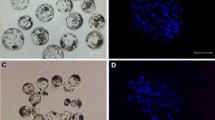
Abstract
Cytoplasmic microinjection (CI) of foreign gene into in vivo fertilized zygotes has emerged as a useful tool for transgenic pig production. In the current study, we investigated factors affecting transgenic efficiency and developmental potential of parthenogenetic (PA) and in vitro-fertilized (IVF) porcine embryos produced by CI. These factors included adding of RNase inhibitor, DNA or RNA concentration, injection time, and different structures of plasmids. Our results showed that adding of 1–4 U/μL of RNase inhibitor did not have negative effect on development potential of CI-PA embryos, and RNase inhibitor injection significantly increased EGFP expressing rate of CI-PA embryos. High injection DNA concentration and long injection interval after PA significantly reduced blastocyst formation. Different molecular structures such as DNA or RNA affected CI-PA embryos development, and RNA had little harmful effect on pig’s early embryonic development. EGFP expression rate of CI-IVF embryos was improved following the increase of foreign DNA concentration, but blastocyst formation rate was decreased. Injection time after IVF did not show any significant difference on embryonic development, but longer interval resulted in a significantly lower EGFP expressing rate. Cas9 mRNA and myostatin (GDF-8) sgRNA co-injection indicated that the mutation rate of CI-IVF group was significantly higher than that of CI-PA. The CI-IVF-generated embryos were then transferred to six recipient pigs, but no live piglets were obtained. The following pronuclear formation assessment showed more than 76.1% IVF zygotes were polyspermy. These results demonstrate that CI-PA and CI-IVF were effective methods for production of transgenic pig embryos. However, polyspermic fertilization and poor quality of porcine IVF blastocysts are still the main problem of resulting in pregnancy failure.




Similar content being viewed by others
References
Bedell VM, Wang Y, Campbell JM, Poshusta TL, Starker CG, Krug RG II, Tan W, Penheiter SG, Ma AC, Leung AY (2012) In vivo genome editing using a high-efficiency TALEN system. Nature 491:114–118
Bogdanove AJ, Voytas DF (2011) TAL effectors: customizable proteins for DNA targeting. Science 333:1843–1846
Brinster RL, Chen HY, Trumbauer ME, Avarbock MR (1980) Translation of globin messenger RNA by the mouse ovum. Nature 283:499–501
Brinster RL, Chen HY, Trumbauer ME, Yagle MK, Palmiter RD (1985) Factors affecting the efficiency of introducing foreign DNA into mice by microinjecting eggs. Proc Natl Acad Sci U S A 82:4438–4442
Cibelli J, Emborg ME, Prockop DJ, Roberts M, Schatten G, Rao M, Harding J, Mirochnitchenko O (2013) Strategies for improving animal models for regenerative medicine. Cell Stem Cell 12:271–274
Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339:819–823
Deltcheva E, Chylinski K, Sharma CM, Gonzales K, Chao Y, Pirzada ZA, Eckert MR, Vogel J, Charpentier E (2011) CRISPR RNA maturation by trans-encoded small RNA and host factor RNase III. Nature 471:602–607
Deng D, Yan C, Pan X, Mahfouz M, Wang J, Zhu J-K, Shi Y, Yan N (2012) Structural basis for sequence-specific recognition of DNA by TAL effectors. Science 335:720–723
Ebert KM, Paynton BV, McKnight GS, Brinster RL (1984) Translation and stability of ovalbumin messenger RNA injected into growing oocytes and fertilized ova of mice. J Embryol Exp Morphol 84:91–103
Fan N, Lai L (2013) Genetically modified pig models for human diseases. J Genet Genomics 40:67–73
Garcia-Vazquez FA, Ruiz S, Matas C, Izquierdo-Rico MJ, Grullon LA, De Ondiz A, Vieira L, Aviles-Lopez K, Gutierrez-Adan A, Gadea J (2010) Production of transgenic piglets using ICSI-sperm-mediated gene transfer in combination with recombinase RecA. Reproduction 140:259–272
Gil MA, Alminana C, Cuello C, Parrilla I, Roca J, Vazquez JM, Martinez EA (2007) Brief coincubation of gametes in porcine in vitro fertilization: role of sperm:oocyte ratio and post-coincubation medium. Theriogenology 67:620–626
Grupen CG (2014) The evolution of porcine embryo in vitro production. Theriogenology 81:24–37
Han Y-M, Abeydeera LR, Kim J-H, Moon H-B, Cabot RA, Day BN, Prather RS (1999a) Growth retardation of inner cell mass cells in polyspermic porcine embryos produced in vitro. Biol Reprod 60:1110–1113
Han Y-M, Wang W-H, Abeydeera LR, Petersen AL, Kim J-H, Murphy C, Day BN, Prather RS (1999b) Pronuclear location before the first cell division determines ploidy of polyspermic pig embryos. Biol Reprod 61:1340–1346
Hirabayashi M, Kato M, Ishikawa A, Kaneko R, Yagi T, Hochi S (2005) Factors affecting production of transgenic rats by ICSI‐mediated DNA transfer: effects of sonication and freeze‐thawing of spermatozoa, rat strains for sperm and oocyte donors, and different constructs of exogenous DNA. Mol Reprod Dev 70:422–428
Hsu PD, Lander ES, Zhang F (2014) Development and applications of CRISPR-Cas9 for genome engineering. Cell 157:1262–1278
Iqbal K, Barg-Kues B, Broll S, Bode J, Niemann H, Kues W (2009) Cytoplasmic injection of circular plasmids allows targeted expression in mammalian embryos. Biotechniques 47:959–968
Ivics Z, Garrels W, Mates L, Yau TY, Bashir S, Zidek V, Landa V, Geurts A, Pravenec M, Rulicke T, Kues WA, Izsvak Z (2014) Germline transgenesis in pigs by cytoplasmic microinjection of Sleeping Beauty transposons. Nat Protoc 9:810–827
Kikuchi K (2004) Developmental competence of porcine blastocysts produced in vitro. J Reprod Dev 50:21–28
Kues WA, Niemann H (2011) Advances in farm animal transgenesis. Prev Vet Med 102:146–156
Liu H, Chen Y, Niu Y, Zhang K, Kang Y, Ge W, Liu X, Zhao E, Wang C, Lin S (2014a) TALEN-mediated gene mutagenesis in rhesus and cynomolgus monkeys. Cell Stem Cell 14:323–328
Liu, S., Cui, K., Li, H. L., Sun, J. M., Lu, X. R., Shen, K. Y., Liu, Q. Y. and Shi, D. S. (2014b) Comparison of chemical, electrical, and combined activation methods for in vitro matured porcine oocytes. In Vitro Cellular & Developmental Biology-Animal, 1–10.
