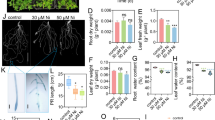
Abstract
Effectively controlling target organisms while reducing the adverse effects of pesticides on non-target organisms is a crucial scientific inquiry and challenge in pesticide ecotoxicology research. Here, we studied the alleviation of herbicide (R)-imazethapyr [(R)-IM] to non-target plant wheat by active regulation between auxin and secondary metabolite 2,4-dihydroxy-7-methoxy-2H-1,4-benzoxazine-3(4H)-one (DIMBOA). We found (R)-IM reduced 32.4% auxin content in wheat leaves and induced 40.7% DIMBOA accumulation compared to the control group, which effortlessly disrupted the balance between wheat growth and defense. Transcriptomic results indicated that restoration of the auxin level in plants promoted the up-regulation of growth-related genes and the accumulation of DIMBOA up-regulated the expression of defense-related genes. Auxin and DIMBOA alleviated herbicide stress primarily through effects in the two directions of wheat growth and defense, respectively. Additionally, as a common precursor of auxin and DIMBOA, indole adopted a combined growth and defense strategy in response to (R)-IM toxicity, i.e., restoring growth development and enhancing the defense system. Future regulation of auxin and DIMBOA levels in plants may be possible through appropriate methods, thus regulating the plant growth-defense balance under herbicide stress. Our insight into the interference mechanism of herbicides to the plant growth-defense system will facilitate the design of improved strategies for herbicide detoxification.
Graphical Abstract







Similar content being viewed by others
Data availability
All data underlying the results are generated and analyzed by the authors and are available as part of the article.
References
Abel S, Theologis A (2010) Odyssey of auxin. Csh Perspect Biol 2:a004572. https://doi.org/10.1101/cshperspect.a004572
Ahmad S, Veyrat N, Gordon-Weeks R, Zhang Y, Martin J, Smart L, Glauser G, Erb M, Flors V, Frey M (2011) Benzoxazinoid metabolites regulate innate immunity against aphids and fungi in maize. Plant Physiol 157(1):317–327. https://doi.org/10.1104/pp.111.180224
Amir SB, Rasheed R, Ashraf MA, Hussain I, Iqbal M (2020) Hydrogen sulfide mediates defense response in safflower by regulating secondary metabolism, oxidative defense, and elemental uptake under drought. Physiol Plantarum 172(2):795–808. https://doi.org/10.1111/ppl.13267
Baerson SR, Sánchez-Moreiras A, Pedrol-Bonjoch N, Schulz M, Kagan IA, Agarwal AK, Reigosa MJ, Duke SO (2005) Detoxification and transcriptome response in Arabidopsis seedlings exposed to the allelochemical benzoxazolin-2(3H)-one. J Biol Chem 280(23):21867–21881. https://doi.org/10.1074/jbc.m500694200
Bao M, Li J, Chen H, Chen Z, Xu D, Wen Y (2021) Enantioselective effects of imazethapyr on the secondary metabolites and nutritional value of wheat seedlings. Sci Total Environ 757:143759. https://doi.org/10.1016/j.scitotenv.2020.143759
Barbez E, Dünser K, Gaidora A, Lendl T, Busch W (2017) Auxin steers root cell expansion via apoplastic pH regulation in Arabidopsis thaliana. P Natl Acad Sci 114(24):E4884–E4893. https://doi.org/10.1073/pnas.1613499114
Brumos J, Robles LM, Yun J, Vu TC, Jackson S, Alonso JM, Stepanova AN (2018) Local auxin biosynthesis is a key regulator of plant development. Dev cell 47(3):306–318. https://doi.org/10.1016/j.devcel.2018.09.022
Chen H, Zhang S, Wu K, Li R, He X, He D, Huang C, Wei H (2020) The effects of exogenous organic acids on the growth, photosynthesis and cellular ultrastructure of Salix variegata Franch. Under Cd stress. Ecotox Environ Safe 187(15):109790. https://doi.org/10.1016/j.ecoenv.2019.109790
Clay NK, Adio AM, Denoux C, Jander G, Ausubel FM (2009) Glucosinolate metabolites required for an Arabidopsis innate immune response. Science 323:95–101. https://doi.org/10.1126/science.1164627
