Abstract
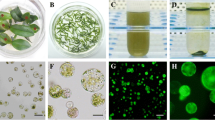
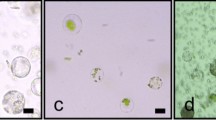
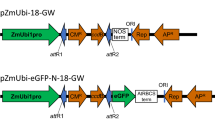
Populus euphratica Oliv., a species of the model woody plant genus Populus, is well known for its tolerance to salinity stress, the underlying mechanism of which is a research hotspot. Transient expression of fluorescent fusion proteins is commonly used for rapid assessment of gene functions and interactions, and thus would be useful to study the genes involved in salt tolerance in this species. Our transient gene expression protocol for P. euphratica included a simple protoplast preparation and transformation procedure from suspension cultured cells. The highest protoplast yield (8 × 107 g−1 fresh weight) with high viability (above 90 %) was obtained using an optimized enzyme mix of 4 % (w/v) cellulase R10, 0.5 % (w/v) pectinase, and 0.2 % (w/v) hemicellulase. Factors affecting protoplast transformation efficiency were also optimized: 20 μg plasmid DNA versus 105 protoplasts, and a transformation time of 20 min using PEG, which resulted in a transformation efficiency greater than 50 %. A pair of known markers was simultaneously and correctly expressed in the same P. euphratica protoplasts by co-transformation. The isolation and transformation protocol took 5 h, and results could be obtained within 24 h. This protoplast transient expression system is suitable for studying gene expression, protein localization, and protein–protein interactions in woody plants. In addition, it would be particularly useful for studying the signaling pathway involved in the salt tolerance of P. euphratica in a homologous system, which may not even be possible using protoplasts prepared from other species.






Similar content being viewed by others
References
Bai Y, Han N, Wu J, Yang Y, Wang J, Zhu M, Bian H (2014) A transient gene expression system using barley protoplasts to evaluate microRNAs for post-transcriptional regulation of their target genes. Plant Cell Tissue Organ Cult (PCTOC) 119(1):211–219
Bargmann BO, Birnbaum KD (2009) Positive fluorescent selection permits precise, rapid, and in-depth overexpression analysis in plant protoplasts. Plant Physiol 149(3):1231–1239. doi:10.1104/pp.108.133975
Bargmann BO, Birnbaum KD (2010) Fluorescence activated cell sorting of plant protoplasts. J Vis Exp JoVE. doi:10.3791/1673
Birnbaum K, Jung JW, Wang JY, Lambert GM, Hirst JA, Galbraith DW, Benfey PN (2005) Cell type-specific expression profiling in plants via cell sorting of protoplasts from fluorescent reporter lines. Nat Methods 2(8):615–619. doi:10.1038/nmeth0805-615
Brosche M, Vinocur B, Alatalo ER, Lamminmaki A, Teichmann T, Ottow EA, Djilianov D, Afif D, Bogeat-Triboulot MB, Altman A, Polle A, Dreyer E, Rudd S, Paulin L, Auvinen P, Kangasjarvi J (2005) Gene expression and metabolite profiling of Populus euphratica growing in the Negev desert. Genome Biol 6(12):R101. doi:10.1186/gb-2005-6-12-r101
Cao J, Yao D, Lin F, Jiang M (2014) PEG-mediated transient gene expression and silencing system in maize mesophyll protoplasts: a valuable tool for signal transduction study in maize. Acta Physiol Plant 36(5):1271–1281
Chen S, Polle A (2010) Salinity tolerance of Populus. Plant Biol 12(2):317–333
Chen S, Fritz E, Wang S, Hüttermann A, Liu Q, Jiang X (2000) Cellular distribution of ions in salt-stressed cells of Populus euphratica and P. tomentosa. For Stud China 2(2):8–16
