Abstract
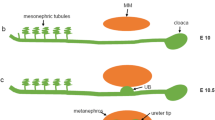
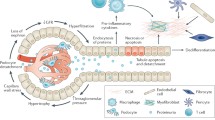
The hedgehog signaling cascade is an evolutionarily conserved pathway that regulates multiple aspects of embryonic development and plays a decisive role in tissue homeostasis. As the best studied member of three hedgehog ligands, sonic hedgehog (Shh) is known to be associated with kidney development and tissue repair after various insults. Recent studies uncover an intrinsic link between dysregulated Shh signaling and renal fibrogenesis. In various types of chronic kidney disease (CKD), Shh is upregulated specifically in renal tubular epithelium but targets interstitial fibroblasts, thereby mediating a dynamic epithelial- mesenchymal communication (EMC). Tubule-derived Shh acts as a growth factor for interstitial fibroblasts and controls a hierarchy of fibrosis-related genes, which lead to the excessive deposition of extracellular matrix in renal interstitium. In this review, we recapitulate the principle of Shh signaling, its activation and regulation in a variety of kidney diseases. We also discuss the potential mechanisms by which Shh promotes renal fibrosis and assess the efficacy of blocking this signaling in preclinical settings. Continuing these lines of investigations will provide novel opportunities for designing effective therapies to improve CKD prognosis in patients.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aberger, F., Kern, D., Greil, R., and Hartmann, T.N. (2012). Canonical and noncanonical Hedgehog/GLI signaling in hematological malignancies. Vitam Horm 88, 25–54.
Alfaro, A.C., Roberts, B., Kwong, L., Bijlsma, M.F., and Roelink, H. (2014). Ptch2 mediates the Shh response in Ptch1-/- cells. Development 141, 3331–3339.
Bielesz, B., Sirin, Y., Si, H., Niranjan, T., Gruenwald, A., Ahn, S., Kato, H., Pullman, J., Gessler, M., Haase, V.H., and Susztak, K. (2010). Epithelial Notch signaling regulates interstitial fibrosis development in the kidneys of mice and humans. J Clin Invest 120, 4040–4054.
Bolanos, A.L., Milla, C.M., Lira, J.C., Ramirez, R., Checa, M., Barrera, L., Garcia-Alvarez, J., Carbajal, V., Becerril, C., Gaxiola, M., Pardo, A., and Selman, M. (2012). Role of Sonic Hedgehog in idiopathic pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 303, L978–990.
Brennan, D., Chen, X., Cheng, L., Mahoney, M., and Riobo, N.A. (2012). Noncanonical Hedgehog signaling. Vitam Horm 88, 55–72.
Carpenter, D., Stone, D.M., Brush, J., Ryan, A., Armanini, M., Frantz, G., Rosenthal, A., and de Sauvage, F.J. (1998). Characterization of two patched receptors for the vertebrate hedgehog protein family. Proc Natl Acad Sci USA 95, 13630–13634.
Chinchilla, P., Xiao, L., Kazanietz, M.G., and Riobo, N.A. (2010). Hedgehog proteins activate pro-angiogenic responses in endothelial cells through non-canonical signaling pathways. Cell Cycle 9, 570–579.
Chung, S.I., Moon, H., Ju, H.L., Cho, K.J., do Y., Han, K.H., Eun, J.W., Nam, S.W., Ribback, S., Dombrowski, F., Calvisi, D.F., and Ro, S.W. (2016). Hepatic expression of Sonic Hedgehog induces liver fibrosis and promotes hepatocarcinogenesis in a transgenic mouse model. J Hepatol 64, 618–627.
Cigna, N., Farrokhi, Moshai, E., Brayer, S., Marchal-Somme, J., Wemeau-Stervinou, L., Fabre, A., Mal, H., Leseche, G., Dehoux, M., Soler, P., Crestani, B., and Mailleux, A.A. (2012). The hedgehog system machinery controls transforming growth factor-beta-dependent myofibroblastic differentiation in humans: involvement in idiopathic pulmonary fibrosis. Am J Pathol 181, 2126–2137.
