Abstract
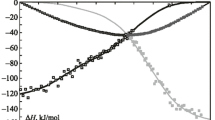
Mixing enthalpies of the liquid alloys in the binary Ca–Ge system were measured by isoperibolic solution calorimetry within the concentration ranges 0 < x Ge < 0.08 at 1270–1300 K, and 0.54 < x Ge < 1 at 1370–1520 K. The experimental data show great exothermic effects of alloy formation; the partial mixing enthalpies at infinite dilution of the components are \( \Delta \overline{H}_{\text{Ca}}^{\infty } \) = −138 to −140 kJ mol−1 and \( \Delta \overline{H}_{\text{Ge}}^{\infty } \) = −200 kJ mol−1 (at 1300 K). The Gibbs energies and entropies of mixing, activities of the components, and molar fractions of associates in the melts were calculated over a wide temperature range, using models of ideal and regular associated solutions, according to the data in literature for the thermodynamic properties of solid alloys and the phase diagram of the Ca–Ge system.





Similar content being viewed by others
References
M. Notin, J. Mejbar, A. Bouhajib, J. Charles, and J. Hertz, The thermodynamic properties of calcium intermetallic compounds, J. Alloys Compd., 1995, 220, p 62-75. doi:10.1016/0925-8388(94)06001-0
J. Mejbar, These de Doctorat, l’Universite de Nancy I (24 September 1993).
Z. Yang, D. Shi, B. Wen, R. Melnik, S. Yao, and T. Li, First-principle studies of Ca–X (X = Si, Ge, Sn, Pb) intermetallic compounds, J. Solid State Chem., 2010, 183, p 136-143. doi:10.1016/j.jssc.2009.11.007
Y. Djaballah, A. Pasturel, and A. Belgacem-Bouzida, Thermodynamic assessment of the calcium–germanium system, J. Alloys Compd., 2010, 497, p 74-79. doi:10.1016/j.jallcom.2010.02.189
B. Predel, Ca–Ge (Calcium–Germanium), Landolt-Börnstein-Group IV Physical Chemistry, Springer-Verlag, Berlin Heidelberg, 2012, 12B, p 160-161. doi:10.1007/978-3-540-44756-6_111
I.V. Nikolaenko, G.I. Batalin, E.A. Beloborodova, and M.A. Turchanin, Enthalpy of mixing of calcium with germanium, Metally, 1987, 2, p 39-40 (in Russian)
V.V. Litovskii, Enthalpies of formation of the binary alloys of earth-alkaline metals with silicon, germanium and tin, Synopsis of diss. Cand. Phys.-Math. Sci., Sverdlovsk (1986) (in Russian)
Yu.O. Yesin, V.V. Litovskii, and M.S. Petrushevskii, Partial and integral enthalpies of formation of the liquid alloys of calcium with silicon, germanium and tin, in V Union Conference on the thermodynamics of metallic alloys, Moscow (19–21 March 1985) 13 (in Russian).
K.A. Bolshakov, E.B. Sokolov, P.I. Fedorov, and A.V. Chirkin, Investigation of the melting diagram of the germanium–calcium system by the method of thermal analysis, Inorg. Mater., 1965, 1(10), p 1646 (in Russ. original, p 1822–1825).
T.B. Massalski, Binary Alloy Phase Diagrams, ASM International, Materials Park, 1990
A. Palenzona, P. Manfrinetti, and M.L. Fornasini, The phase diagram of the Ca–Ge system, J. Alloys Compd., 2002, 345, p 144-147. doi:10.1016/S0925-8388(02)00326-2
H. Okamoto, Ca-Ge (Calcium-Germanium), J. Phase Equilib. Diff., 2013, 34(2), p 172-173. doi:10.1007/s11669-012-0179-9
P.H. Tobash and S. Bobev, Synthesis, structure and electronic structure of a new polymorph of CaGe2, J. Solid State Chem., 2007, 180, p 1575-1581. doi:10.1016/j.jssc.2007.03.003
H. Bouderba, Y. Djaballah, A. Belgacem-Bouzida, and R. Beddiaf, Temperature and pressure effects on phase stabilities in the Ca–Ge system from first-principles calculations and Debye-Gruneisen model, Intermetallics, 2012, 28, p 108-119. doi:10.1016/j.intermet.2012.04.013
M. Ivanov, V. Berezutski, and N. Usenko, Mixing enthalpies in liquid alloys of manganese with the lanthanides, Int. J. Mater. Res., 2011, 102(3), p 277-281. doi:10.3139/146.110474
A.T. Dinsdale, SGTE data for pure elements, CALPHAD, 1991, 15, p 319-427. doi:10.1016/0364-5916(91)90030-N
I. Barin, Thermochemical data of pure substances, VCH, Weinheim; New York; Basel; Cambridge; Tokyo, 1995
D.R. Lide (ed), CRC Handbook of Chemistry and Physics, 84th Edition. CRC Press. Boca Raton, Florida (2003); Section 6, Fluid Properties; Vapor Pressure.
