Abstract
Purpose
Bariatric surgery is a treatment option for those affected by severe obesity. This study investigated changes in gut microbiota and serum biomarkers after laparoscopic sleeve gastrectomy (LSG).
Materials and Methods
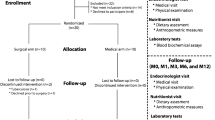
A total of 126 patients with morbid obesity who underwent LSG were enrolled in this study. Routine biochemical tests, hormonal (insulin and glucagon), and cytokine levels (IL-6, IL-1β, TNF-α, IL-10, and TGF–β 1) were measured, in addition, real-time PCR (quantitative PCR, qPCR) quantitated gut microbiota. All the parameters were measured pre-operatively, 3, and 12 months post-surgery (F0, F3, and F12, respectively).
Results
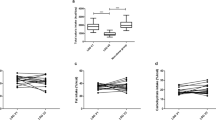
At F3, the level of FBS, HbA1c, HOMA-IR, triglyceride, cholesterol, LDL, BUN, creatinine, urea, SGOT, SGPT, IL-1β, IL-6, IFNγ, insulin, glucagon, the abundance of Prevotella and Bacteroides fragilis group, as well as the concentration of Firmicutes spp. showed significant decrease (P < 0.01), and HDL level, Akkermansia muciniphila and Roseburia spp. abundance, and Bacteroidetes and Bifidobacterium spp. concentration showed significant increase (P < 0.0001). The observed pattern continued or remained stable at F12 for all of these variables. IL-10 and TGF-β1 remained unchanged until F3 and showed a significant drop at F12. At F3, Clostridium cluster IV increased significantly and remained at that level afterward. Moreover, concentration of Phylum Actinobacteria showed an initial drop at F3 and a later increase at F12 (P < 0.0001).
Conclusion
LSG is associated with a significant improvement in serum biomarkers, as well as significant changes in fecal microbiota. Future systems biology analyses would shed more light on the underlying interactions of these parameters, and could help in developing novel diagnostic and therapeutic strategies for obesity management.
Similar content being viewed by others
Data Availability
All data generated or analyzed in this study are included in the present article.
References
Chooi YC, Ding C, Magkos F. The epidemiology of obesity. Metabolism. 2019;92:6–10.
Sivamaruthi BS, Kesika P, Suganthy N, et al. A review on role of microbiome in obesity and antiobesity properties of probiotic supplements. Biomed Res Int. 2019;2019:1–20.
Organization WH. Obesity and overweight. Fact sheet no. 311. Updated January 2015. World Health Organization [Cited: 2015 November 20] Available from: http://www.who.int/mediacentre/factsheets/fs311/en 2015.
Hruby A, Hu FB. The epidemiology of obesity: a big picture. Pharmacoeconomics. 2015;33(7):673–89.
Hady HR, Olszewska M, Czerniawski M, Groth D, Diemieszczyk I, Pawluszewicz P, Kretowski A, Ladny JR, Dadan J. Different surgical approaches in laparoscopic sleeve gastrectomy and their influence on metabolic syndrome: a retrospective study. Medicine. 2018;97(4).
Tabasi M, Ashrafian F, Khezerloo JK, et al. Changes in gut microbiota and hormones after bariatric surgery: a bench-to-bedside review. Obes Surg. 2019;29(5):1663–74.
Rosenbaum M, Knight R, Leibel RL. The gut microbiota in human energy homeostasis and obesity. Trends in Endocrinology & Metabolism. 2015;26(9):493–501.
Rajoka MSR, Shi J, Mehwish HM, et al. Interaction between diet composition and gut microbiota and its impact on gastrointestinal tract health. Food Sci Human Wellness. 2017;6(3):121–30.
Graessler J, Qin Y, Zhong H, et al. Metagenomic sequencing of the human gut microbiome before and after bariatric surgery in obese patients with type 2 diabetes: correlation with inflammatory and metabolic parameters. The pharmacogenomics journal. 2013;13(6):514–22.
Magouliotis DE, Tasiopoulou VS, Sioka E, et al. Impact of bariatric surgery on metabolic and gut microbiota profile: a systematic review and meta-analysis. Obes Surg. 2017;27(5):1345–57.
Hasan N, Yang H. Factors affecting the composition of the gut microbiota, and its modulation. PeerJ. 2019;7:e7502.
Owen-Smith A, Kipping R, Donovan J, et al. A NICE example? Variation in provision of bariatric surgery in England. Bmj. 2013;346:f2453.