Coresh, J., Selvin, E., Stevens, L.A., Manzi, J., Kusek, J.W., Eggers, P., Van Lente, F., and Levey, A.S. (2007). Prevalence of chronic kidney disease in the United States. JAMA 298, 2038–2047.
Dai, P., Shinagawa, T., Nomura, T., Harada, J., Kaul, S.C., Wadhwa, R., Khan, M.M., Akimaru, H., Sasaki, H., Colmenares, C., and Ishii, S. (2002). Ski is involved in transcriptional regulation by the repressor and full-length forms of Gli3. Genes Dev 16, 2843–2848.
Ding, H., Zhou, D., Hao, S., Zhou, L., He, W., Nie, J., Hou, F.F., and Liu, Y. (2012). Sonic hedgehog signaling mediates epithelial-mesenchymal communication and promotes renal fibrosis. J Am Soc Nephrol 23, 801–813.
Duffield, J.S. (2014). Cellular and molecular mechanisms in kidney fibrosis. J Clin Invest 124, 2299–2306.
El-Zaatari, M., Zavros, Y., Tessier, A., Waghray, M., Lentz, S., Gumucio, D., Todisco, A., and Merchant, J.L. (2010). Intracellular calcium release and protein kinase C activation stimulate sonic hedgehog gene expression during gastric acid secretion. Gastroenterology 139, 2061–2071 e2062.
Fabian, S.L., Penchev, R.R., St-Jacques, B., Rao, A.N., Sipila, P., West, K.A., McMahon, A.P., and Humphreys, B.D. (2012). Hedgehog-Gli pathway activation during kidney fibrosis. Am J Pathol 180, 1441–1453.
Gill, P.S., and Rosenblum, N.D. (2006). Control of murine kidney development by sonic hedgehog and its GLI effectors. Cell Cycle 5, 1426–1430.
Gonnissen, A., Isebaert, S., and Haustermans, K. (2015). Targeting the Hedgehog signaling pathway in cancer: beyond Smoothened. Oncotarget 6, 13899–13913.
Grande, M.T., Sanchez-Laorden, B., Lopez-Blau, C., De Frutos, C.A., Boutet, A., Arevalo, M., Rowe, R.G., Weiss, S.J., Lopez-Novoa, J.M., and Nieto, M.A. (2015). Snail1-induced partial epithelial-tomesenchymal transition drives renal fibrosis in mice and can be targeted to reverse established disease. Nat Med 21, 989–997.
He, W., and Dai, C. (2015). Key fibrogenic signaling. Curr Pathobiol Rep 3, 183–192.
He, W., Dai, C., Li, Y., Zeng, G., Monga, S.P., and Liu, Y. (2009). Wnt/beta-catenin signaling promotes renal interstitial fibrosis. J Am Soc Nephrol 20, 765–776.
Hirsova, P., and Gores, G.J. (2015). Ballooned hepatocytes, undead cells, sonic hedgehog, and vitamin E: therapeutic implications for nonalcoholic steatohepatitis. Hepatology 61, 15–17.
Horn, A., Palumbo, K., Cordazzo, C., Dees, C., Akhmetshina, A., Tomcik, M., Zerr P., Avouac, J., Gusinde, J., Zwerina, J., Roudaut, H., Traiffort, E., Ruat, M., Distler, O., Schett, G., and Distler, J.H. (2012). Hedgehog signaling controls fibroblast activation and tissue fibrosis in systemic sclerosis. Arthritis Rheum 64, 2724–2733.
Hu, B., Liu, J., Wu, Z., Liu, T., Ullenbruch, M.R., Ding, L., Henke, C.A., Bitterman, P.B., and Phan, S.H. (2015). Reemergence of hedgehog mediates epithelial-mesenchymal crosstalk in pulmonary fibrosis. Am J Respir Cell Mol Biol 52, 418–428.
