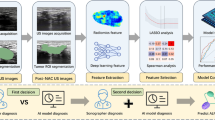
Abstract
Background
This study aimed to develop and validate radiomics and deep learning (DL) signatures for predicting distal metastasis (DM) of non-small cell lung cancer (NSCLC) in low-dose computed tomography (LDCT).
Methods
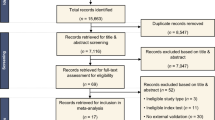
Images and clinical data were retrospectively collected for 381 NSCLC patients and prospectively collected for 114 patients at the Fifth Affiliated Hospital of Sun Yat-Sen University. Additionally, we enrolled 179 patients from the Jiangmen Central Hospital to externally validate the signatures. Machine-learning algorithms were employed to develop radiomics signature while the DL signature was developed using neural architecture search. The diagnostic efficiency was primarily quantified with the area under receiver operating characteristic curve (AUC). We interpreted the reasoning process of the radiomics signature and DL signature by radiomics voxel mapping and attention weight tracking.
Results
A total of 674 patients with pathologically-confirmed NSCLC were included from two institutions, with 143 of them having DM. The radiomics signature achieved AUCs of 0.885, 0.854, and 0.733 in the internal validation, prospective validation, and external validation while those for DL signature were 0.893, 0.786, and 0.780. The proposed signatures achieved a promising performance in predicting the DM of NSCLC and outperformed the approaches proposed in previous studies. Interpretability analysis revealed that both radiomics and DL signatures could detect the variations among voxels inside tumors, which helped in identifying the DM of NSCLC.
Conclusions
Our study demonstrates the potential of LDCT-based radiomics and DL signatures for predicting DM in NSCLC. These signatures could help improve lung cancer screening regarding further diagnostic tests and treatment strategies.





Similar content being viewed by others
Availability of data and materials
The datasets analysed during the current study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Duma N, Santana-Davila R, Molina JR (2019) Non-small cell lung cancer: epidemiology, screening, diagnosis, and treatment. Mayo Clin Proc 94:1623–1640. https://doi.org/10.1016/j.mayocp.2019.01.013
National Cancer Institute (2023) The Surveillance E, and End Results (SEER) Program. Cancer Stat Facts: Lung and Bronchus Cancer. https://seer.cancer.gov/statfacts/html/lungb.html. Published December 11, 2011. Updated March 30, 2022. Accessed June 30
Ashok A, Jiwnani SS, Karimundackal G et al (2021) Controversies in mediastinal staging for nonsmall cell lung cancer. Indian J Med Paediatr Oncol 42:406–414. https://doi.org/10.1055/s-0041-1739345
Lam S, Bai C, Baldwin D et al (2023) Current and future perspectives on CT screening for lung cancer: a road map for 2023–2027 from the IASLC. J Thorac Oncol. https://doi.org/10.1016/j.jtho.2023.07.019
Lv XY, Wu ZG, Cao JL et al (2021) A nomogram for predicting the risk of lymph node metastasis in T1–2 non-small-cell lung cancer based on PET/CT and clinical characteristics. Transl Lung Cancer Res 10:430–438. https://doi.org/10.21037/tlcr-20-1026
Manafi-Farid R, Askari E et al (2022) [18F]FDG-PET/CT radiomics and artificial intelligence in lung cancer: technical aspects and potential clinical applications. Semin Nucl Med 52:759–780. https://doi.org/10.1053/j.semnuclmed.2022.04.004
Kandathil A, Kay FU, Butt YM, Wachsmann JW, Subramaniam RM (2018) Role of FDG PET/CT in the eighth edition of TNM staging of non-small cell lung cancer. Radiographics 38:2134–2149. https://doi.org/10.1148/rg.2018180060
Esteva A, Kuprel B, Novoa RA et al (2017) Dermatologist-level classification of skin cancer with deep neural networks. Nature 542:115–118. https://doi.org/10.1038/nature21056