M.I. Ivanov, V.V. Berezutski, M.O. Shevchenko, V.G. Kudin, and V.S. Sudavtsova, Interaction in the alloys of the systems containing europium, Dopovidi (Reports) NAS of Ukraine, 2013, 8, p 90-99 (in Ukrainian)
V.G. Kudin, M.A. Shevchenko, I.V. Mateiko, and V.S. Sudavtsova, Thermodynamic properties of melts of the Al-La system, Russ. J. Phys. Chem., 2013, 87(3), p 364-370 (in Russian)
V.S. Sudavtsova, M.I. Ivanov, V.V. Berezutski, V.G. Kudin, and M.A. Shevchenko, Enthalpy of mixing of the Al–Yb melts, Russ. J. Phys. Chem., 2012, 86(8), p 1311-1315 (in Russian)
M.A. Shevchenko, V.G. Kudin, N.G. Kobylinskaya, and V.S. Sudavtsova, Thermodynamic properties and phase diagram of the Ce–Si system, Ukr. Chem. J., 2012, 78(6), p 96-102 (in Russian)
V.V. Berezutskii, M.A. Shevchenko, M.I. Ivanov, and V.S. Sudavtsova, Thermodynamic properties of liquid alloys Ni–Eu and Ni–Yb, Russ. J. Phys. Chem. A, 2014, 88(9), p 1463-1471. doi:10.1134/S0036024414090064
G. Kaptay, On the abilities and limitations of the linear, exponential and combined models to describe the temperature dependence of the excess Gibbs energy of solutions, CALPHAD, 2014, 44, p 81-94. doi:10.1016/j.calphad.2013.08.007
G. Kaptay, On the tendency of solutions to tend towards ideal solutions at high temperatures, Metall. Mater. Trans. A, 2012, 43, p 531-543. doi:10.1007/s11661-011-0902-x
M. Ohno, A. Kozlov, R. Arroyave, Z.K. Liu, and R. Schmid-Fetzer, Thermodynamic modeling of the Ca-Sn system based on finite temperature quantities from first-principles and experiment, Acta Mater., 2006, 54, p 4939-4951. doi:10.1016/j.actamat.2006.06.017
K. Li, S. Liu, C. Sha, and Y. Du, A thermodynamic reassessment of the Si-Sr system, CALPHAD, 2011, 35, p 594-600. doi:10.1016/j.calphad.2011.09.007
M. Idbenali, C. Servant, N. Selhaoui, and L. Bouirden, A thermodynamic reassessment of the Ca–Pb system, CALPHAD, 2008, 32, p 64-73. doi:10.1016/j.calphad.2007.10.004
C.B. Alcock, M.W. Chase, and V. Itkin, Thermodynamic properties of the Group IIA Elements, J. Phys. Chem. Ref. Data, 1993, 22(1), p 1-85. doi:10.1063/1.555935
A.A.A.P. da Silva, G.C. Coelho, C.A. Nunes, P.A. Suzuki, J.M. Fiorani, N. David, and M. Vilasi, Experimental determination of the Ta–Ge phase diagram, J. Alloys Compd., 2013, 576, p 38-42. doi:10.1016/j.jallcom.2013.04.070
F.R. de Boer, R. Boom, W.C.M. Mattens, A.R. Miedema, and A.K. Niessen, Cohesion in Metals Transition Metal Alloys, North Holland, Amsterdam, 1988
T. Geng, C. Li, Z. Du, C. Guo, X. Zhao, and H. Xu, Thermodynamic assessment of the Nb–Ge system, J. Alloys Compd., 2011, 509, p 3080-3088. doi:10.1016/j.jallcom.2010.12.005
E.A. Beloborodova, Thermodynamic properties of liquid alloys of germanium with d-transition metals, Powder Metall. Metal Ceram., 1996, 35(7–8), p 389-391
I.H. Jung, D.H. Kang, W.J. Park, N.J. Kim, and S.H. Ahn, Thermodynamic modeling of the Mg–Si–Sn system, CALPHAD, 2007, 31, p 192-200. doi:10.1016/j.calphad.2006.12.003