Incardona, J.P., Gaffield, W., Kapur, R.P., and Roelink, H. (1998). The teratogenic Veratrum alkaloid cyclopamine inhibits sonic hedgehog signal transduction. Development 125, 3553–3562.
Ingham, P.W. (2012). Hedgehog signaling. Cold Spring Harb Perspect Biol 4, a011221.
Jenkins, D. (2009). Hedgehog signalling: emerging evidence for non-canonical pathways. Cell Signal 21, 1023–1034.
Johnson, R.L., Milenkovic, L., and Scott, M.P. (2000). In vivo functions of the patched protein: requirement of the C terminus for target gene inactivation but not Hedgehog sequestration. Mol Cell 6, 467–478.
Kagawa, H., Shino, Y., Kobayashi, D., Demizu, S., Shimada, M., Ariga, H., and Kawahara, H. (2011). A novel signaling pathway mediated by the nuclear targeting of C-terminal fragments of mammalian Patched 1. PLoS One 6, e18638.
Kang, H.M., Ahn, S.H., Choi, P., Ko, Y.A., Han, S.H., Chinga, F., Park, A.S., Tao, J., Sharma, K., Pullman, J., Bottinger, E.P., Goldberg, I.J., and Susztak, K. (2015). Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat Med 21, 37–46.
Katoh, Y., and Katoh, M. (2008). Hedgehog signaling, epithelial-tomesenchymal transition and miRNA. Int J Mol Med 22, 271–275.
Katoh, Y., and Katoh, M. (2009). Hedgehog target genes: mechanisms of carcinogenesis induced by aberrant hedgehog signaling activation. Curr Mol Med 9, 873–886.
Kramann, R., Fleig, S.V., Schneider, R.K., Fabian, S.L., DiRocco, D.P., Maarouf, O., Wongboonsin, J., Ikeda, Y., Heckl, D., Chang, S.L., Rennke, H.G., Waikar, S.S., and Humphreys, B.D. (2015). Pharmacological GLI2 inhibition prevents myofibroblast cell-cycle progression and reduces kidney fibrosis. J Clin Invest 125, 2935–2951.
Kramann, R., Schneider, R.K., DiRocco, D.P., Machado, F., Fleig, S., Bondzie, P.A., Henderson, J.M., Ebert, B.L., and Humphreys, B.D. (2015). Perivascular Gli1+ progenitors are key contributors to injury- induced organ fibrosis. Cell Stem Cell 16, 51–66.
Krauss, S., Concordet, J.P., and Ingham, P.W. (1993). A functionally conserved homolog of the Drosophila segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 75, 1431–1444.
Li, X., Deng, W., Lobo-Ruppert, S.M., and Ruppert, J.M. (2007). Gli1 acts through Snail and E-cadherin to promote nuclear signaling by beta- catenin. Oncogene 26, 4489–4498.
Li, Y., Wen, X., and Liu, Y. (2012). Tubular cell dedifferentiation and peritubular inflammation are coupled by the transcription regulator Id1 in renal fibrogenesis. Kidney Int 81, 880–891.
Lindstrom, E., Shimokawa, T., Toftgard, R., and Zaphiropoulos, P.G. (2006). PTCH mutations: distribution and analyses. Hum Mutat 27, 215–219.
Lipinski, R.J., Hutson, P.R., Hannam, P.W., Nydza, R.J., Washington, I.M., Moore, R.W., Girdaukas, G.G., Peterson, R.E., and Bushman, W. (2008). Dose- and route-dependent teratogenicity, toxicity, and pharmacokinetic profiles of the hedgehog signaling antagonist cyclopamine in the mouse. Toxicol Sci 104, 189–197.
Liu, Y. (2006). Renal fibrosis: new insights into the pathogenesis and therapeutics. Kidney Int 69, 213–217.
