Abstract
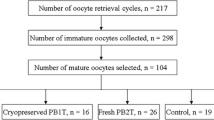
Despite many studies in humans and mice using genome transfer (GT), there are few reports using this technique in oocytes of wild or domestic animals. Therefore, we aimed to establish a GT technique in bovine oocytes using the metaphase plate (MP) and polar body (PB) as the sources of genetic material. In the first experiment, GT was established using MP (GT-MP), and a sperm concentration of 1 × 106 or 0.5 × 106 spermatozoa/ml gave similar fertilization rates. The cleavage rate (50%) and blastocyst rate (13.6%) in the GT-MP group was lower than that of the in vitro production control group (80.2% and 32.6%, respectively). The second experiment evaluated the same parameters using PB instead of MP; the GT-PB group had lower fertilization (82.3% vs. 96.2%) and blastocyst (7.7% vs. 36.8%) rates than the control group. No differences in the amount of mitochondrial DNA (mtDNA) were observed between groups. Finally, GT-MP was performed using vitrified oocytes (GT-MPV) as a source of genetic material. The cleavage rate of the GT-MPV group (68.4%) was similar to that of the vitrified oocytes (VIT) control group (70.0%) and to that of the control IVP group (81.25%, P < 0.05). The blastocyst rate of GT-MPV (15.7) did not differ neither from the VIT control group (5.0%) nor from the IVP control group (35.7%). The results suggested that the structures reconstructed by the GT-MPV and GT-PB technique develop in embryos even if vitrified oocytes are used.

Similar content being viewed by others
References
Arcarons N, Morato R, Spricigo JF, Ferraz MA, Mogas T. Spindle configuration and developmental competence of in vitro-matured bovine oocytes exposed to NaCl or sucrose prior to Cryotop vitrification. Reprod Fertil Dev. 2015;28:1560–9. https://doi.org/10.1071/RD14516.
Mrowiec P, Nowak A, Kochan J, Mlodawska W. Using Rapid I Method for vitrification of bovine oocytes. Cryo-Lett. 2019;40(2):123–8.
Arav A, Natan Y, Kalo D, Komsky-Elbaz A, Roth Z, Levi-Setti PE, et al. A new, simple, automatic vitrification device: preliminary results with murine and bovine oocytes and embryos. J Assist Reprod Genet. 2018;35(7):1161–8. https://doi.org/10.1007/s10815-018-1210-9.
Cobo A, Garrido N, Pellicer A, Remohi J. Six years’ experience in ovum donation using vitrified oocytes: report of cumulative outcomes, impact of storage time, and development of a predictive model for oocyte survival rate. Fertil Steril. 2015;104(6):1426–34 e1-8. https://doi.org/10.1016/j.fertnstert.2015.08.020.
Cao YX, Xing Q, Li L, Cong L, Zhang ZG, Wei ZL, et al. Comparison of survival and embryonic development in human oocytes cryopreserved by slow-freezing and vitrification. Fertil Steril. 2009;92(4):1306–11. https://doi.org/10.1016/j.fertnstert.2008.08.069.
Fisch B, Abir R. Female fertility preservation: past, present and future. Reprod. 2018;156(1):11–27. https://doi.org/10.1530/REP-17-0483.
Kuwayama M, Vajta G, Kato O, Leibo SP. Highly efficient vitrification method for cryopreservation of human oocytes. Reprod Biomed Online. 2005;11(3):300–8. https://doi.org/10.1016/S1472-6483(10)60837-1.
Spricigo JF, Morais K, Ferreira AR, Machado GM, Gomes AC, Rumpf R, et al. Vitrification of bovine oocytes at different meiotic stages using the Cryotop method: assessment of morphological, molecular and functional patterns. Cryobiology. 2014;69(2):256–65. https://doi.org/10.1016/j.cryobiol.2014.07.015.