Li X, Zhang S, Zhang Q et al (2019) Diagnosis of thyroid cancer using deep con-volutional neural network models applied to sonographic images: a retro-spective, multicohort, diagnostic study. Lancet Oncol 20:193–201. https://doi.org/10.1016/S1470-2045(18)30762-9
Rajpurkar P, Irvin J, Ball RL et al (2018) Deep learning for chest radiograph diagnosis: a retrospective comparison of the CheXNeXt algorithm to practicing radiologists. PLoS Med 15:e1002686. https://doi.org/10.1371/journal.pmed.1002686
Bi WL, Hosny A, Schabath MB et al (2019) Artificial intelligence in cancer imaging: clinical challenges and applications. CA Cancer J Clin 69:127–157. https://doi.org/10.3322/caac.21552
Ma X, Xia L, Chen J, Wan W, Zhou W (2023) Development and validation of a deep learning signature for predicting lymph node metastasis in lung adenocarcinoma: comparison with radiomics signature and clinical-semantic model. Eur Radiol 33:1949–1962. https://doi.org/10.1007/s00330-022-09153-z
Cong M, Feng H et al (2020) Development of a predictive radiomics model for lymph node metastases in pre-surgical CT-based stage IA non-small cell lung cancer. Lung Cancer 139:73–79. https://doi.org/10.1016/j.lungcan.2019.11.003
Sun Q, Lin X, Zhao Y et al (2020) Deep learning vs. radiomics for predicting axillary lymph node metastasis of breast cancer using ultrasound images: don’t forget the peritumoral region. Front Oncol 10:53. https://doi.org/10.3389/fonc.2020.00053
Dong D, Fang MJ, Tang L et al (2020) Deep learning radiomic nomogram can predict the number of lymph node metastasis in locally advanced gastric cancer: an international multicenter study. Ann Oncol 31:912–920. https://doi.org/10.1016/j.annonc.2020.04.003
Lee JH, Ha EJ, Kim D et al (2020) Application of deep learning to the diagnosis of cervical lymph node metastasis from thyroid cancer with CT: external validation and clinical utility for resident training. Eur Radiol 30:3066–3072. https://doi.org/10.1007/s00330-019-06652-4
Tau N, Stundzia A, Yasufuku K, Hussey D, Metser U (2020) Convolutional neural networks in predicting nodal and distant metastatic potential of newly diagnosed non-small cell lung cancer on FDG PET images. AJR Am J Roentgenol 215:192–197. https://doi.org/10.2214/AJR.19.22346
Liu Y, Sun Y et al (2021) A survey on evolutionary neural architecture search. IEEE Trans Neural Netw Learn Syst. https://doi.org/10.1109/TNNLS.2021.3100554
van Griethuysen JJM et al (2017) Computational radiomics system to decode the radiographic phenotype. Can Res 77:e104–e107. https://doi.org/10.1158/0008-5472.CAN-17-0339
Woo S, Park J, Lee JY, Kweon IS, (2018) CBAM: Convolutional block attention module. In: Proceedings of the European Conference on Computer Vision (ECCV), pp 3–19. https://doi.org/10.1007/978-3-030-01234-2_1
Lee SM, Park CM, Paeng JC et al (2012) Accuracy and predictive features of FDG-PET/CT and CT for diagnosis of lymph node metastasis of T1 non-small-cell lung cancer manifesting as a subsolid nodule. Eur Radiol 22:1556–1563. https://doi.org/10.1007/s00330-012-2395-4
Li CJ, Tian YL et al (2023) Usefulness of [68Ga]FAPI-04 and [18F]FDG PET/CT for the detection of primary tumour and metastatic lesions in gastrointestinal carcinoma: a comparative study. Eur Radiol 33:2779–2791. https://doi.org/10.1007/s00330-022-09251-y
Gao SJ, Kim AW et al (2017) Indications for invasive mediastinal staging in patients with early non-small cell lung cancer staged with PET-CT. Lung Cancer 109:36–41. https://doi.org/10.1016/j.lungcan.2017.04.018
Qi YM, Wu SS et al (2021) Development of nomograms for predicting lymph node metastasis and distant metastasis in newly diagnosed T1–2 non-small cell lung cancer: a population-based analysis. Front Oncol 11:683282. https://doi.org/10.3389/fonc.2021.683282
Real E, Aggarwal A, Huang Y, Le QV (2019) Aging evolution for image classifier architecture search. In: AAAI Conference on Artificial Intelligence, 2, p 2
