Abstract
Objective
To test whether Shenfu Injection (参附注射液, SFI) might attenuate the impact of cerebral energy dysfunction after resuscitation in a pig model of cardiac arrest (CA).
Methods
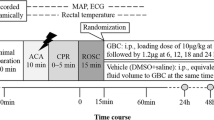
Thirty-four Wuzhishan miniature inbred pigs were randomly divided into three groups: the SFI group (n=12), the saline group (SA group, n=12), and the sham-operated group (sham group, n=10). Following successful return of spontaneous circulation (ROSC) from 8-min untreated ventricular fibrillation, animals received a continuous infusion of either SFI (0.2 mL/min) or saline for 6 h. Cerebral performance category score was evaluated at 24 and 48 h after ROSC, followed by positron emission tomography and computed tomography scans of cerebral glucose uptake. Surviving pigs were euthanized 48 h after ROSC, and the brains were removed for detecting mitochondrial function.
Results
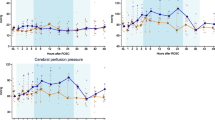
Compared with the SA group, SFI treatment produced a better neurologic outcome 48 h after ROSC (P<0.05). However, there was no significant difference of survival rate between the SA and SFI groups (83.3% vs. 81.8%, P>0.05). After ROSC, the SA group showed a decrease in the maximum standardized uptake value of different regions in the brain tissue, where SFI treatment can ameliorate these decreases (P<0.01 or P<0.05). Improved mitochondrial respiratory properties and higher mitochondrial membrane potential were also found following SFI treatment compared with the SA group at 48 h after ROSC (P<0.05 or P<0.01).
Conclusion
SFI treatment after resuscitation has significant neuroprotective effects against disruption of cerebral energy metabolism from CA by improving glucose uptake and by normalizing mitochondrial function.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Iwami T, Nichol G, Hiraide A, Hayashi Y, Nishiuchi T, Kajino K, et al. Continuous improvements in “chain of survival” increased survival after out-of-hospital cardiac arrests: a large-scale population-based study. Circulation 2009;119:728–734.
Busl KM, Greer DM. Hypoxic-ischemic brain injury: pathophysiology, neuropathology and mechanisms. NeuroRehabilitation 2010;26:5–13.
Laver S, Farrow C, Turner D, Nolan J. Mode of death after admission to an intensive care unit following cardiac arrest. Intensive Care Med 2004;30:2126–2128.
Jiang J, Fang X, Fu Y, Xu W, Jiang L, Huang Z. Impaired cerebral mitochondrial oxidative phosphorylation function in a rat model of ventricular fibrillation and cardiopulmonary resuscitation. Biomed Res Int 2014;2014:192769.
Sharma AB, Barlow MA, Yang SH, Simpkins JW, Mallet RT. Pyruvate enhances neurological recovery following cardiopulmonary arrest and resuscitation. Resuscitation 2008;76:108–119.
Gazmuri RJ, Radhakrishnan J. Protecting mitochondrial bioenergetic function during resuscitation from cardiac arrest. Crit Care Clin 2012;28:245–270.
Gong P, Hua R, Zhang Y, Zhao H, Tang Z, Mei X, et al. Hypothermia-induced neuroprotection is associated with reduced mitochondrial membrane permeability in a swine model of cardiac arrest. J Cereb Blood Flow Metab 2013;33:928–934.
Cao J, Zheng CD, Zhang GX, Zhang YJ, Min S. Protective effect of Shenfu Injection on myocardial mitochondria injured by ischemia-reperfusion in rabbits. Chin Med J 2005;118:505–507.
Lu JM, Yao Q, Chen C. Ginseng compounds: an update on their molecular mechanisms and medical applications. Curr Vasc Pharmacol 2009;7:293–302.
Leung KW, Wong AS. Pharmacology of ginsenosides: a literature review. Chin Med J 2010;5:20–24.
Nah SY, Kim DH, Rhim H. Ginsenosides: are any of them candidates for drugs acting on the central nervous system? CNS Drug Rev 2007;13:381–340.
