Abstract
Background
Immunotherapy has revolutionized the treatment of hepatocellular carcinoma (HCC). However, whether adding immunotherapy to antiangiogenic therapy benefits patients with unresectable HCC (uHCC) more in the first-line setting remains controversial.
Objective
In this analysis, we compared the clinical outcomes of lenvatinib monotherapy with atezolizumab plus bevacizumab combination therapy in advanced uHCC in real-world clinical practice.
Methods
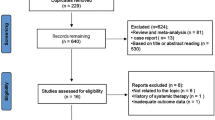
The MEDLINE, Embase, and Cochrane CENTRAL databases were systematically searched on 23 April 2023. The “metaSurvival” and “meta” packages of the R software (version 4.2.2) were used to summarize the survival curves and meta-analyze the survival data. Overall survival (OS) and progression-free survival (PFS) were defined as dual primary endpoints. Secondary endpoints included the objective response rate (ORR) and disease control rate (DCR).
Results
Overall, the pooled median OS was 18.4 months in the lenvatinib group versus 18.5 months in the atezolizumab plus bevacizumab group; the pooled median PFS was 6.9 months in the lenvatinib group versus 7.3 months in the atezolizumab plus bevacizumab group. Lenvatinib therapy showed similar OS [hazard ratio (HR): 0.91, 95% confidence interval (CI): 0.55–1.52, p = 0.72] and PFS (HR: 0.79, 95% CI: 0.56–1.12, p = 0.19) compared with atezolizumab plus bevacizumab therapy. In addition, a comparable ORR [odds ratio (OR): 0.89, 95% CI: 0.65–1.20, p = 0.44) was observed between lenvatinib and atezolizumab plus bevacizumab.
Conclusions
Comprehensive analysis suggested that lenvatinib monotherapy exhibited survival outcomes comparable to those of atezolizumab plus bevacizumab combination therapy, which may provide useful insights for clinicians in future clinical practice.






Similar content being viewed by others
References
Ducreux M, Abou-Alfa GK, Bekaii-Saab T, Berlin J, Cervantes A, de Baere T, et al. The management of hepatocellular carcinoma. Current expert opinion and recommendations derived from the 24th ESMO/world congress on gastrointestinal cancer, Barcelona, 2022. ESMO Open. 2023;8: 101567. https://doi.org/10.1016/j.esmoop.2023.101567.
Gordan JD, Kennedy EB, Abou-Alfa GK, Beg MS, Brower ST, Gade TP, et al. Systemic therapy for advanced hepatocellular carcinoma: ASCO guideline. J Clin Oncol. 2020;38:4317–45. https://doi.org/10.1200/JCO.20.02672.
Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391:1163–73. https://doi.org/10.1016/S0140-6736(18)30207-1.
Finn RS, Ikeda M, Zhu AX, Sung MW, Baron AD, Kudo M, et al. Phase Ib study of lenvatinib plus pembrolizumab in patients with unresectable hepatocellular carcinoma. J Clin Oncol. 2020;38:2960–70. https://doi.org/10.1200/JCO.20.00808.
Finn RS, Kudo M, Merle P, Meyer T, Qin S, Ikeda M, et al. LBA34 Primary results from the phase III LEAP-002 study: Lenvatinib plus pembrolizumab versus lenvatinib as first-line (1L) therapy for advanced hepatocellular carcinoma (aHCC). Ann Oncol. 2022;33:S1401. https://doi.org/10.1016/j.annonc.2022.08.031.
Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382:1894–905. https://doi.org/10.1056/NEJMoa1915745.
Cheng AL, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, et al. Updated efficacy and safety data from IMbrave150: atezolizumab plus bevacizumab vs. sorafenib for unresectable hepatocellular carcinoma. J Hepatol. 2022;76:862–73. https://doi.org/10.1016/j.jhep.2021.11.030.