Liu, Y. (2010). New insights into epithelial-mesenchymal transition in kidney fibrosis. J Am Soc Nephrol 21, 212–222.
Liu, Y. (2011). Cellular and molecular mechanisms of renal fibrosis. Nat Rev Nephrol 7, 684–696.
Lovisa, S., LeBleu, V.S., Tampe, B., Sugimoto, H., Vadnagara, K., Carstens, J.L., Wu, C.C., Hagos, Y., Burckhardt, B.C., Pentcheva-Hoang, T., Nischal, H., Allison, J.P., Zeisberg, M., and Kalluri, R. (2015). Epithelial- to-mesenchymal transition induces cell cycle arrest and parenchymal damage in renal fibrosis. Nat Med 21, 998–1009.
Lum, L., and Beachy, P.A. (2004). The Hedgehog response network: sensors, switches, and routers. Science 304, 1755–1759.
Makino, S., Masuya, H., Ishijima, J., Yada, Y., and Shiroishi, T. (2001). A spontaneous mouse mutation, mesenchymal dysplasia (mes), is caused by a deletion of the most C-terminal cytoplasmic domain of patched (ptc). Dev Biol 239, 95–106.
McGowan, S.E., and McCoy, D.M. (2013). Platelet-derived growth factor- A and sonic hedgehog signaling direct lung fibroblast precursors during alveolar septal formation. Am J Physiol Lung Cell Mol Physiol 305, L229–239.
Meng, X.M., Tang, P.M., Li, J., and Lan, H.Y. (2015). TGF-beta/Smad signaling in renal fibrosis. Front Physiol 6, 82.
Nieuwenhuis, E., Barnfield, P.C., Makino, S., and Hui, C.C. (2007). Epidermal hyperplasia and expansion of the interfollicular stem cell compartment in mutant mice with a C-terminal truncation of Patched1. Dev Biol 308, 547–560.
Nusslein-Volhard, C., and Wieschaus, E. (1980). Mutations affecting segment number and polarity in Drosophila. Nature 287, 795–801.
O’Hara, W.A., Azar, W.J., Behringer, R.R., Renfree, M.B., and Pask, A.J. (2011). Desert hedgehog is a mammal-specific gene expressed during testicular and ovarian development in a marsupial. BMC Dev Biol 11, 72.
Ochi, H., Pearson, B.J., Chuang, P.T., Hammerschmidt, M., and Westerfield, M. (2006). Hhip regulates zebrafish muscle development by both sequestering Hedgehog and modulating localization of Smoothened. Dev Biol 297, 127–140.
Pan, A., Chang, L., Nguyen, A., and James, A.W. (2013). A review of hedgehog signaling in cranial bone development. Front Physiol 4, 61.
Polizio, A.H., Chinchilla, P., Chen, X., Manning, D.R., and Riobo, N.A. (2011). Sonic Hedgehog activates the GTPases Rac1 and RhoA in a Gli-independent manner through coupling of smoothened to Gi proteins. Sci Signal 4, pt7.
Rangwala, F., Guy, C.D., Lu, J., Suzuki, A., Burchette, J.L., Abdelmalek, M.F., Chen, W., and Diehl, A.M. (2011). Increased production of sonic hedgehog by ballooned hepatocytes. J Pathol 224, 401–410.
Rauhauser, A.A., Ren, C., Lu, D., Li, B., Zhu, J., McEnery, K., Vadnagara, K., Zepeda-Orozco, D., Zhou, X.J., Lin, F., Jetten, A.M., and Attanasio, M. (2015). Hedgehog signaling indirectly affects tubular cell survival after obstructive kidney injury. Am J Physiol Renal Physiol 309, F770–778.
Reichrath, J., and Reichrath, S. (2013). The relevance of the vitamin D endocrine system (VDES) for tumorigenesis, prevention, and treatment of non-melanoma skin cancer (NMSC): present concepts and future perspectives. Dermatoendocrinol 5, 38–50.