Bergström A, Licht TR, Wilcks A, et al. Introducing GUt low-density array (GULDA)–a validated approach for qPCR-based intestinal microbial community analysis. FEMS Microbiol Lett. 2012;337(1):38–47.
Matsuki T, Watanabe K, Fujimoto J, et al. Use of 16S rRNA gene-targeted group-specific primers for real-time PCR analysis of predominant bacteria in human feces. Appl Environ Microbiol. 2004;70(12):7220–8.
Hermann-Bank ML, Skovgaard K, Stockmarr A, et al. The gut microbiota assay: a high-throughput qPCR approach combinable with next generation sequencing to study gut microbial diversity. BMC Genomics. 2013;14(1):788.
Koliada A, Syzenko G, Moseiko V, et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017;17(1):120.
Queipo-Ortuño MI, Seoane LM, Murri M, et al. Gut microbiota composition in male rat models under different nutritional status and physical activity and its association with serum leptin and ghrelin levels. PLoS One. 2013;8(5):e65465.
Schelbert KB. Comorbidities of obesity. Prim Care. 2009;36(2):271–85.
Webber J. The comorbidities of obesity. Practical Diabetes International. 2001;18(8):293–6.
Ruiz-Tovar J, Oller I, Priego P, et al. Short-and mid-term changes in bone mineral density after laparoscopic sleeve gastrectomy. Obes Surg. 2013;23(7):861–6.
Mihmanli M, Isil RG, Isil CT, et al. Effects of laparoscopic sleeve gastrectomy on parathyroid hormone, vitamin D, calcium, phosphorus, and albumin levels. Obes Surg. 2017;27(12):3149–55.
Anhê FF, Varin TV, Schertzer JD, et al. The gut microbiota as a mediator of metabolic benefits after bariatric surgery. Can J Diabetes. 2017;41(4):439–47.
Johnson EL, Heaver SL, Walters WA, et al. Microbiome and metabolic disease: revisiting the bacterial phylum Bacteroidetes. J Mol Med. 2017;95(1):1–8.
Reyed M. The role of bifidobacteria in health. Res J Med Med Sci. 2007;2(1):14–24.
Schneeberger M, Everard A, Gómez-Valadés AG, et al. Akkermansia muciniphila inversely correlates with the onset of inflammation, altered adipose tissue metabolism and metabolic disorders during obesity in mice. Sci Rep. 2015;5:16643.
Tamanai-Shacoori Z, Smida I, Bousarghin L, et al. Roseburia spp.: a marker of health? Future Microbiol. 2017;12(2):157–70.
Koliada A, Syzenko G, Moseiko V, et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017;17(1):1–6.
Miranda VP, dos Santos Amorim PR, Bastos RR, de Faria ER, de Castro Moreira ME, do Carmo Castro Franceschini S, do Carmo Gouveia Peluzio M, de Luces Fortes Ferreira CL, Priore SE. Abundance of gut microbiota, concentration of short-Chain fatty acids, and inflammatory markers associated with elevated body fat, overweight, and obesity in female adolescents. Mediators of Inflammation. 2019.
Acknowledgments
This article was part of the project conducted by Mohsen Tabasi to fulfill the requirement for a Ph.D. degree. We would like to express our gratitude to the Pasteur Institute of Iran and Legal Medicine Research Center for providing financial support. We also would like to express our appreciation to Dr. Nader Shahrokhi, Dr. Sara Ahmadi Badi, Mr. Milad Kheirvari, Mr. Mohammad Reza Yazdannasab, and staff in Shariati Hospital of Tehran, Iran for their technical assistance.
Funding
This project was financially supported by the Pasteur Institute of Iran (grant no. TP-9567).
Author information
Authors and Affiliations
Contributions
MT and SE design the study and data analysis. SDS and FE data: collecting and writing paper. ARS and SB: conceptualization, investigation, formal analysis, writing-review and editing. All co-authors commented on the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
This study was conducted in accordance with the principles of the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All the participants were thoroughly informed about the study and procedures before signing consent forms. Participants were assured of anonymity and confidentiality. The Research Ethics Committee of the Pasteur Institute of Iran, Tehran, approved this study (IR.PII. REC.1397.029).
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tabasi, M., Eybpoosh, S., Siadat, S.D. et al. Modulation of the Gut Microbiota and Serum Biomarkers After Laparoscopic Sleeve Gastrectomy: a 1-Year Follow-Up Study. OBES SURG 31, 1949–1956 (2021). https://doi.org/10.1007/s11695-020-05139-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-05139-2