–1552. e12. https://doi.org/10.1053/j.gastro.2018.07.028
Corazzari M, Gagliardi M, Fimia GM, Piacentini M (2017) Endoplasmic reticulum stress, unfolded protein response, and cancer cell fate. Front Oncol 7:78. https://doi.org/10.3389/fonc.2017.00078
Cybulsky AV (2017) Endoplasmic reticulum stress, the unfolded protein response and autophagy in kidney diseases. Nat Rev Nephrol 13(11):681–696. https://doi.org/10.1038/nrneph.2017.129
Davenport EL, Morgan GJ, Davies FE (2008) Untangling the unfolded protein response. Cell Cycle 7(7):865–869. https://doi.org/10.4161/cc.7.7.5615
Dikic I, Elazar Z (2018) Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol 19(6):349. https://doi.org/10.1038/s41580-018-0003-4
Fujimoto T, Yoshimatsu K, Watanabe K, Yokomizo H, Otani T, Matsumoto A, Osawa G, Onda M, Ogawa K (2007) Overexpression of human X-box binding protein 1 (XBP-1) in colorectal adenomas and adenocarcinomas. Anticancer Res 27(1A):127–131
Garcia-Rudaz C, Luna F, Tapia V, Kerr B, Colgin L, Galimi F, Dissen GA, Rawlings ND, Ojeda SR (2007) Fxna, a novel gene differentially expressed in the rat ovary at the time of folliculogenesis, is required for normal ovarian histogenesis. Development 134(5):945–957. https://doi.org/10.1242/dev.02795
Ghavami S, Eshraghi M, Kadkhoda K, Mutawe MM, Maddika S, Bay GH, Wesselborg S, Halayko AJ, Klonisch T, Los M (2009) Role of BNIP3 in TNF-induced cell death--TNF upregulates BNIP3 expression. Biochim Biophys Acta 1793(3):546–560. https://doi.org/10.1016/j.bbamcr.2009.01.002
Ghavami S, Mutawe MM, Hauff K, Stelmack GL, Schaafsma D, Sharma P, McNeill KD, Hynes TS, Kung SK, Unruh H, Klonisch T, Hatch GM, Los M, Halayko AJ (2010) Statin-triggered cell death in primary human lung mesenchymal cells involves p53-PUMA and release of Smac and Omi but not cytochrome c. Biochim Biophys Acta 1803(4):452–467. https://doi.org/10.1016/j.bbamcr.2009.12.005
Glembotski CC (2008) The role of the unfolded protein response in the heart. J Mol Cell Cardiol 44(3):453–459. https://doi.org/10.1016/j.yjmcc.2007.10.017
Grandi A, Santi A, Campagnoli S, Parri M, De Camilli E, Song C, Jin B, Lacombe A, Castori-Eppenberger S, Sarmientos P, Grandi G, Viale G, Terracciano L, Chiarugi P, Pileri P, Grifantini R (2016) ERMP1, a novel potential oncogene involved in UPR and oxidative stress defense, is highly expressed in human cancer. Oncotarget 7(39):63596. https://doi.org/10.18632/oncotarget.11550
Grootjans J, Kaser A, Kaufman RJ, Blumberg RS (2016) The unfolded protein response in immunity and inflammation. Nat Rev Immunol 16(8):469. https://doi.org/10.1038/nri.2016.62
Gupta SS, Zeglinski MR, Rattan SG, Landry NM, Ghavami S, Wigle JT, Klonisch T, Halayko AJ, Dixon IMC (2016) Inhibition of autophagy inhibits the conversion of cardiac fibroblasts to cardiac myofibroblasts. Oncotarget 7(48):78516–78531. https://doi.org/10.18632/oncotarget.12392
Hetz C (2012) The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 13(2):89. https://doi.org/10.1038/nrm3270
Hetz Flores C, Chevet E, Harding HP (2013) Targeting the unfolded protein response in disease. Nat Rev Drug Discov 12(9):703–719. https://doi.org/10.1038/nrd3976
Jin C, Jin Z, Chen N-z LM, Liu C-b HW-L, Zheng C-g (2016) Activation of IRE1α-XBP1 pathway induces cell proliferation and invasion in colorectal carcinoma. Biochem Biophys Res Commun 470(1):75–81. https://doi.org/10.1016/j.bbrc.2015.12.119
Kavousipour S, Khademi F, Zamani M, Vakili B, Mokarram P (2017) Novel biotechnology approaches in colorectal cancer diagnosis and therapy. Biotechnol Lett 39(6):785–803. https://doi.org/10.1007/s10529-017-2303-8
Kim Y, Santos R, Gage FH, Marchetto MC (2017) Molecular mechanisms of bipolar disorder: progress made and future challenges. Front Cell Neurosci 11:30. https://doi.org/10.3389/fncel.2017.00030