Dalal VK, Tripathy BC (2018) Water-stress induced downsizing of light-harvesting antenna complex protects developing rice seedlings from photo-oxidative damage. Sci Rep 8(5955):1–16. https://doi.org/10.1038/s41598-017-14419-4
Dixon SJ, Stockwell BR (2014) The role of iron and reactive oxygen species in cell death. Nat Chem Biol 10:9–17. https://doi.org/10.1038/nchembio.1416
Du M, Spalding EP, Gray WM (2020) Rapid auxin-mediated cell expansion. Annu Rev Plant Biol 71:379. https://doi.org/10.1146/annurev-arplant-073019-025907
Gomes MP, Manac’h L, Sarah G, Hénault-Ethier L, Labrecque M, Lucotte M, Juneau P (2017) Glyphosate-dependent inhibition of photosynthesis in willow. Front Plant Sci 8:207. https://doi.org/10.3389/fpls.2017.00207
Govindasamy P, Singh V, Palsaniya D, Srinivasan R, Chaudhary M, Kantwa S (2021) Herbicide effect on weed control, soil health parameters and yield of Egyptian clover (Trifolium alexandrinum L.). Crop Prot 139:105389. https://doi.org/10.1016/j.cropro.2020.105389
Grones P, Chen X, Simon S, Kaufmann WA, De Rycke R, Nodzyński T, Zažímalová E, Friml J (2015) Auxin-binding pocket of ABP1 is crucial for its gain-of-function cellular and developmental roles. J Exp Bot 66(16):5055–5065. https://doi.org/10.1093/jxb/erv177
Guo Q, Major IT, Howe GA (2018) Resolution of growth-defense conflict: mechanistic insights from jasmonate signaling. Curr Opin Plant Biol 44:72–81. https://doi.org/10.1016/j.pbi.2018.02.009
Hu L, Mateo P, Ye M, Zhang X, Berset JD, Handrick V, Radisch D, Grabe V, Koellner TG, Gershenzon J, Robert CAM, Erb M (2018) Plant iron acquisition strategy exploited by an insect herbivore. Science 361:694–697. https://doi.org/10.1126/science.aat4082
Hu L, Wu Z, Robert CA, Ouyang X, Züst T, Mestrot A, Xu J, Erb M (2021) Soil chemistry determines whether defensive plant secondary metabolites promote or suppress herbivore growth. P Natl Acad Sci 118(43):e2109602118. https://doi.org/10.1073/pnas.2109602118
Hu Y, Vandenbussche F, Van Der Straeten D (2017) Regulation of seedling growth by ethylene and the ethylene–auxin crosstalk. Planta 245:467–489. https://doi.org/10.1007/s00425-017-2651-6
Huang J, Bao M, Li J, Chen H, Xu D, Chen Z, Wen Y (2022) Enantioselective Response of Wheat Seedlings to Imazethapyr: From the Perspective of Fe and the Secondary Metabolite DIMBOA. J Agr Food Chem 70(18):5516–5525. https://doi.org/10.1021/acs.jafc.1c07727
Itkin M, Rogachev I, Alkan N, Rosenberg T, Malitsky S, Masini L, Meir S, Iijima Y, Aoki K, de Vos R (2011) GLYCOALKALOID METABOLISM1 is required for steroidal alkaloid glycosylation and prevention of phytotoxicity in tomato. Plant Cell 23(12):4507–4525. https://doi.org/10.1105/tpc.111.088732
Jin J, Wang M, Lu W, Zhang L, Jiang Q, Jin Y, Lu K, Sun S, Cao Q, Wang Y (2019) Effect of plants and their root exudate on bacterial activities during rhizobacterium–plant remediation of phenol from water. Environ Int 127:114–124. https://doi.org/10.1016/j.envint.2019.03.015
Kong C, Zhang S, Li Y, Xia Z, Yang X, Meiners SJ, Wang P (2018) Plant neighbor detection and allelochemical response are driven by root-secreted signaling chemicals. Nat Commun 9:1–9. https://doi.org/10.1038/s41467-018-06429-1
Li L, Verstraeten I, Roosjen M, Takahashi K, Rodriguez L, Merrin J, Chen J, Shabala L, Smet W, Ren H (2021) Cell surface and intracellular auxin signalling for H+ fluxes in root growth. Nature 599:273–277. https://doi.org/10.1038/s41586-021-04037-6
Lin W, Zhou X, Tang W, Takahashi K, Pan X, Dai J, Ren H, Zhu X, Pan S, Zheng H (2021) TMK-based cell-surface auxin signalling activates cell-wall acidification. Nature 599:278–282. https://doi.org/10.1038/s41586-021-03976-4
Lin X, Ye Y, Fan S, Jin C, Zheng S (2016) Increased sucrose accumulation regulates iron-deficiency responses by promoting auxin signaling in Arabidopsis plants. Plant physiol 170(2):907–920. https://doi.org/10.1104/pp.15.01598
Liu X, Du M, Wang Y, Liu S, Liu X (2018) BMP9 overexpressing adipose-derived mesenchymal stem cells promote cartilage repair in osteoarthritis-affected knee joint via the Notch1/Jagged1 signaling pathway. Exp Ther Med 16(6):4623–4631. https://doi.org/10.3892/etm.2018.6754