Robarge, K.D., Brunton, S.A., Castanedo, G.M., Cui, Y., Dina, M.S., Goldsmith, R., Gould, S.E., Guichert, O., Gunzner, J.L., Halladay, J., Jia, W., Khojasteh, C., Koehler, M.F., Kotkow, K., La H., Lalonde, R.L., Lau, K., Lee, L., Marshall, D., Marsters, J.C., Murray, L.J., Qian, C., Rubin, L.L., Salphati, L., Stanley, M.S., Stibbard, J.H., Sutherlin, D.P., Ubhayaker, S., Wang, S., Wong, S., and Xie, M. (2009). GDC-0449-a potent inhibitor of the hedgehog pathway. Bioorg Med Chem Lett 19, 5576–5581.
Robbins, D.J., Fei, D.L., and Riobo, N.A. (2012). The Hedgehog signal transduction network. Sci Signal 5, re6.
Ruat, M. (2015). The smoothened receptor in cancer and regenerative medicine. In Topics in Medicinal Chemistry (New York: Springer Berlin Heidelberg).
Ruiz-Gomez, A., Molnar, C., Holguin, H., Mayor, F., and de Celis, J.F. (2007). The cell biology of Smo signalling and its relationships with GPCRs. Biochim Biophys Acta 1768, 901–912.
Santos, P.C., Krieger, J.E., and Pereira, A.C. (2012). Renin-angiotensin system, hypertension, and chronic kidney disease: pharmacogenetic implications. J Pharmacol Sci 120, 77–88.
Strand, M.F., Wilson, S.R., Dembinski, J.L., Holsworth, D.D., Khvat, A., Okun, I., Petersen, D., and Krauss, S. (2011). A novel synthetic smoothened antagonist transiently inhibits pancreatic adenocarcinoma xenografts in a mouse model. PLoS One 6, e19904.
Sweetwyne, M.T., Tao, J., and Susztak, K. (2014). Kick it up a notch: Notch signaling and kidney fibrosis. Kidney Int Suppl 4, 91–96.
Tan, R.J., Zhou, D., Zhou, L., and Liu, Y. (2014). Wnt/beta-catenin signaling and kidney fibrosis. Kidney Int Suppl 4, 84–90.
Teperino, R., Aberger, F., Esterbauer, H., Riobo, N., and Pospisilik, J.A. (2014). Canonical and non-canonical Hedgehog signalling and the control of metabolism. Semin Cell Dev Biol 33, 81–92.
Thibert, C., Teillet, M.A., Lapointe, F., Mazelin, L., Le Douarin, N.M., and Mehlen, P. (2003). Inhibition of neuroepithelial patched-induced apoptosis by sonic hedgehog. Science 301, 843–846.
Tukachinsky, H., Lopez, L.V., and Salic, A. (2010). A mechanism for vertebrate Hedgehog signaling: recruitment to cilia and dissociation of SuFu-Gli protein complexes. J Cell Biol 191, 415–428.
Wang, D., Dai, C., Li, Y., and Liu, Y. (2011). Canonical Wnt/beta-catenin signaling mediates transforming growth factor-beta1-driven podocyte injury and proteinuria. Kidney Int 80, 1159–1169.
Yam, P.T., Langlois, S.D., Morin, S., and Charron, F. (2009). Sonic hedgehog guides axons through a noncanonical, Src-family-kinasedependent signaling pathway. Neuron 62, 349–362.
Yang, L., Wang, Y., Mao, H., Fleig, S., Omenetti, A., Brown, K.D., Sicklick, J.K., Li, Y.X., and Diehl, A.M. (2008). Sonic hedgehog is an autocrine viability factor for myofibroblastic hepatic stellate cells. J Hepatol 48, 98–106.