Fulgenzi CAM, D’Alessio A, Airoldi C, Scotti L, Demirtas CO, Gennari A, et al. Comparative efficacy of novel combination strategies for unresectable hepatocellular carcinoma: a network metanalysis of phase III trials. Eur J Cancer. 2022;174:57–67. https://doi.org/10.1016/j.ejca.2022.06.058.
Shamseer L, Moher D, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015;350: g7647. https://doi.org/10.1136/bmj.g7647.
Wang BC, Fu C, Lin GH. The efficacy of adebrelimab compared with durvalumab and atezolizumab in untreated extensive-stage small-cell lung cancer: a survival analysis of reconstructed patient-level data. Front Immunol. 2023;14:1185577. https://doi.org/10.3389/fimmu.2023.1185577.
Jackson D, White IR, Thompson SG. Extending DerSimonian and Laird’s methodology to perform multivariate random effects meta-analyses. Stat Med. 2010;29:1282–97. https://doi.org/10.1002/sim.3602.
Jackson D, White IR, Riley RD. Quantifying the impact of between-study heterogeneity in multivariate meta-analyses. Stat Med. 2012;31:3805–20. https://doi.org/10.1002/sim.5453.
Combescure C, Foucher Y, Jackson D. Meta-analysis of single-arm survival studies: a distribution-free approach for estimating summary survival curves with random effects. Stat Med. 2014;33:2521–37. https://doi.org/10.1002/sim.6111.
Rimini M, Persano M, Tada T, Suda G, Shimose S, Kudo M, et al. Survival outcomes from atezolizumab plus bevacizumab versus lenvatinib in Child Pugh B unresectable hepatocellular carcinoma patients. J Cancer Res Clin Oncol. 2023;149(10):7565–77. https://doi.org/10.1007/s00432-023-04678-2.
Persano M, Rimini M, Tada T, Suda G, Shimose S, Kudo M, et al. Clinical outcomes with atezolizumab plus bevacizumab or lenvatinib in patients with hepatocellular carcinoma: a multicenter real-world study. J Cancer Res Clin Oncol. 2023;149(9):5591–602. https://doi.org/10.1007/s00432-022-04512-1.
Su CW, Teng W, Lin PT, Jeng WJ, Chen KA, Hsieh YC, et al. Similar efficacy and safety between lenvatinib versus atezolizumab plus bevacizumab as the first-line treatment for unresectable hepatocellular carcinoma. Cancer Med. 2023;12:7077–89. https://doi.org/10.1002/cam4.5506.
Casadei-Gardini A, Rimini M, Tada T, Suda G, Shimose S, Kudo M, et al. Atezolizumab plus bevacizumab versus lenvatinib for unresectable hepatocellular carcinoma: a large real-life worldwide population. Eur J Cancer. 2023;180:9–20. https://doi.org/10.1016/j.ejca.2022.11.017.
Niizeki T, Tokunaga T, Takami Y, Wada Y, Harada M, Shibata M, et al. Comparison of efficacy and safety of atezolizumab plus bevacizumab and lenvatinib as first-line therapy for unresectable hepatocellular carcinoma: a propensity score matching analysis. Target Oncol. 2022;17:643–53. https://doi.org/10.1007/s11523-022-00921-x.
Rimini M, Rimassa L, Ueshima K, Burgio V, Shigeo S, Tada T, et al. Atezolizumab plus bevacizumab versus lenvatinib or sorafenib in non-viral unresectable hepatocellular carcinoma: an international propensity score matching analysis. ESMO Open. 2022;7: 100591. https://doi.org/10.1016/j.esmoop.2022.100591.
Hiraoka A, Kumada T, Tada T, Hirooka M, Kariyama K, Tani J, et al. Does first-line treatment have prognostic impact for unresectable HCC?—atezolizumab plus bevacizumab versus lenvatinib. Cancer Med. 2023;12:325–34. https://doi.org/10.1002/cam4.4854.
Maesaka K, Sakamori R, Yamada R, Doi A, Tahata Y, Miyazaki M, et al. Comparison of atezolizumab plus bevacizumab and lenvatinib in terms of efficacy and safety as primary systemic chemotherapy for hepatocellular carcinoma. Hepatol Res. 2022;52:630–40. https://doi.org/10.1111/hepr.13771.