Chen S, Ding S, Li J (2002) Isolation of protoplast and ion channel recording in plasma membrane of suspension cells of Populus euphratica. For Stud China 4(1):1–4
Chen S, Li J, Wang S, Fritz E, Hüttermann A, Altman A (2003) Effects of NaCl on shoot growth, transpiration, ion compartmentation, and transport in regenerated plants of Populus euphratica and Populus tomentosa. Can J For Res 33(6):967–975
Gries D, Zeng F, Foetzki A, Arndt SK, Bruelheide H, Thomas FM, Zhang X, Runge M (2003) Growth and water relations of Tamarix ramosissima and Populus euphratica on Taklamakan desert dunes in relation to depth to a permanent water table. Plant Cell Environ 26(5):725–736
Gu R, Fonseca S, Puskas LG, Hackler L Jr, Zvara A, Dudits D, Pais MS (2004) Transcript identification and profiling during salt stress and recovery of Populus euphratica. Tree Physiol 24(3):265–276
Guo Y, Bai J, Zhang Z (2007) Plant regeneration from embryogenic suspension-derived protoplasts of ginger (Zingiber officinale Rosc.). Plant Cell Tissue Organ Cult 89(2–3):151–157
Guo J, Morrell-Falvey JL, Labbe JL, Muchero W, Kalluri UC, Tuskan GA, Chen JG (2012) Highly efficient isolation of Populus mesophyll protoplasts and its application in transient expression assays. Plos One 7(9):e44908. doi:10.1371/journal.pone.0044908
Larkin PJ (1976) Purification and viability determinations of plant protoplasts. Planta 128(3):213–216. doi:10.1007/BF00393231
Li B, Qin Y, Duan H, Yin W, Xia X (2011) Genome-wide characterization of new and drought stress responsive microRNAs in Populus euphratica. J Exp Bot 62(11):3765–3779. doi:10.1093/jxb/err051
Lin YC, Li W, Sun YH, Kumari S, Wei H, Li Q, Tunlaya-Anukit S, Sederoff RR, Chiang VL (2013) SND1 transcription factor-directed quantitative functional hierarchical genetic regulatory network in wood formation in Populus trichocarpa. Plant Cell 25(11):4324–4341. doi:10.1105/tpc.113.117697
Lin YC, Li W, Chen H, Li Q, Sun YH, Shi R, Lin CY, Wang JP, Chen HC, Chuang L, Qu GZ, Sederoff RR, Chiang VL (2014) A simple improved-throughput xylem protoplast system for studying wood formation. Nat Protoc 9(9):2194–2205. doi:10.1038/nprot.2014.147
Lu Y, Chen X, Wu Y, Wang Y, He Y, Wu Y (2013) Directly transforming PCR-amplified DNA fragments into plant cells is a versatile system that facilitates the transient expression assay. Plos One 8(2):e57171
Ma H-C, Fung L, Wang S-S, Altman A, Hüttermann A (1997) Photosynthetic response of Populus euphratica to salt stress. For Ecol Manage 93(1):55–61
Ma T, Liu Q, Li Z, Zhang X (2002) Tonoplast H+-ATPase in response to salt stress in Populus euphratica cell suspensions. Plant Sci 163(3):499–505
Ma T, Wang J, Zhou G, Yue Z, Hu Q, Chen Y, Liu B, Qiu Q, Wang Z, Zhang J (2013) Genomic insights into salt adaptation in a desert poplar. Nat Commun 4:2797. doi:10.1038/ncomms3797
Marion J, Bach L, Bellec Y, Meyer C, Gissot L, Faure JD (2008) Systematic analysis of protein subcellular localization and interaction using high-throughput transient transformation of Arabidopsis seedlings. Plant J 56(1):169–179
Menges M, Murray JA (2002) Synchronous Arabidopsis suspension cultures for analysis of cell-cycle gene activity. Plant J 30(2):203–212
Nagata T, Nemoto Y, Hasezawa S (1992) Tobacco BY-2 cell line as the “HeLa” cell in the cell biology of higher plants. Int Rev Cytol 132(1):1–30
Ning J, Zhang B, Wang N, Zhou Y, Xiong L (2011) Increased leaf angle1, a Raf-like MAPKKK that interacts with a nuclear protein family, regulates mechanical tissue formation in the lamina joint of rice. Plant Cell Online 23(12):4334–4347