Ye R, Li N, Han J, Kong X, Cao R, Rao Z, et al. Neuroprotective effects of ginsenoside Rd against oxygen-glucose deprivation in cultured hippocampal neurons. Neurosci Res 2009;64:306–310.
Lee YS, Kang YJ, Kim HJ, Park MK, Seo HG, Lee JH, et al. Higenamine reduces apoptotic cell death by induction of heme oxygenase-1 in rat myocardial ischemia-reperfusion injury. Apoptosis 2006;11:1091–1100.
Hou X, Li C, Gu W, Guo Z, Yin W, Zhang D. Effect of Shenfu on inflammatory cytokine release and brain edema after prolonged cardiac arrest in the swine. Am J Emerg Med 2013;31:1159–1164.
Yang LJ, Wang J, Tian ZF, Yuan YF. Shenfu Injection attenuates neonatal hypoxic-ischemic brain damage in rat. Neurol Sci 2013;34:1571–1574.
Li J, Gu L, Feng DF, Ding F, Zhu GG, Rong JD. Exploring temporospatial changes in glucose metabolic disorder, learning, and memory dysfunction in a rat model of diffuse axonal injury. J Neurotrauma 2012;29:2635–2646.
Lou M, Zhang H, Wang J, Wen SQ, Tang ZQ, Chen YZ, et al. Hyperbaric oxygen treatment attenuated the decrease in regional glucose metabolism of rats subjected to focal cerebral ischemia: a high resolution positron emission tomography study. Neuroscience 2007;11:555–561.
Ell PJ, von Schulthess GK. PET/CT: a new road map. Eur J Nucl Med Mol Imaging 2002;29:719–720.
De Volder AG, Michel C, Guérit JM, Bol A, Georges B, de Barsy T. Brain glucose metabolism in postanoxic syndrome due to cardiac arrest. Acta Neurol Belg 1994;94:183–189.
Neumar RW, Nolan JP, Adrie C, Aibiki M, Berg RA, Böttiger BW, et al. Post-cardiac arrest syndrome: epidemiology, pathophysiology, treatment, and prognostication: a consensus statement from the International Liaison Committee on Resuscitation (American Heart Association, Australian and New Zealand Council on Resuscitation, European Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Council of Asia, and the Resuscitation Council of Southern Africa); the American Heart Association Emergency Cardiovascular Care Committee; the Council on Cardiovascular Surgery and Anesthesia; the Council on Cardiopulmonary, Perioperative, and Critical Care; the Council on Clinical Cardiology; and the Stroke Council. Circulation 2008;118:2452–2483.
Wu JY, Li CS, Liu ZX, Wu CJ, Zhang GC. A comparison of 2 types of chest compressions in a porcine model of cardiac arrest. Am J Emerg Med 2009;27:823–829.
Killingsworth CR, Wei CC, Dell’Italia LJ, Ardell JL, Kingsley MA, Smith WM, et al. Short-acting beta-adrenergic antagonist esmolol given at reperfusion improves survival after prolonged ventricular fibrillation. Circulation 2004;109:2469–2474.
Pantazopoulos IN, Xanthos TT, Vlachos I, Troupis G, Kotsiomitis E, Johnson E, et al. Use of the impedance threshold device improves survival rate and neurological outcome in a swine model of asphyxial cardiac arrest. Crit Care Med 2012;40:861–868.
Ajam K, Gold LS, Beck SS, Damon S, Phelps R, Rea TD. Reliability of the cerebral performance category to classify neurological status among survivors of ventricular fibrillation arrest: a cohort study. Scand J Trauma Resusc Emerg Med 2011;19:38.
Herlitz J, Engdahl J, Svensson L, Angquist KA, Silfverstolpe J, Holmberg S. Major differences in 1-month survival between hospitals in Sweden among initial survivors of out-of-hospital cardiac arrest. Resuscitation 2006;70:404–409.
Noppens RR, Kelm RF, Lindemann R, Engelhard K, Werner C, Kempski O. Effects of a single-dose hypertonic saline hydroxyethyl starch on cerebral blood flow, long-term outcome, neurogenesis, and neuronal survival after cardiac arrest and cardiopulmonary resuscitation in rats. Crit Care Med 2012;40:2149–2156.