Kroeger H, Chiang WCH, Felden J, Nguyen A, Lin JH (2019) ER stress and unfolded protein response in ocular health and disease. FEBS J 286(2):399–412. https://doi.org/10.1111/febs.14522
Kumar R, Krause GS, Yoshida H, Mori K, DeGracia DJ (2003) Dysfunction of the unfolded protein response during global brain ischemia and reperfusion. J Cereb Blood Flow Metab 23(4):462–471. https://doi.org/10.1097/01.WCB.0000056064.25434.CA
Kuroda H, Kutner RH, Bazan NG, Reiser J (2009) Simplified lentivirus vector production in protein-free media using polyethylenimine-mediated transfection. J Virol Methods 157(2):113–121. https://doi.org/10.1016/j.jviromet.2008.11.021
Lee J, Ozcan U (2014) Unfolded protein response signaling and metabolic diseases. J Biol Chem 289(3):1203–1211. https://doi.org/10.1074/jbc.R113.534743
Levy JMM, Towers CG, Thorburn A (2017) Targeting autophagy in cancer. Nat Rev Cancer 17(9):528. https://doi.org/10.1038/nrc.2017.53
Likus W, Siemianowicz K, Bie’nkc K, Pakuład M, Pathake H, Duttaf CH, Wangg Q, Shojaeih SH, Assarafi YG, Ghavami S, Cie’slar-Pobudal A, Łos MJ (2016) Could drugs inhibiting the mevalonate pathway also target cancer stem cells? Drug Resist Update 25:13–25. https://doi.org/10.1016/j.drup.2016.02.001
Lindholm D, Korhonen L, Eriksson O, Kõks S (2017) Recent insights into the role of unfolded protein response in ER stress in health and disease. Front Cell Dev Biol 5:48. https://doi.org/10.3389/fcell.2017.00048
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Niederreiter L, Fritz TMJ, Adolph TE, Krismer AM, Offner FA, Tschurtschenthaler M, Flak MB, Hosomi SH, Tomczak MF, Kaneider NC, Sarcevic E, Kempster SL, Raine T, Esser D, Rosenstiel PH, Kohno K, Iwawaki T, Tilg H, Blumberg RS, Kaser A (2013) ER stress transcription factor Xbp1 suppresses intestinal tumorigenesis and directs intestinal stem cells. J Exp Med 210(10):2041–2056. https://doi.org/10.1084/jem.20122341
Osorio F, Lambrecht B, Janssens S (2013) The UPR and lung disease. Semin Immunopathol 35:293–306. https://doi.org/10.1007/s00281-013-0368-6
Qu J, Zhang L, Li L, Su Y (2018) miR-148b functions as a tumor suppressor by targeting endoplasmic reticulum metallo protease 1 in human endometrial cancer cells. Oncol Res 27(1):81–88. https://doi.org/10.3727/096504018X15202988139874
Roller C, Maddalo D (2013) The molecular chaperone GRP78/BiP in the development of chemoresistance: mechanism and possible treatment. Front Pharmacol 4:10. https://doi.org/10.3389/fphar.2013.00010
Ron D, Walter P (2007) Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol 8(7):519. https://doi.org/10.1038/nrm2199
Rouschop KM, Dubois LJ, Keulers TG, Beucken TVD, Lambin PH, Bussink J, Kogel AJVD, Koritzinsky M, Wouters BG (2013) PERK/eIF2α signaling protects therapy resistant hypoxic cells through induction of glutathione synthesis and protection against ROS. Proc Natl Acad Sci U S A 110(12):4622–4627. https://doi.org/10.1073/pnas.1210633110
Saito S, Furuno A, Sakurai J, Sakamoto A, Park HR, Shinya K, Tsuruo T, Tomida A (2009) Chemical genomics identifies the unfolded protein response as a target for selective cancer cell killing during glucose deprivation. Cancer Res 69(10):4225–4234. https://doi.org/10.1158/0008-5472.CAN-08-2689
Scheper W, Hoozemans JJ (2015) The unfolded protein response in neurodegenerative diseases: a neuropathological perspective. Acta Neuropathol 130(3):315–331. https://doi.org/10.1007/s00401-015-1462-8
Schwarz DS, Blower MD (2016) The endoplasmic reticulum: structure, function and response to cellular signaling. Cell Mol Life Sci 73(1):79–94. https://doi.org/10.1007/s00018-015-2052