Page RL, Butler SP, Subramanian A, Gwazdauskas FC, Johnson JL, Velander WH (1995) Transgenesis in mice by cytoplasmic injection of polylysine/DNA mixtures. Transgenic Res 4:353–360
Paynton B, Ebert K, Brinster R (1983) Synthesis and secretion of ovalbumin by mouse-growing oocytes following microinjection of chick ovalbumin mRNA. Exp Cell Res 144:214–218
Perry AC, Wakayama T, Kishikawa H, Kasai T, Okabe M, Toyoda Y, Yanagimachi R (1999) Mammalian transgenesis by intracytoplasmic sperm injection. Science 284:1180–1183
Sumiyama K, Ruddle FH (2003) Regulation of Dlx3 gene expression in visceral arches by evolutionarily conserved enhancer elements. Proc Natl Acad Sci 100:4030–4034
Thouas G, Korfiatis N, French A, Jones G, Trounson A (2001) Simplified technique for differential staining of inner cell mass and trophectoderm cells of mouse and bovine blastocysts. Reprod Biomed Online 3:25–29
Urasaki, A. and Kawakami, K. (2009) Analysis of genes and genome by the tol2-mediated gene and enhancer trap methods. In Zebrafish Springer, pp. 85–102
Wang H, Hu Y-C, Markoulaki S, Welstead GG, Cheng AW, Shivalila CS, Pyntikova T, Dadon DB, Voytas DF, Bogdanove AJ (2013) TALEN-mediated editing of the mouse Y chromosome. Nat Biotechnol 31:530–532
Whyte JJ, Prather RS (2011) Genetic modifications of pigs for medicine and agriculture. Mol Reprod Dev 78:879–891
Wiedenheft B, Sternberg SH, Doudna JA (2012) RNA-guided genetic silencing systems in bacteria and archaea. Nature 482:331–338
Wyman C, Kanaar R (2006) DNA double-strand break repair: all’s well that ends well. Annu Rev Genet 40:363–383
Yong HY, Hao Y, Lai L, Li R, Murphy CN, Rieke A, Wax D, Samuel M, Prather RS (2006) Production of a transgenic piglet by a sperm injection technique in which no chemical or physical treatments were used for oocytes or sperm. Mol Reprod Dev 73:595–599
Yoshioka K, Suzuki C, Tanaka A, Anas IM, Iwamura S (2002) Birth of piglets derived from porcine zygotes cultured in a chemically defined medium. Biol Reprod 66:112–119
Zu Y, Tong X, Wang Z, Liu D, Pan R, Li Z, Hu Y, Luo Z, Huang P, Wu Q (2013) TALEN-mediated precise genome modification by homologous recombination in zebrafish. Nat Methods 10:329–331
Acknowledgments
This work was funded by the Hebei Provincial National Natural Science Fund (H2016314002) and the China 863 high-tec Project (2011AA100607).
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Tetsuji Okamoto
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 13 kb)
ESM 2
(DOCX 14 kb)
ESM 3
(DOCX 13 kb)
ESM 4
(DOCX 13 kb)
ESM 5
(DOCX 15 kb)
ESM 6
(DOCX 15 kb)
ESM 7
(DOCX 15 kb)
ESM 8
(DOCX 15 kb)
ESM 9
(DOCX 14 kb)
ESM 10
(DOCX 15 kb)
Fig. 1
Transgene vector constructions. pcDNA-EGFP-N1 (GIF 7 kb)
PRL-EGFP: EGFP Expression Vector with PRL ORF Controled by BCN promoter (GIF 11 kb)
TALEN-BMP15 and TALEN- FoxO3: The TALEN expression plasmid for knocking-out BMP15 and FoxO3 of porcine (GIF 9 kb)
hSpCas9 expression plasmid with a NLS at the N-terminal (GIF 9 kb)
Cas-BMP15, Cas-FoxO3 and Cas-GDF8 gRNA expression vector with a PsiI restriction endonuclease site for linearized (GIF 8 kb)
Fig. 2
Sequence alignment of GDF8 IVF mutant embryos (GIF 187 kb)
(GIF 244 kb)
(GIF 231 kb)
Rights and permissions
About this article
Cite this article
Liu, S., Liu, X., Huang, H. et al. Factors affecting efficiency of introducing foreign DNA and RNA into parthenogenetic or in vitro-fertilized porcine eggs by cytoplasmic microinjection. In Vitro Cell.Dev.Biol.-Animal 52, 713–722 (2016). https://doi.org/10.1007/s11626-016-0025-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-016-0025-1