Abstract
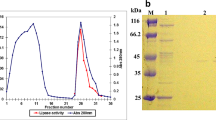
An extracellular lipase (EC 3.1.1.3, AN0512Lip) from Aspergillus niger AN0512 was purified and its characteristics were investigated. After the process of ammonium sulfate precipitation followed by ion-exchange chromatography and gel filtration, the purified lipase was achieved with 203.6-fold purification and 22.1 % recovery. AN0512Lip exhibited the highest activity at 50 °C and pH 5.0. It was thermostable and pH-stable, as indicated by that more than 50 % activity retained at 60 °C for 20 h and more than 90 % activity retained at pH 3.0 for 20 h, respectively. AN0512Lip activity was stimulated by some divalent metal ions (especially Cu2+, Ca2+), while greatly suppressed by EDTA, indicating that AN0512Lip was a metal-activated enzyme. Moreover, AN0512Lip exhibited high tolerance for various polar organic solvents with log P < 0.8, and the highest lipase activity (476 % of its original activity) was achieved after addition of 90 % (V/V) isopropanol to the reaction mixture. AN0512Lip also displayed 3-regiospecificity and great affinity for the long-chain fatty ester. The preliminary test showed that AN0512Lip was a candidate for enriching EPA and DHA in fish oil. All the unique properties, such as thermostability, Cu2+-dependent, 3-regiospecificity, and polar organic solvent-tolerance, indicated that AN0512Lip could have potential applications in the food industry, even in organic synthesis and the pharmaceutical industry.






Similar content being viewed by others
Abbreviations
- AN0512Lip:
-
Lipase from Aspergillus niger AN0512
- DH:
-
Degree of hydrolysis
- EDTA:
-
Ethylene diamine tetraacetic acid
- K m :
-
Michaelis constant
- PMSF:
-
Phenylmethanesulfonyl fluoride
- p-NPP:
-
p-Nitrophenyl palmitate
- p-NPD:
-
p-Nitrophenyl dodecanoate
- p-NPO:
-
p-Nitrophenyl octanoate
- p-NPA:
-
p-Nitrophenyl acetate
- SDS:
-
Sodium dodecyl sulfonate
- TLC:
-
Thin-layer chromatography
- V max :
-
The maximum velocity for the reaction
References
Ellaiah P, Prabhakar T, Ramakrishna B, Taleb AT, Adinarayana K (2004) Production of lipase by immobilized cells of Aspergillus niger. Process Biochem 39:525–528
Hasan F, Shah AA, Hameed A (2006) Industrial applications of microbial lipases. Enzyme Microb Technol 39:235–251
Gutarra ML, Godoy MG, Maugeri F, Rodrigues MI, Freire DM, Castilho LR (2009) Production of an acidic and thermostable lipase of the mesophilic fungus Penicillium simplicissimum by solid-state fermentation. Bioresour Technol 100:5249–5254
Carvalho PDO, Contesini FJ, Bizaco R, Macedo GA (2005) Kinetic properties and enantioselectivity of the lipases produced by four Aspergillus species. Food Biotechnol 19:183–192
Singh AK, Mukhopadhyay M (2012) Overview of fungal lipase: a review. Appl Biochem Biotechnol 166:486–520
Romero CM, Pera LM, Loto F, Vallejos C, Castro G, Baigori MD (2012) Purification of an organic solvent-tolerant lipase from Aspergillus niger MYA 135 and its application in ester synthesis. Biocatal Agric Biotechnol 1:25–31
Yamaguchi S, Mase T (1991) Purification and characterization of mono- and diacylglycerol lipase isolated from Penicillium camembertii U-150. Appl Microbiol Biotechnol 34:720–725
Abel Hiol MDJ, Druet D, Comeau L (1999) Production, purification and characterization of an extracellular lipase from Mucor hiemalis f. hiemalis. Enzyme Microb Technol 25:80–87
Rodrigues RC, Fernandez-Lafuente R (2010) Lipase from Rhizomucor miehei as an industrial biocatalyst in chemical process. J Mol Catal B Enzym 64:1–22
Saxena RK, Davidson WS, Sheoran A, Giri B (2003) Purification and characterization of an alkaline thermostable lipase from Aspergillus carneus. Process Biochem 39:239–247
Mateos Diaz JC, Rodríguez JA, Roussos S, Cordova J, Abousalham A, Carriere F, Baratti J (2006) Lipase from the thermotolerant fungus Rhizopus homothallicus is more thermostable when produced using solid state fermentation than liquid fermentation procedures. Enzyme Microb Technol 39:1042–1050
Timberlake WE, Marshall MA (1989) Genetic engineering of filamentous fungi. Science 244:1313–1317
Contesini FJ, Lopes DB, Macedo GA, Nascimento M, Carvalho P (2010) Aspergillus sp. lipase: potential biocatalyst for industrial use. J Mol Catal B Enzym 67:163–171
Schuster E, Dunn-Coleman N, Frisvad JC, Van Dijck PW (2002) On the safety of Aspergillus niger—a review. Appl Microbiol Biotechnol 59:426–435
Zamost B, Nielsen H, Starnes R (1991) Thermostable enzymes for industrial applications. J Ind Microbiol 8:71–81
Todida J, Arikawa Y, Kondou K, Fukuzawa M, Sekiguchi J (1998) Purification and characterization of triacylglycerol lipase from Aspergillus oryzae. Biosci Biotechnol Biochem 62:759–763
Mander P, Cho SS, Simkhada JR, Choi YH, Park DJ, Yoo JC (2012) An organic solvent-tolerant lipase from Streptomyces sp. CS133 for enzymatic transesterification of vegetable oils in organic media. Process Biochem 47:635–642
Pokorny D, Cimerman A, Steiner W (1997) Aspergillus niger lipases: induction, isolation and characterization of two lipases from a MZKI Al 16 strain. J Mol Catal B Enzym 2:215–222