Mironczuk-Chodakowska I, Witkowska AM, Zujko ME (2018) Endogenous non-enzymatic antioxidants in the human body. Adv Med Sci 63(1):68–78. https://doi.org/10.1016/j.advms.2017.05.005
Moraes MCB, Birkett MA, Gordon-Weeks R, Smart LE, Martin JL, Pye BJ, Bromilow R, Pickett JA (2008) cis-Jasmone induces accumulation of defence compounds in wheat, Triticum aestivum. Phytochemistry 69(1):9–17. https://doi.org/10.1016/j.phytochem.2007.06.020
Nam H-I, Shahzad Z, Dorone Y, Clowez S, Zhao K, Bouain N, Lay-Pruitt KS, Cho H, Rhee SY, Rouached H (2021) Interdependent iron and phosphorus availability controls photosynthesis through retrograde signaling. Nat Commun 12:1–13. https://doi.org/10.1038/s41467-021-27548-2
Niemeyer HM (2009) Hydroxamic acids derived from 2-hydroxy-2H-1,4-benzoxazin-3(4H)-one: key defense chemicals of cereals. J Agr Food Chem 57(5):1677–1696. https://doi.org/10.1021/jf8034034
Powles SB, Yu Q (2010) Evolution in action: plants resistant to herbicides. Annu Rev Plant Biol 61:317–347. https://doi.org/10.1146/annurev-arplant-042809-112119
Qian H, Hu H, Mao Y, Ma J, Zhang A, Liu W, Fu Z (2009) Enantioselective phytotoxicity of the herbicide imazethapyr in rice. Chemosphere 76(7):885–892. https://doi.org/10.1016/j.scitotenv.2020.143759
Qu Q, Zhang Z, Li Y, Zhou Z, Ye Y, Lu T, Sun L, Qian H (2019) Comparative molecular and metabolic responses of wheat seedlings (Triticum aestivum L.) to the imazethapyr enantiomers S-IM and R-IM. Sci Total Environ 692:723–731. https://doi.org/10.1016/j.scitotenv.2019.07.333
Richter A, Powell AF, Mirzaei M, Wang LJ, Movahed N, Miller JK, Piñeros MA, Jander G (2021) Indole-3-glycerolphosphate synthase, a branchpoint for the biosynthesis of tryptophan, indole, and benzoxazinoids in maize. Plant J 106(1):245–257. https://doi.org/10.1111/tpj.15163
Schulz R, Bub S, Petschick LL, Stehle S, Wolfram J (2021) Applied pesticide toxicity shifts toward plants and invertebrates, even in GM crops. Science 372:81–84. https://doi.org/10.1126/science.abe1148
Sharma A, Kumar V, Shahzad B, Ramakrishnan M, Singh Sidhu GP, Bali AS, Handa N, Kapoor D, Yadav P, Khanna K (2020) Photosynthetic response of plants under different abiotic stresses: a review. J Plant Growth Regul 39:509–531. https://doi.org/10.1007/s00344-019-10018-x
Shi W, Han Y, Sun S, Tang Y, Zhou W, Du X, Liu G (2020) Immunotoxicities of microplastics and sertraline, alone and in combination, to a bivalve species: size-dependent interaction and potential toxication mechanism. J Hazard Mater 396(5):122603. https://doi.org/10.1016/j.jhazmat.2020.122603
Singh A, Singh S, Singh R, Kumar S, Singh SK, Singh IK (2021) Dynamics of Zea mays transcriptome in response to a polyphagous herbivore, Spodoptera litura. Funct Integr Genomic 21:571–592. https://doi.org/10.1007/s10142-021-00796-7
Sroka Z, Zbikowska B, Janicki K, Franiczek R, Krzyzanowska B, Drys A (2014) Antimicrobial and antiradical activity of extracts obtained from leaves of three species of the genus Pyrus. Microb Drug Resist 20(4):337–343. https://doi.org/10.1089/mdr.2013.0155
Su P, Zhao L, Li W, Zhao J, Yan J, Ma X, Li A, Wang H, Kong L (2021) Integrated metabolo-transcriptomics and functional characterization reveals that the wheat auxin receptor TIR1 negatively regulates defense against Fusarium graminearum. J Integr Plant Biol 63(2):340–352. https://doi.org/10.1111/jipb.12992
Sun X, Zhang J, Wang X, Zhao J, Pan W, Yu G, Qu Y, Wang J (2020) Colorimetric and fluorimetric dual mode detection of Fe2+ in aqueous solution based on a carbon dots/phenanthroline system. Arab J Chem 13(4):5075–5083. https://doi.org/10.1016/j.arabjc.2020.02.007
Wang X, Wang N, Rui Q, Zhang P, Xu L (2013) Jasmonates modulate the promotion effects induced by SNP on root development of wheat under osmotic stress through lipoxygenase activation. J Plant Biochem Biot 22:295–303. https://doi.org/10.1007/s13562-012-0158-6