Hwang I-S, Kwon D-J, Im G-S, Tashima K, Hochi S, Hwang S. High incidence of polyspermic fertilization in bovine oocytes matured in vitro after Cryotop vitrification. Cryo-Lett. 2016;37(1):27–33.
Punyawai K, Anakkul N, Srirattana K, Aikawa Y, Sangsritavong S, Nagai T, et al. Comparison of Cryotop and micro volume air cooling methods for cryopreservation of bovine matured oocytes and blastocysts. J Reprod Dev. 2015;61(5):431–7. https://doi.org/10.1262/jrd.2014-163.
Amstislavsky S, Mokrousova V, Brusentsev E, Okotrub K, Comizzoli P. Influence of cellular lipids on cryopreservation of mammalian oocytes and preimplantation embryos: a review. Biopreserv Biobank. 2019;17(1):76–83. https://doi.org/10.1089/bio.2018.0039.
Ezoe K, Yabuuchi A, Tani T, Mori C, Miki T, Takayama Y, et al. Developmental competence of vitrified-warmed bovine oocytes at the germinal-vesicle stage is improved by cyclic adenosine monophosphate modulators during in vitro maturation. PLoS One. 2015;10(5):e0126801. https://doi.org/10.1371/journal.pone.0126801.
Monteiro CAS, Leal GR, Saraiva HFRA, Garcia JM, Camargo AJR, Serapião RV, et al. Short term culture with cAMP modulators before vitrification significantly improve actin integrity in bovine oocytes. Livest. Sci. 2017;197:96–101. https://doi.org/10.1016/j.livsci.2017.01.013.
Barrera N, Dos Santos Neto PC, Cuadro F, Bosolasco D, Mulet AP, Crispo M, et al. Impact of delipidated estrous sheep serum supplementation on in vitro maturation, cryotolerance and endoplasmic reticulum stress gene expression of sheep oocytes. PLoS One. 2018;13(6):e0198742. https://doi.org/10.1371/journal.pone.0198742.
Spricigo JF, Morais KS, Yang BS, Dode MA. Effect of the exposure to methyl-beta-cyclodextrin prior to chilling or vitrification on the viability of bovine immature oocytes. Cryobiology. 2012;65(3):319–25. https://doi.org/10.1016/j.cryobiol.2012.09.001.
Spricigo JF, Diogenes MN, Leme LO, Guimaraes AL, Muterlle CV, Silva BD, et al. Effects of different maturation systems on bovine oocyte quality, plasma membrane phospholipid composition and resistance to vitrification and warming. PLoS One. 2015;10(6):e0130164. https://doi.org/10.1371/journal.pone.0130164.
Sprícigo JF, Morató R, Arcarons N, Yeste M, Dode MA, López-Bejar M, et al. Assessment of the effect of adding L-carnitine and/or resveratrol to maturation medium before vitrification on in vitro-matured calf oocytes. Theriogenology. 2017;89:47–57. https://doi.org/10.1016/j.theriogenology.2016.09.035.
Tachibana M, Sparman M, Mitalipov S. Chromosome transfer in mature oocytes. Nat Protoc. 2010;5(6):1138–47. https://doi.org/10.1038/nprot.2010.75.
Graves-Herring JE, Wildt DE, Comizzoli P. Retention of structure and function of the cat germinal vesicle after air-drying and storage at suprazero temperature. Biol Reprod. 2013;88(6):139. https://doi.org/10.1095/biolreprod.113.108472.
Paull D, Emmanuele V, Weiss KA, Treff N, Stewart L, Hua H, et al. Nuclear genome transfer in human oocytes eliminates mitochondrial DNA variants. Nat. 2013;493(7434):632–7. https://doi.org/10.1038/nature11800.
Tachibana M, Amato P, Sparman M, Woodward J, Sanchis DM, Ma H, et al. Towards germline gene therapy of inherited mitochondrial diseases. Nat. 2013;493(7434):627–31. https://doi.org/10.1038/nature11647.