Romero CM, Pera LM, Olivaro C, Vazquez A, Baigori MD (2012) Tailoring chain length selectivity of a solvent-tolerant lipase activity from Aspergillus niger MYA 135 by submerged fermentation. Fuel Process Technol 98:23–29
Haki GD, Rakshit SK (2003) Developments in industrially important thermostable enzymes: a review. Bioresour Technol 89:17–34
Uttatree S, Winayanuwattikun P, Charoenpanich J (2010) Isolation and characterization of a novel thermophilic-organic solvent stable lipase from Acinetobacter baylyi. Appl Biochem Biotechnol 162:1362–1376
Sun T, Pigott GM, Herwig RP (2002) Lipase-assisted concentration of n-3 polyunsaturated fatty acids from viscera of farmed Atlantic salmon (Salmo salar L.). J Food Sci 67:130–136
Bispo P, Batista I, Bernardino RJ, Bandarra NM (2014) Preparation of triacylglycerols rich in omega-3 fatty acids from sardine oil using a Rhizomucor miehei lipase: focus in the EPA/DHA ratio. Appl Biochem Biotech 172:1866–1881
Guil-Guerrero JL, López-Martínez JC, Rincón-Cervera MA, Campra-Madrid P (2007) One-step extraction and concentration of polyunsaturated fatty acids from fish liver. J Am Oil Chem Soc 84:357–361
Hu S, Wei H, Guo S, Li L, Hou Y (2011) Flavor evaluation of yak butter in Tsinghai-Tibet Plateau and isolation of microorganisms contributing flavor. Anim Sci J 82:122–126
Winkler UK, Stuckmann M (1979) Glycogen, hyaluronate, and some other polysaccharides greatly enhance the formation of exolipase by Serratia marcescens. J Bacteriol 138:663–670
Salameh MA, Wiegel J (2007) Purification and characterization of two highly thermophilic alkaline lipases from Thermosyntropha lipolytica. Appl Environ Microbiol 73:7725–7731
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Cao Y, Zhuang Y, Yao C, Wu B, He B (2012) Purification and characterization of an organic solvent-stable lipase from Pseudomonas stutzeri LC2-8 and its application for efficient resolution of (R, S)-1-phenylethanol. Biochem Eng J 64:55–60
Mhetras NC, Bastawde KB, Gokhale DV (2009) Purification and characterization of acidic lipase from Aspergillus niger NCIM 1207. Bioresour Technol 100:1486–1490
Kojima Y, Shimizu S (2003) Purification and characterization of the lipase from Pseudomonas fluorescens HU380. J Biosci Bioeng 96:219–226
Wanasundara UN, Shahidi F (1998) Lipase-assisted concentration of n-3 polyunsaturated fatty acids in acylglycerols from marine oils. J Am Oil Chem Soc 75:945–951
Pan XX, Xu L, Zhang Y, Xiao X, Wang XF, Liu Y, Zhang HJ, Yan YJ (2012) Efficient display of active Geotrichum sp. lipase on Pichia pastoris cell wall and its application as a whole-cell biocatalyst to enrich EPA and DHA in fish oil. J Agric Food Chem 60:9673–9679
Namboodiri VM, Chattopadhyaya R (2000) Purification and biochemical characterization of a novel thermostable lipase from Aspergillus niger. Lipids 35:495–502
Yadav RP, Saxena RK, Gupta R, Davidson WS (1998) Purification and characterization of a regiospecific lipase from Aspergillus terreus. Biotechnol Appl Biochem 28:243–249
Lee D, Koh Y, Kim K, Kim B, Choi H, Kim D, Suhartono MT, Pyun Y (1999) Isolation and characterization of a thermophilic lipase from Bacillus thermoleovorans ID-1. FEMS Microbiol Lett 179:393–400
Talon R, Dublet N, Montel MC, Cantonnet M (1995) Purification and characterization of extracellular Staphylococcus warneri lipase. Curr Microbiol 30:11–16
Kasana RC, Kaur B, Yadav SK (2008) Isolation and identification of a psychrotrophic Acinetobacter sp. CR9 and characterization of its alkaline lipase. J Basic Microbiol 48:207–212
Castro-Ochoa LD, Rodríguez-Gómez C, Valerio-Alfaro G, Oliart Ros R (2005) Screening, purification and characterization of the thermoalkalophilic lipase produced by Bacillus thermoleovorans CCR11. Enzyme Microb Technol 37:648–654
Song X, Qi X, Hao B, Qu Y (2008) Studies of substrate specificities of lipases from different sources. Eur J Lipid Sci Technol 110:1095–1101
Ramani K, John Kennedy L, Ramakrishnan M, Sekaran G (2010) Purification, characterization and application of acidic lipase from Pseudomonas gessardii using beef tallow as a substrate for fats and oil hydrolysis. Process Biochem 45:1683–1691
Gaur R, Gupta A, Khare SK (2008) Purification and characterization of lipase from solvent tolerant Pseudomonas aeruginosa PseA. Process Biochem 43:1040–1046
Acknowledgments
We are grateful for financial support from the National Natural Science Fund of China (Grant Numbers 31130042, 31171630 and 21206046) and the Specialized Research Fund for the Doctoral Program of Higher Education of China (Number 20130172110018).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Liu, G., Hu, S., Li, L. et al. Purification and Characterization of a Lipase with High Thermostability and Polar Organic Solvent-Tolerance from Aspergillus niger AN0512. Lipids 50, 1155–1163 (2015). https://doi.org/10.1007/s11745-015-4052-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-015-4052-6