Abstract
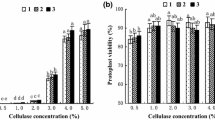
Despite the availability of the Populus genome sequence and the development of genetic, genomic, and transgenic approaches for its improvement, the lengthy life span of Populus and the cumbersome process required for its transformation have impeded rapid characterization of gene functions in Populus. Protoplasts provide a versatile and physiologically relevant cell system for high-throughput analysis and functional characterization of plant genes. Here, a highly efficient transient expression system using Populus mesophyll protoplasts was developed based on the following three steps. The first step involved formulating a new enzyme cocktail containing 2 % Cellulase C2605 and 0.5 % Pectinase P2611, which was shown to enable efficient large-scale isolation of homogenous Populus mesophyll protoplasts. The second step involved optimization of transfection conditions, such as the polyethylene glycol concentration and amount of plasmid DNA to ensure a >80 % transfection efficiency for Populus protoplasts. The third step involved using the Populus protoplast transient expression system to successfully determine the subcellular localizations of proteins, emulate signaling events during pathogen infection, and prepare protein extracts for Western blotting and protein–protein interaction assays. This rapid and highly efficient transient gene expression system in Populus mesophyll protoplasts will facilitate the rapid identification of gene functions and elucidation of signaling pathways in Populus.




Similar content being viewed by others
References
Asai T, Tena G, Plotnikova J, Willmann MR, Chiu WL, Gomez-Gomez L, Boller T, Ausubel FM, Sheen J (2002) MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415:977–983
Axtell MJ, Staskawicz BJ (2003) Initiation of RPS2-specified disease resistance in Arabidopsis is coupled to the AvrRpt2-directed elimination of RIN4. Cell 112:369–377
Boudsocq M, Willmann MR, McCormack M, Lee H, Shan L, He P, Bush J, Cheng SH, Sheen J (2010) Differential innate immune signalling via Ca2+ sensor protein kinases. Nature 464:418–422
Bradshaw HD, Ceulemans R, Davis J, Stettler R (2000) Emerging model systems in plant biology: poplar (Populus) as a model forest tree. J Plant Growth Regul 19:306–313
Busov VB, Strauss SH, Pilate G (2010) Transformation as a tool for genetic analysis in Populus genetics and genomics of Populus. In: Jansson S, Bhalerao RP, Groover AT (eds), genetics and genomics of Populus, 1st edn. Springer, New York, pp 113–133
Davey MR, Anthony P, Power JB, Lowe KC (2005) Plant protoplasts: status and biotechnological perspectives. Biotechnol Adv 23:131–171
Dekeyser RA, Claes B, De Rycke R, Habets ME, Van Montagu M, Caplan AB (1990) Transient gene expression in intact and organized rice tissues. Plant Cell 2:591–602
Du N, Liu X, Li Y, Chen S, Zhang J, Ha D, Deng W, Sun C, Zhang Y, Pijut P (2012) Genetic transformation of Populus tomentosa to improve salt tolerance. Plant Cell Tiss Org Cult 108(2):181–189
Faraco M, Di Sansebastiano GP, Spelt K, Koes RE, Quattrocchio FM (2011) One protoplast is not the other! Plant Physiol 156:474–478
Guo J, Morrell-Falvey JL, Labbé JL, Muchero W, Kalluri UC, Tuskan GA, Chen JG (2012) Highly efficient isolation of Populus mesophyll protoplasts and its application in transient expression assays. PLoS ONE 7:e44908
He S, Shan W, Kuang JF, Xie H, Xiao YY, Lu WJ, Chen JY (2012) Molecular characterization of a stress-response bZIP transcription factor in banana. Plant Cell Tiss Org Cult:1–15. doi:10.1007/s11240-012-0258-y
Jansson S, Douglas CJ (2007) Populus: a model system for plant biology. Annu Rev Plant Biol 58:435–458
Jiang L, Wang J, Liu Z, Wang L, Zhang F, Liu GC, Zhong Q (2010) Silencing induced by inverted repeat constructs in protoplasts of Nicotiana benthamiana. Plant Cell Tiss Org Cult 100:139–148
Kang J, Ide Y, Sasaki S (1995) Isolation and culture of leaf protoplasts from in vitro subcultured Poplars: Populus tomentosa, Populus alba cv. Pyramidalis × Populus tomentosa and Populus maximowiczii × Populus plantierensis. Bull Tokyo Uni For 93:59–63