Ji XF, Yang L, Zhang MY, Li CS, Wang S, Cong LH. Shen-fu Injection attenuates postresuscitation myocardial dysfunction in a porcine model of cardiac arrest. Shock 2013;35:530–536.
Kawai N, Maeda Y, Kudomi N, Yamamoto Y, Nishiyama Y, Tamiya T. Focal neuronal damage in patients with neuropsychological impairment after diffuse traumatic brain injury: evaluation using 11C-flumazenil positron emission tomography with statistical image analysis. J Neurotrauma 2010;27:2131–2138.
Nakamura T, Kuroda Y, Torigoe N, Abe Y, Yamashita S, Kawakita K, et al. Cerebral metabolism monitoring during hypothermia following resuscitation from cardiopulmonary arrest. Acta Neurochir Suppl 2008;102:203–206.
Schaafsma A, de Jong BM, Bams JL, Haaxma-Reiche H, Pruim J, Zijlstra JG. Cerebral perfusion and metabolism in resuscitated patients with severe post-hypoxic encephalopathy. J Neurol Sci 2003;210:23–30.
Shang W, Yang Y, Zhou L, Jiang B, Jin H, Chen M. Ginsenoside Rb1 stimulates glucose uptake through insulin-like signaling pathway in 3T3-L1 adipocytes. J Endocrinol 2008;198:561–569.
Kann O, Kovács R. Mitochondria and neuronal activity. Am J Physiol Cell Physiol 2007;292:C641–C657.
Safar P, Behringer W, Böttiger BW, Sterz F. Cerebral resuscitation potentials for cardiac arrest. Crit Care Med 2002;30(Suppl):S140–S144.
Weber J, Senior AE. ATP synthesis driven by proton transport in F1F0-ATP synthase. FEBS Lett 2003;545:61–70.
Mitchell M, Schulz SL, Armstrong DT, Lane M. Metabolic and mitochondrial dysfunction in early mouse embryos following maternal dietary protein intervention. Biol Reprod 2009;80:622–630.
Acton BM, Jurisicova A, Jurisica I, Casper RF. Alterations in mitochondrial membrane potential during preimplantation stages of mouse and human embryo development. Mol Hum Reprod 2004;10:23–32.
Tian J, Zhang S, Li G, Liu Z, Xu B. 20(S)-ginsenoside Rg3, a neuroprotective agent, inhibits mitochondrial permeability transition pores in rat brain. Phytother Res 2009;23:486–491.
Ye R, Li N, Han J, Kong X, Cao R, Rao Z, et al. Neuroprotective effects of ginsenoside Rd against oxygen-glucose deprivation in cultured hippocampal neurons. Neurosci Res 2009;64:306–310.
Peberdy MA, Callaway CW, Neumar RW, Geocadin RG, Zimmerman JL, Donnino M, et al. Part 9: post-cardiac arrest care: 2010 American Heart Association Guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2010;122(Suppl):S768–S786.
Beylin ME, Perman SM, Abella BS, Leary M, Shofer FS, Grossestreuer AV, et al. Higher mean arterial pressure with or without vasoactive agents is associated with increased survival and better neurological outcomes in comatose survivors of cardiac arrest. Intensive Care Med 2013;39:1981–1988.
Isaev NK, Stelmashook EV, Plotnikov EY, Khryapenkova TG, Lozier ER, Doludin YV, et al. Role of acidosis, NMDA receptors, and acid-sensitive ion channel 1a (ASIC1a) in neuronal death induced by ischemia. Biochemistry (Mosc) 2008;73:1171–1175.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Beijing Natural Science Foundation (No. 7132092) and Beijing Scientific Research Project for Outstanding Doctoral Thesis Guidance Teacher (No. 20121002501)
Rights and permissions
About this article
Cite this article
Zhang, Y., Li, Cs., Wu, Cj. et al. Neuroprotective effect of Shenfu Injection (参附注射液) following cardiac arrest in pig correlates with improved mitochondrial function and cerebral glucose uptake. Chin. J. Integr. Med. 20, 835–843 (2014). https://doi.org/10.1007/s11655-014-1890-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-014-1890-7