Yu, J., Carroll, T.J., and McMahon, A.P. (2002). Sonic hedgehog regulates proliferation and differentiation of mesenchymal cells in the mouse metanephric kidney. Development 129, 5301–5312.
Zeisberg, M., and Neilson, E.G. (2010). Mechanisms of tubulointerstitial fibrosis. J Am Soc Nephrol 21, 1819–1834.
Zhang, L., Wang, F., Wang, L., Wang, W., Liu, B., Liu, J., Chen, M., He, Q., Liao, Y., Yu, X., Chen, N., Zhang, J.E., Hu, Z., Liu, F., Hong, D., Ma, L., Liu, H., Zhou, X., Chen, J., Pan, L., Chen, W., Wang, W., Li, X., and Wang, H. (2012). Prevalence of chronic kidney disease in China: a cross-sectional survey. Lancet 379, 815–822.
Zhao, C., Chen, A., Jamieson, C.H., Fereshteh, M., Abrahamsson, A., Blum, J., Kwon, H.Y., Kim, J., Chute, J.P., Rizzieri, D., Munchhof, M., VanArsdale, T., Beachy, P.A., and Reya, T. (2009). Hedgehog signalling is essential for maintenance of cancer stem cells in myeloid leukaemia. Nature 458, 776–779.
Zhao, Q., Beck, A., and Fraidenraich, D. (2010). A direct, non-canonical pathway for Hedgehog proteins in the endothelium. Cell Cycle 9, 647–648.
Zhao, X.P., Liao, M.C., Chang, S.Y., Abdo, S., Aliou, Y., Chenier, I., Ingelfinger, J.R., and Zhang, S.L. (2014). Maternal diabetes modulates kidney formation in murine progeny: the role of hedgehog interacting protein (HHIP). Diabetologia 57, 1986–1996.
Zhou, D., Li, Y., Zhou, L., Tan, R.J., Xiao, L., Liang, M., Hou, F.F., and Liu, Y. (2014). Sonic hedgehog is a novel tubule-derived growth factor for interstitial fibroblasts after kidney injury. J Am Soc Nephrol 25, 2187–2200.
Zhou, D., Li, Y., Lin, L., Zhou, L., Igarashi, P., and Liu, Y. (2012). Tubule- specific ablation of endogenous beta-catenin aggravates acute kidney injury in mice. Kidney Int 82, 537–547.
Zhou, D., and Liu, Y. (2016). Renal fibrosis in 2015: understanding the mechanisms of kidney fibrosis. Nat Rev Nephrol 12, 68–70.
Zhou, D., Tan, R.J., Fu, H., and Liu, Y. (2016). Wnt/beta-catenin signaling in kidney injury and repair: a double-edged sword. Lab Invest 96, 156–167.
Zhou, L., Li, Y., Hao, S., Zhou, D., Tan, R.J., Nie, J., Hou, F.F., Kahn, M., and Liu, Y. (2015). Multiple genes of the renin-angiotensin system are novel targets of Wnt/beta-catenin signaling. J Am Soc Nephrol 26, 107–120.
Zhou, L., and Liu, Y. (2015). Wnt/beta-catenin signalling and podocyte dysfunction in proteinuric kidney disease. Nat Rev Nephrol 11, 535–545.
Zhulyn, O., Nieuwenhuis, E., Liu, Y.C., Angers, S., and Hui, C.C. (2015). Ptch2 shares overlapping functions with Ptch1 in Smo regulation and limb development. Dev Biol 397, 191–202.
Zoccali, C., Kramer, A., and Jager, K.J. (2010). Epidemiology of CKD in Europe: an uncertain scenario. Nephrol Dial Transplant 25, 1731–1733.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at link.springer.com
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhou, D., Tan, R.J. & Liu, Y. Sonic hedgehog signaling in kidney fibrosis: a master communicator. Sci. China Life Sci. 59, 920–929 (2016). https://doi.org/10.1007/s11427-016-0020-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-016-0020-y