Neupane J, Vandewoestyne M, Ghimire S, Lu Y, Qian C, Van Coster R, et al. Assessment of nuclear transfer techniques to prevent the transmission of heritable mitochondrial disorders without compromising embryonic development competence in mice. Mitochondrion. 2014;18:27–33. https://doi.org/10.1016/j.mito.2014.09.003.
Otsuki J, Nagai Y, Sankai T. Aggregated chromosomes transfer in human oocytes. Reprod Biomed Online. 2014;28(3):401–4. https://doi.org/10.1016/j.rbmo.2013.10.024.
Wang T, Sha H, Ji D, Zhang HL, Chen D, Cao Y, et al. Polar body genome transfer for preventing the transmission of inherited mitochondrial diseases. Cell. 2014;157(7):1591–604. https://doi.org/10.1016/j.cell.2014.04.042.
Lee YJ, Lin W, Peng SY, Lee JW, Lin YH, Yu C, et al. Effects of intracytoplasmic sperm injection timing and fertilization methods on the development of bovine spindle transferred embryos. Theriogenology. 2022;180:63–71. https://doi.org/10.1016/j.theriogenology.2021.12.012.
Wang Z, Li Y, Yang X, Wang Y, Nie Y, Xu Y, et al. Mitochondrial replacement in macaque monkey offspring by first polar body transfer. Cell Res. 2021;31(2):233–6. https://doi.org/10.1038/s41422-020-0381-y.
Tachibana M, Sparman M, Sritanaudomchai H, Ma H, Clepper L, Woodward J, et al. Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nat. 2009;461:367. https://doi.org/10.1038/nature08368.
Wang Z-W, Zhang G-L, Schatten H, Carroll J, Sun Q-Y. Cytoplasmic determination of meiotic spindle size revealed by a unique inter-species germinal vesicle transfer model. Sci Rep. 2016;6:19827. https://doi.org/10.1038/srep19827.
Zhang J, Liu H. Cytoplasm replacement following germinal vesicle transfer restores meiotic maturation and spindle assembly in meiotically arrested oocytes. Reprod Biomed Online. 2015;31(1):71–8. https://doi.org/10.1016/j.rbmo.2015.03.012.
Zhang S-P, Lu C-F, Gong F, Xie P-Y, Hu L, Zhang S-J, et al. Polar body transfer restores the developmental potential of oocytes to blastocyst stage in a case of repeated embryo fragmentation. J Assist Reprod Genet. 2017;34(5):563–71. https://doi.org/10.1007/s10815-017-0881-y.
Yamada M, Sato S, Ooka R, Akashi K, Nakamura A, Miyado K, et al. Mitochondrial replacement by genome transfer in human oocytes: efficacy, concerns, and legality. Reprod Med Biol. 2021;20(1):53–61. https://doi.org/10.1002/rmb2.12356.
Franciosi F, Perazzoli F, Lodde V, Modina SC, Luciano AM. Developmental competence of gametes reconstructed by germinal vesicle transplantation from fresh and cryopreserved bovine oocytes. Fertil Steril. 2010;93(1):229–38. https://doi.org/10.1016/j.fertnstert.2008.09.078.
Luciano A, Franciosi F, Lodde V, Perazzoli F, Slezáková M, Modina S. Cryopreservation of immature bovine oocytes to reconstruct artificial gametes by germinal vesicle transplantation. Reprod Domest Anim. 2009;44(3):480–8. https://doi.org/10.1111/j.1439-0531.2008.01194.x.
Holm P, Booth PJ, Schmidt MH, Greve T, Callesen H. High bovine blastocyst development in a static in vitro production system using sofaa medium supplemented with sodium citrate and myo-inositol with or without serum-proteins. Theriogenology. 1999;52(4):683–700. https://doi.org/10.1016/S0093-691X(99)00162-4.
Parrish JJ, Krogenaes A, Susko-Parrish JL. Effect of bovine sperm separation by either swim-up or Percoll method on success of in vitro fertilization and early embryonic development. Theriogenology. 1995;44(6):859–69. https://doi.org/10.1016/0093-691X(95)00271-9.