Nowak K, Luniak N, Meyer S, Schulze J, Mendel RR, Hansch R (2004) Fluorescent proteins in poplar: a useful tool to study promoter function and protein localization. Plant Biol 6(1):65–73. doi:10.1055/s-2004-815730
Ottow EA, Polle A, Brosche M, Kangasjarvi J, Dibrov P, Zorb C, Teichmann T (2005) Molecular characterization of PeNhaD1: the first member of the NhaD Na+/H+ antiporter family of plant origin. Plant Mol Biol 58(1):75–88. doi:10.1007/s11103-005-4525-8
Qiao J, Ishihara Y, Kuroda H, Sakai F, Sakai H, Komano T (1997) Transient expression of goat growth hormone gene in poplar (Populus alba L.) protoplasts: a quick method for detection of foreign gene expression in mRNA level. Biosci Biotechnol Biochem 61(9):1580–1581
Rezazadeh R, Williams RR, Harrison DK (2011) Factors affecting mango (Mangifera indica L.) protoplast isolation and culture. Sci Hortic 130(1):214–221
Sheen J (2001) Signal transduction in maize and Arabidopsis mesophyll protoplasts. Plant Physiol 127:1467
Stafford A, Warren G (1991) Plant cell and tissue culture. Open University Press, Philadelphia
Tan B, Xu M, Chen Y, Huang M (2013) Transient expression for functional gene analysis using Populus protoplasts. PCTOC 114(1):11–18
Tuskan GA, Difazio S, Jansson S (2006) The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 313(5793):1596–604. doi:10.1126/science.1128691
Wang S, Li H, Chen B (1996) Euphrates poplar forest. China Environmental Science Press, Beijing
Xiong G, Li R, Qian Q, Song X, Liu X, Yu Y, Zeng D, Wan J, Li J, Zhou Y (2010) The rice dynamin-related protein DRP2B mediates membrane trafficking, and thereby plays a critical role in secondary cell wall cellulose biosynthesis. Plant J 64(1):56–70
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2(7):1565–1572. doi:10.1038/nprot.2007.199
Zhang C, Gai Y, Zhu Y, Chen X, Jiang X (2008) Construction of a bidirectional promoter and its transient expression in Populus tomentosa. Front For China 3(1):112–116
Zhang Y, Su J, Duan S, Ao Y, Dai J, Liu J, Wang P, Li Y, Liu B, Feng D, Wang J, Wang H (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7(1):30. doi:10.1186/1746-4811-7-30
Zhang Y, Wang Y, Wang C (2012) Gene overexpression and gene silencing in Birch using an Agrobacterium-mediated transient expression system. Mol Biol Rep 39(5):5537–5541
Zhong R, Ye ZH (2010) The poplar PtrWNDs are transcriptional activators of secondary cell wall biosynthesis. Plant Signal Behav 5(4):469–472
Zhong R, McCarthy RL, Lee C, Ye ZH (2011) Dissection of the transcriptional program regulating secondary wall biosynthesis during wood formation in poplar. Plant Physiol 157(3):1452–1468. doi:10.1104/pp.111.181354
Zhuge Q, Huang M, Wang M (2000) A study on poplar somatic hybridization. J Nanjing For Univ 24(2):6–10
Acknowledgments
This work was supported by a grant from the Special Fund on Essential Research for National Non-profit Institutions to the Chinese Academy of Forestry (CAFYBB2011001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Moriguchi.
Rights and permissions
About this article
Cite this article
Guo, Y., Song, X., Zhao, S. et al. A transient gene expression system in Populus euphratica Oliv. protoplasts prepared from suspension cultured cells. Acta Physiol Plant 37, 160 (2015). https://doi.org/10.1007/s11738-015-1906-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-015-1906-8