Shapiro DJ, Livezey M, Yu L, Zheng X, Andruska N (2016) Anticipatory UPR activation: a protective pathway and target in cancer. Trends Endocrinol Metab 27(10):731–741. https://doi.org/10.1016/j.tem.2016.06.002
Shirazi F, Jones RJ, Singh RK, Zou J, Kuiatse I, Berkova Z, Wang H, Lee HC, Hong S, Dick L, Chattopadhyay N, Orlowski RZ (2020) Activating KRAS, NRAS, and BRAF mutants enhance proteasome capacity and reduce endoplasmic reticulum stress in multiple myeloma. Proc Natl Acad Sci 117(33):20004–20014. https://doi.org/10.1073/pnas.2005052117
Spaan CN, Smit WL, de Jeude JFVL, Meijer BJ, Muncan V, van den Brink GR, Heijmans J (2019) Expression of UPR effector proteins ATF6 and XBP1 reduce colorectal cancer cell proliferation and stemness by activating PERK signaling. Cell Death Dis 10(7):1–10. https://doi.org/10.1038/s41419-019-1729-4
Suh DH, Kim MK, Kim HS, Chung HH, Song YS (2012) Unfolded protein response to autophagy as a promising druggable target for anticancer therapy. Ann N Y Acad Sci 1271(1):20–32. https://doi.org/10.1111/j.1749-6632.2012.06739.x
Walczak A, Gradzik K, Kabzinski J, Przybylowska-Sygut K, Majsterek I (2019) The role of the ER-induced UPR pathway and the efficacy of its inhibitors and inducers in the inhibition of tumor progression. Oxidative Med Cell Longev 2019:5729710. https://doi.org/10.1155/2019/5729710
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334(6059):1081–1086. https://doi.org/10.1126/science.1209038
Wu J, Liu S, Liu G, Dombkowski A, Abrams J, Martin-Trevino R, Wicha MS, Ethier SP, Yang ZQ (2012) Identification and functional analysis of 9p24 amplified genes in human breast cancer. Oncogene 31(3):333. https://doi.org/10.1038/onc.2011.227
Xing X, Lai M, Wang Y, Xu E, Huang Q (2006) Overexpression of glucose-regulated protein 78 in colon cancer. Clin Chim Acta 364(1–2):308–315. https://doi.org/10.1016/j.cca.2005.07.016
Yang Y, Cheung HH, Tu J, Miu KK, Chan WY (2016) New insights into the unfolded protein response in stem cells. Oncotarget 7(33):54010–54027. https://doi.org/10.18632/oncotarget.9833
Zheng X, Andruska N, Yu L, Mao CH, Kim JE, Livezey M, Helferich WG, Shapiro DJ (2016) Interplay between steroid hormone activation of the unfolded protein response and nuclear receptor action. Steroids 114:2–6. https://doi.org/10.1016/j.steroids.2016.03.014
Zhou AX, Tabas I (2013) The UPR in atherosclerosis. Semin Immunopathol 35:321–332. https://doi.org/10.1007/s00281-013-0372-x
Acknowledgments
The authors would like to express their sincere gratitude to Shiraz University of Medical Sciences for financially supporting the study (No. 16117). The authors would also like to acknowledge, Ms. Sarah Masoumi, English editor and internal manager of the Iranian Journal of Blood and Cancer for her valuable assistance in English editing of this manuscript.
Code availability
Not Applicable.
Funding
This work was financially supported by Shiraz University of Medical Sciences (Grant No.16117).
Author information
Authors and Affiliations
Contributions
M.Z and P.M coordinated and designed the study. M.Z acquired funding. S.D. and M.E. conducted the experiments. M. Z, P.M, S.Gh and S.V.H analyzed and interpreted the data. M.Z and S.D. wrote the manuscript, P.M. and S.Gh, and SV.H revised and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All the authors agree to participate in this work.
Consent for publication
All the authors consent to publish this manuscript in Biologia.
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dastghaib, S., Mokarram, P., Erfani, M. et al. Endoplasmic reticulum Metallo protease 1, a triggering factor for unfolded protein response and promising target in colorectal cancer. Biologia 76, 2403–2411 (2021). https://doi.org/10.1007/s11756-021-00769-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00769-y