Machado GM, Carvalho JO, Filho ES, Caixeta ES, Franco MM, Rumpf R, et al. Effect of Percoll volume, duration and force of centrifugation, on in vitro production and sex ratio of bovine embryos. Theriogenology. 2009;71(8):1289–97. https://doi.org/10.1016/j.theriogenology.2009.01.002.
Iwata H, Goto H, Tanaka H, Sakaguchi Y, Kimura K, Kuwayama T, et al. Effect of maternal age on mitochondrial DNA copy number, ATP content and IVF outcome of bovine oocytes. Reprod Fertil Dev. 2011;23(3):424–32. https://doi.org/10.1071/RD10133.
Sripunya N, Liang Y, Panyawai K, Srirattana K, Ngernsoungnern A, Ngernsoungnern P, et al. Cytochalasin B efficiency in the cryopreservation of immature bovine oocytes by Cryotop and solid surface vitrification methods. Cryobiology. 2014;69(3):496–9. https://doi.org/10.1016/j.cryobiol.2014.09.001.
Sprícigo JFW, Netto SBS, Muterlle CV, Rodrigues SAD, Leme LO, Guimarães AL, et al. Intrafollicular transfer of fresh and vitrified immature bovine oocytes. Theriogenology. 2016;86(8):2054–62. https://doi.org/10.1016/j.theriogenology.2016.07.003.
García-Martínez T, Vendrell-Flotats M, Martínez-Rodero I, Ordóñez-León EA, Álvarez-Rodríguez M, López-Béjar M, et al. Glutathione ethyl ester protects in vitro-maturing bovine oocytes against oxidative stress induced by subsequent vitrification/warming. Int J Mol Sci. 2020;21(20):7547. https://doi.org/10.3390/ijms21207547.
Sun W-S, Jang H, Kwon HJ, Kim KY, Ahn SB, Hwang S, et al. The protective effect of Leucosporidium-derived ice-binding protein (LeIBP) on bovine oocytes and embryos during vitrification. Theriogenology. 2020;151:137–43. https://doi.org/10.1016/j.theriogenology.2020.04.016.
Ding C, Li T, Zeng Y, Hong P, Xu Y, Zhou C. Effects of in-vitro or in-vivo matured ooplasm and spindle-chromosome complex on the development of spindle-transferred oocytes. Reprod Biomed Online. 2014;29(6):708–16. https://doi.org/10.1016/j.rbmo.2014.08.012.
Magata F, Tsuchiya K, Okubo H, Ideta A. Application of intracytoplasmic sperm injection to the embryo production in aged cows. J Vet Med Sci. 2018:18-0284. https://doi.org/10.1292/jvms.18-0284.
Zhang J, Liu H, Luo S, Lu Z, Chávez-Badiola A, Liu Z, et al. Live birth derived from oocyte spindle transfer to prevent mitochondrial disease. Reprod Biomed Online. 2017;34(4):361–8. https://doi.org/10.1016/j.rbmo.2017.01.013.
Bianchi E, Wright GJ. Sperm meets egg: the genetics of mammalian fertilization. Annu Rev Genet. 2016;50(1):93–111. https://doi.org/10.1146/annurev-genet-121415-121834.
Acknowledgements
We thank the Brazilian Agricultural Research Corporation (EMBRAPA) and the Coordination for the Improvement of Higher Education Personnel (CAPES) for financial support, the Qualimax (Luziania GO) and Nippobras (Formosa, GO) slaughterhouses for providing biological material to perform these experiments, and all of the students and workers of EMBRAPA for their support during the course of these experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dode, M.A.N., Caixeta, F.M.C., Vargas, L.N. et al. Genome transfer technique for bovine embryo production using the metaphase plate and polar body. J Assist Reprod Genet 40, 943–951 (2023). https://doi.org/10.1007/s10815-023-02758-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-023-02758-3