D. Nassyrov and I.H. Jung, Thermodynamic modeling of the Mg–Ge–Pb system, CALPHAD, 2009, 33, p 521-529. doi:10.1016/j.calphad.2009.01.005
M. Heyrman and P. Chartrand, Thermodynamic evaluation and optimization of the Ca–Si system, J. Phase Equilib. Diff., 2006, 27(3), p 220-230. doi:10.1361/154770306X109755
Y. Du, L. Li, J. Wang, J. Wang, and Z. Jin, A thermodynamic description of the Ge–Sr system acquired via a hybrid approach of CALPHAD and first-principles calculations, CALPHAD, 2009, 33, p 719-722. doi:10.1016/j.calphad.2009.09.004
J. Zhao, Y. Du, L. Zhang, A. Wang, L. Zhou, D. Zhao, and J. Liang, Thermodynamic assessment of the Sn–Sr system supported by first-principles calculations, Thermochim. Acta, 2012, 529, p 74-79. doi:10.1016/j.tca.2011.11.026
G. Bruzzone, E. Franceschi, and F. Merlo, On the Sr–Pb system, J. Less Common Met., 1981, 81, p 155-160
H. Okamoto, Ba–Si (Barium–Silicon), J. Phase Equilib. Diff., 2008, 29(5), p 464. doi:10.1007/s11669-008-9361-5
H. Okamoto, Ba–Ge (Barium–Germanium), J. Phase Equilib. Diff., 2009, 30(1), p 114. doi:10.1007/s11669-008-9428-3
M. Idbenali, C. Servant, N. Selhaoui, and L. Bouirden, A thermodynamic modelling of the Ba–Pb system, CALPHAD, 2007, 31, p 479-489. doi:10.1016/j.calphad.2007.04.003
V.V. Litovski, Enthalpies of formation of liquid binary alloys of alkaline-earth metals with silicon, germanium and tin, Thesis for Ph.D. in physics and mathematics, Sverdlovsk, 1986 (in Russian)
R.C. King and O.J. Kleppa, A thermochemical study of some selected Laves phases, Acta Metall., 1964, 12(1), p 87-97
J.R. Guadagno, M.J. Pool, and S.S. Shen, Thermodynamic investigation of liquid Ca–Sn, Sr–Sn, and Ba–Sn alloys, Metall. Trans., 1970, 1, p 1779-1780
V.S. Sudavtsova and G.I. Batalin, Thermodynamic properties of binary melts of the systems Ca–Si(Sn), Izv. AN USSR, Inorg. Mater., 1988, 24(9), p 1578-1580 (in Russian)
A. Yassin and R. Castanet, Enthalpies of dissolution of elements in liquid tin: II. Transition, alkali and alkaline-earth metals, J. Alloys Compd., 2001, 314, p 160-166. doi:10.1016/S0925-8388(00)01228-7
V.S. Sudavtsova, G.I. Batalin, and L.N. Zelenina, Thermodynamic properties of melts of the systems Si–(Mg, Sb), Ukr. Chim. Zhurn., 1988, 54(6), p 655-657 (in Russian)
F. Sommer, G. Borzone, N. Parodi, and R. Ferro, Enthalpy of formation of Ca–Pb and Ba–Pb alloys, Intermetallics, 2006, 14, p 287-296. doi:10.1016/j.intermet.2005.06.003
F. Sommer, J.J. Lee, and B. Predel, Temperature dependence of the mixing enthalpies of liquid magnesium-lead and magnesium-tin alloys, Z. Metallkd., 1980, 71(12), p 818-821
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shevchenko, M.O., Ivanov, M.I., Berezutski, V.V. et al. Thermodynamic Properties of Alloys in the Binary Ca–Ge System. J. Phase Equilib. Diffus. 36, 554–572 (2015). https://doi.org/10.1007/s11669-015-0408-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-015-0408-0