Kim BK, Cheon J, Kim H, Kang B, Ha Y, Kim DY, et al. Atezolizumab/bevacizumab vs. lenvatinib as first-line therapy for unresectable hepatocellular carcinoma: a real-world, multi-center study. Cancer. 2022;14:1747. https://doi.org/10.3390/cancers14071747.
Laethem JLV, Borbath I, Karwal M, Verslype C, VanVlierberghe H, Kardosh A, et al. 933P Updated results for pembrolizumab (pembro) monotherapy as first-line therapy for advanced hepatocellular carcinoma (HCC) in the phase II KEYNOTE-224 study. Annal Oncol. 2021;32:S819. https://doi.org/10.1016/j.annonc.2021.08.153.
Abou-Alfa GK, Chan SL, Kudo M, Lau G, Kelley RK, Furuse J, et al. Phase 3 randomized, open-label, multicenter study of tremelimumab (T) and durvalumab (D) as first-line therapy in patients (pts) with unresectable hepatocellular carcinoma (uHCC): HIMALAYA. J Clin Oncol. 2022;40:379. https://doi.org/10.1200/JCO.2022.40.4_suppl.379.
Abou-Alfa Ghassan K, Lau G, Kudo M, Chan Stephen L, Kelley Robin K, Furuse J, et al. Tremelimumab plus durvalumab in unresectable hepatocellular carcinoma. NEJM Evid. 2022. https://doi.org/10.1056/EVIDoa2100070.
Ren Z, Xu J, Bai Y, Xu A, Cang S, Du C, et al. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): a randomised, open-label, phase 2–3 study. Lancet Oncol. 2021;22:977–90. https://doi.org/10.1016/S1470-2045(21)00252-7.
Yamashita T, Kudo M, Ikeda K, Izumi N, Tateishi R, Ikeda M, et al. REFLECT-a phase 3 trial comparing efficacy and safety of lenvatinib to sorafenib for the treatment of unresectable hepatocellular carcinoma: an analysis of Japanese subset. J Gastroenterol. 2020;55:113–22. https://doi.org/10.1007/s00535-019-01642-1.
Acknowledgements
We thank Wang’s group for providing statistical support and for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this manuscript.
Conflict of Interest
Bi-Cheng Wang, Bo-Hua Kuang, and Guo-He Lin declare that they have no conflicts of interest that might be relevant to the contents of this manuscript.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Materials
All included real-world studies were searched for and downloaded from official websites. 1. https://link.springer.com/article/https://doi.org/10.1007/s00432-023-04678-2; 2. https://link.springer.com/article/https://doi.org/10.1007/s00432-022-04512-1; 3. https://onlinelibrary.wiley.com/doi/https://doi.org/10.1002/cam4.5506; 4. https://linkinghub.elsevier.com/retrieve/pii/S0959-8049(22)01762-2; 5. https://link.springer.com/article/https://doi.org/10.1007/s11523-022-00921-x; 6. https://linkinghub.elsevier.com/retrieve/pii/S2059-7029(22)00221-6; 7. https://onlinelibrary.wiley.com/doi/https://doi.org/10.1002/cam4.4854; 8. https://onlinelibrary.wiley.com/doi/https://doi.org/10.1111/hepr.13771; 9. https://www.mdpi.com/2072-6694/14/7/1747.
Code Availability
Not applicable
Author Contributions
Study design: Bi-Cheng Wang; data extraction: Bi-Cheng Wang and Guo-He Lin; data analysis: Bi-Cheng Wang and Bo-Hua Kuang; Manuscript writing and editing: all authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, BC., Kuang, BH. & Lin, GH. Lenvatinib Versus Atezolizumab Plus Bevacizumab in the First-Line Treatment for Unresectable Hepatocellular Carcinoma: A Meta-Analysis of Real-World Studies. Targ Oncol 19, 203–212 (2024). https://doi.org/10.1007/s11523-024-01035-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-024-01035-2