Larkin PJ (1976) Purification and viability determinations of plant protoplasts. Planta 128:213–216
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497
Petřivalský M, Vaníčková P, Ryzí M, Navrátilová B, Piterková J, Sedlářová M, Luhová L (2012) The effects of reactive nitrogen and oxygen species on the regeneration and growth of cucumber cells from isolated protoplasts. Plant Cell Tiss Org Cult 108(2):237–249
Pitzschke A, Persak H (2012) Poinsettia protoplasts—a simple, robust and efficient system for transient gene expression studies. Plant Methods 8:14
Rouxel T, Balesdent MH (2010) Avirulence genes. In: Encyclopedia of Life Sciences (ELS). John Wiley & Sons. doi:10.1002/9780470015902.a00212672010
Russell JA, McCown BH (1986) Techniques for enhanced release of leaf protoplasts in Populus. Plant Cell Rep 5:284–287
Sasamoto H, Hosoi Y, Ishii K, Sato T, Saito A (1989) Factors affecting the formation of callus from leaf protoplasts of Populus alba. J Jpn For Soc 71:449–455
Sheen J (2001) Signal transduction in maize and Arabidopsis mesophyll protoplasts. Plant Physiol 127:1466–1475
Sodhi A, Biswas SK (2002) Monocyte chemoattractant protein-1-induced activation of p42/44 MAPK and c-Jun in murine peritoneal macrophages: a potential pathway for macrophage activation. J Interferon Cytokine Res 22:517–526
Taylor G (2002) Populus: Arabidopsis for forestry. Do we need a model tree? Ann Bot 90:681–689
Tuskan GA, DiFazio S, Jansson S, Bohlmann J, Grigoriev I, Hellsten U, Putnam N, Ralph S, Rombauts S, Salamov A, Schein J, Sterck L, Aerts A, Bhalerao RR, Bhalerao RP, Blaudez D, Boerjan W, Brun A, Brunner A, Busov V, Campbell M, Carlson J, Chalot M, Chapman J, Chen GL, Cooper D, Coutinho PM, Couturier J, Covert S, Cronk Q, Cunningham R, Davis J, Degroeve S, Déjardin A, Depamphilis C, Detter J, Dirks B, Dubchak I, Duplessis S, Ehlting J, Ellis B, Gendler K, Goodstein D, Gribskov M, Grimwood J, Groover A, Gunter L, Hamberger B, Heinze B, Helariutta Y, Henrissat B, Holligan D, Holt R, Huang W, Islam-Faridi N, Jones S, Jones-Rhoades M, Jorgensen R, Joshi C, Kangasjärvi J, Karlsson J, Kelleher C, Kirkpatrick R, Kirst M, Kohler A, Kalluri U, Larimer F, Leebens-Mack J, Leplé JC, Locascio P, Lou Y, Lucas S, Martin F, Montanini B, Napoli C, Nelson DR, Nelson C, Nieminen K, Nilsson O, Pereda V, Peter G, Philippe R, Pilate G, Poliakov A, Razumovskaya J, Richardson P, Rinaldi C, Ritland K, Rouzé P, Ryaboy D, Schmutz J, Schrader J, Segerman B, Shin H, Siddiqui A, Sterky F, Terry A, Tsai CJ, Uberbacher E, Unneberg P, Vahala J, Wall K, Wessler S, Yang G, Yin T, Douglas C, Marra M, Sandberg G, Van de Peer Y, Rokhsar D (2006) The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 313:1596–1604
Walter M, Chaban C, Schütze K, Batistic O, Weckermann K, Näke C, Blazevic D, Grefen C, Schumacher K, Oecking C, Harter K, Kudla J (2004) Visualization of protein interactions in living plant cells using bimolecular fluorescence complementation. Plant J 40:428–438
Weinthal D, Tzfira T (2009) Imaging protein–protein interactions in plant cells by bimolecular fluorescence complementation assay. Trends Plant Sci 14:59–63
Wu F, Shen S, Lee L, Lee SH, Chan MT, Lin CS (2009) Tape-Arabidopsis Sandwich—a simpler Arabidopsis protoplast isolation method. Plant Methods 5:16
Yoo S, Cho Y, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572
Zhang Y, Su J, Duan S, Ao Y, Dai J, Liu J, Wang P, Li Y, Liu B, Feng D, Wang J, Wang H (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7(1):30
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (31100484, 31200507), the High-level Talents Program of Nanjing Forestry University (GXL201202), and the National High Technology Research and Development Program of China (2013AA102703).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Biyue Tan and Meng Xu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tan, B., Xu, M., Chen, Y. et al. Transient expression for functional gene analysis using Populus protoplasts. Plant Cell Tiss Organ Cult 114, 11–18 (2013). https://doi.org/10.1007/s11240-013-0299-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-013-0299-x