Faes L, Wagner SK, Fu DJ et al (2019) Automated deep learning design for medical image classification by health-care professionals with no coding experience: a feasibility study. Lancet Digit Health 1:e232–e242. https://doi.org/10.1016/S2589-7500(19)30108-6
Yu Q, Yang D, et al. (2020) C2FNAS: Coarse–to-fine neural architecture search for 3D medical image segmentation. In: Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition, pp 4126–4135
Jiang H, Shen F, Gao F, Han W (2021) Learning efficient, explainable and discriminative representations for pulmonary nodules classification. Pattern Recogn 113:107825. https://doi.org/10.1016/j.patcog.2021.107825
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762. https://doi.org/10.1038/nrclinonc.2017.141
Bradshaw TJ, Boellaard R, Dutta J et al (2022) Nuclear medicine and artificial intelligence: best practices for algorithm development. J Nucl Med 63:500–510. https://doi.org/10.2967/jnumed.121.262567
Coroller T, Yip S, Kim J et al (2016) SU-D-207B-03: A PET-CT radiomicscomparison to predict distant metastasis in lung adenocarcinoma. J Med Phys 43:3349–3349. https://doi.org/10.1118/1.4955671
Wu J, Aguilera T, Shultz D et al (2016) Early-stage non-small cell lung cancer: quantitative imaging characteristics of (18)F fluorodeoxyglucose PET/CT allow prediction of distant metastasis. Radiology 281:270–278
Zhong Y, She Y, Multi-omics Classifier for Pulmonary Nodules (MISSION) Collaborative Group et al (2022) Deep learning for prediction of N2 metastasis and survival for clinical stage I non-small cell lung cancer. Radiology 302:200–211. https://doi.org/10.1148/radiol.2021210902
Rajpurkar P, Lungren MP (2023) The current and future state of AI interpretation of medical images. N Engl J Med 388:1981–1990. https://doi.org/10.1056/NEJMra2301725
Funding
This study was funded by grants from the National Natural Science Foundation of China (grants number 82000628 and 62176104), the Medical Scientific Research Foundation of Guangdong Province of China (grants number B2022113), and the Guangdong-Hong Kong-Macao University Joint Laboratory of Interventional Medicine Foundation of Guangdong Province (2023LSYS001).
Author information
Authors and Affiliations
Contributions
Guarantors of integrity of entire study, XYS, XBD, XHH, YBW, KWL, BXD, XMC, YW, ML, HS; study concepts/study design or data acquisition or data analysis/interpretation, all authors; manuscript drafting or manuscript revision for important intellectual content, all authors; approval of final version of submitted manuscript, all authors; agrees to ensure any questions related to the work are appropriately resolved, all authors; literature research, XYS, XBD, WY, ML, HS; clinical studies, XYS, XBD, XHH, YBW, KWL, BXD, XMC, YW, HS; experimental studies, XYS, XBD, XHH, YBW, ML, HS; statistical analysis, XYS, XBD, ML; and manuscript editing, XYS, XBD, ML, HS.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This study was approved by our institutional review board: Ethics Committee of the Fifth Affiliated Hospital of Sun Yat-sen University (approval number: [2021] K01-1). For patients in retrospective cohort, the written informed consents were waived while those of patients in prospective cohort were obtained in this study.
Consent to participate
For patients in retrospective cohort, the written informed consents were waived while those of patients in prospective cohort were obtained in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Song, X., Duan, X., He, X. et al. Computer-aided diagnosis of distal metastasis in non-small cell lung cancer by low-dose CT based radiomics and deep learning signatures. Radiol med 129, 239–251 (2024). https://doi.org/10.1007/s11547-024-01770-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-024-01770-6