Wang X, Li Q, Xie J, Huang M, Cai J, Zhou Q, Dai T, Jiang D (2021) Abscisic acid and jasmonic acid are involved in drought priming-induced tolerance to drought in wheat. Crop J 9(1):120–132. https://doi.org/10.1016/j.cj.2020.06.002
Wu H, Chen H, Jin C, Tang C, Zhang Y (2019) The chirality of imazethapyr herbicide selectively affects the bacterial community in soybean field soil. Environ Sci Pollut R 26:2531–2546. https://doi.org/10.1007/s11356-018-3736-x
Wu Y, Ma L, Liu Q, Vestergård M, Topalovic O, Wang Q, Zhou Q, Huang L, Yang X, Feng Y (2020) The plant-growth promoting bacteria promote cadmium uptake by inducing a hormonal crosstalk and lateral root formation in a hyperaccumulator plant Sedum alfredii. J Hazard Mater 395(5):122661. https://doi.org/10.1016/j.jhazmat.2020.122661
Ye J, Zhong T, Zhang D, Ma C, Wang L, Yao L, Zhang Q, Zhu M, Xu M (2019) The auxin-regulated protein ZmAuxRP1 coordinates the balance between root growth and stalk rot disease resistance in maize. Mol Plant 12(3):360–373. https://doi.org/10.1016/j.molp.2018.10.005
Yuan J, Tranel PJ, Stewart CN Jr (2007) Non-target-site herbicide resistance: a family business. Trends Plant Sci 12(1):6–13. https://doi.org/10.1016/j.tplants.2006.11.001
Yuan S, Kim SC, Deng X, Hong Y, Wang X (2019) Diacylglycerol kinase and associated lipid mediators modulate rice root architecture. New Phytol 223(1):261–276. https://doi.org/10.1111/nph.15801
Zhang H, Zhao Y, Zhu J-K (2020) Thriving under stress: how plants balance growth and the stress response. Dev Cell 55(5):529–543. https://doi.org/10.1016/j.devcel.2020.10.012
Zhang M, Wang Y, Chen X, Xu F, Ding M, Ye W, Kawai Y, Toda Y, Hayashi Y, Suzuki T (2021) Plasma membrane H+-ATPase overexpression increases rice yield via simultaneous enhancement of nutrient uptake and photosynthesis. Nat Commun 12:1–12. https://doi.org/10.1038/s41467-021-20964-4
Zhang Y, Huang L, Liu L, Cao X, Sun C, Lin X (2022) Metabolic disturbance in lettuce (Lactuca sativa) plants triggered by imidacloprid and fenvalerate. Sci Total Environ 802:149764. https://doi.org/10.1016/j.scitotenv.2021.149764
Zhao Z, Gao X, Ke Y, Chang M, Xie L, Li X, Gu M, Liu J, Tang X (2019) A unique aluminum resistance mechanism conferred by aluminum and salicylic-acid-activated root efflux of benzoxazinoids in maize. Plant Soil 437:273–289. https://doi.org/10.1007/s11104-019-03971-9
Zust T, Agrawal AA (2016) Mechanisms and evolution of plant resistance to aphids. Nat Plants 2:15206. https://doi.org/10.1038/nplants.2015.206
Funding
This study was supported by the National Natural Science Foundation of China (Nos. 22176174 and 22006078).
Author information
Authors and Affiliations
Contributions
Hui Chen: methodology, investigation, formal analysis, and writing original draft; Jinye Huang: methodology, investigation, formal analysis, and writing original draft; Jun Li: validation and editing; Chensi Shen: validation and review; Yuezhong Wen: resources, supervision, validation, writing, review, and editing.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Gangrong Shi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
The Supplementary Information is available free of charge. Primer sequences for 10 DEGs and the internal reference gene β-actin (Table S1). Effects of different exogenous treatments on the phenotype of wheat seedlings (Fig. S1). Effects of treatments on the growth index of wheat seedlings (Fig. S2). Effects of IAA, DIMBOA, and indole on growth and defense systems under (R)-IM stress (Fig. S3). Real-time qPCR validation of differentially expressed genes (Fig. S4). Regulation of plasma membrane H+-ATPase activity on wheat seedlings (Fig. S5). (DOCX 317 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, ., Li, J., Chen, H. et al. Phytotoxicity alleviation of imazethapyr to non-target plant wheat: active regulation between auxin and DIMBOA. Environ Sci Pollut Res 30, 116004–116017 (2023). https://doi.org/10.1007/s11356-023-30608-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30608-3