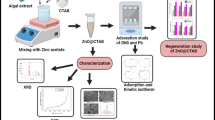
Abstract
Heavy metal pollution in wastewater poses a grave danger to the environment and the human body. Pumice is a mineral with abundant reserves and low prices, and its prospect of heavy metal adsorbent is very broad. In this work, we modified pumice with basic magnesium carbonate nanosheets by a convenient hydrothermal synthesis. The adsorption capacity of heavy metals is greatly improved. The effects of different pH and adsorption dosages are investigated. All the optimum pH values for Cu2+, Pb2+, and Cd2+ are 5. The adsorption of three kinds of ions conforms to the quasi-second-order adsorption kinetics model. The theoretical adsorption capacities of Cu2+, Pb2+, and Cd2+, which are calculated by the Langmuir model, are 235.29 mg/L, 595.24 mg/L, and 370.34 mg/L, respectively. The adsorption of Cu2+ and Cd2+ fit the Langmuir model better. The Freundlich model is fitted well with the adsorption of Pb2+. In the experiment simulating real wastewater, the adsorption capacity of heavy metals is not affected. It also shows good reusability in three regeneration cycles. And Mg5(CO3)4(OH)2·4H2O@pumice adsorption column showed the good removal efficiency of three heavy metals at different concentrations and different spatial velocities in the column experiment. Thus, it is believed that the Mg5(CO3)4(OH)2·4H2O@pumice is a promising adsorbent for the efficient removal of heavy metals.















Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Aljerf L (2018) High-efficiency extraction of bromocresol purple dye and heavy metals as chromium from industrial effluent by adsorption onto a modified surface of zeolite: Kinetics and equilibrium study. J Environ Manage 225:120–132. https://doi.org/10.1016/j.jenvman.2018.07.048
Amin MS, Hashem FS, El-Gamal SMA (2018) Utilization of OPC-Pumice composites for efficient heavy metals removal. J Taibah Univ Sci 12:765–773. https://doi.org/10.1080/16583655.2018.1510150
Asere TG, De Clercq J, Verbeken K et al (2017) Uptake of arsenate by aluminum (hydr)oxide coated red scoria and pumice. Appl Geochem 78:83–95. https://doi.org/10.1016/j.apgeochem.2016.12.013
Asere TG, Stevens CV, Du Laing G (2019) Use of (modified) natural adsorbents for arsenic remediation: a review. Sci Total Environ 676:706–720. https://doi.org/10.1016/j.scitotenv.2019.04.237
Baby R, Saifullah B, Hussein MZ (2019) Palm kernel shell as an effective adsorbent for the treatment of heavy metal contaminated water. Sci Rep 9:18955. https://doi.org/10.1038/s41598-019-55099-6
Bello OS, Adegoke KA, Oyewole RO (2014) Insights into the adsorption of heavy metals from wastewater using diatomaceous Earth. Sep Sci Technol 49:1787–1806. https://doi.org/10.1080/01496395.2014.910223
Can N, Ömür BC, Altındal A (2016) Modeling of heavy metal ion adsorption isotherms onto metallophthalocyanine film. Sens Actuators B Chem 237:953–961. https://doi.org/10.1016/j.snb.2016.07.026
Chakraborty B, Bera B, Roy S et al (2021) Assessment of non-carcinogenic health risk of heavy metal pollution: evidences from coal mining region of eastern India. Environ Sci Pollut Res 28:47275–47293. https://doi.org/10.1007/s11356-021-14012-3
Chen Q, Yao Y, Li X et al (2018) Comparison of heavy metal removals from aqueous solutions by chemical precipitation and characteristics of precipitates. J Water Process Eng 26:289–300. https://doi.org/10.1016/j.jwpe.2018.11.003
Chi T, Zuo J, Liu F (2017) Performance and mechanism for cadmium and lead adsorption from water and soil by corn straw biochar. Front Environ Sci Eng 11:15. https://doi.org/10.1007/s11783-017-0921-y
Çifçi Dİ, Meriç S (2017) Manganese adsorption by iron impregnated pumice composite. Colloids Surf Physicochem Eng Asp 522:279–286. https://doi.org/10.1016/j.colsurfa.2017.03.004
Ciosek AL, Luk GK (2017) An innovative dual-column system for heavy metallic ion sorption by natural zeolite. Appl Sci 7:795. https://doi.org/10.3390/app7080795
Dursun AY (2006) A comparative study on determination of the equilibrium, kinetic and thermodynamic parameters of biosorption of copper(II) and lead(II) ions onto pretreated Aspergillus niger. Biochem Eng J 28:187–195. https://doi.org/10.1016/j.bej.2005.11.003
El-Safty SA, Shenashen MA, Ismael M et al (2012) Optical mesosensors for monitoring and removal of ultra-trace concentration of Zn(ii) and Cu(ii) ions from water. Analyst 137:5278. https://doi.org/10.1039/c2an35484e
Fang Y, Wen J, Zeng G et al (2018) From nZVI to SNCs: development of a better material for pollutant removal in water. Environ Sci Pollut Res 25:6175–6195. https://doi.org/10.1007/s11356-017-1143-3
Figas A, Tomaszewska-Sowa M, Kobierski M et al (2021) Hazard of contamination with heavy metals in Thymus serpyllum L. Herbs from rural areas. Agriculture 11:375. https://doi.org/10.3390/agriculture11040375
Gevorgyan G, Mamyan A, Boshyan T et al (2021) Heavy metal contamination in an industrially affected river catchment basin: assessment, effects, and mitigation. Int J Environ Res Public Health 18:2881. https://doi.org/10.3390/ijerph18062881
Ghosal PS, Gupta AK (2015) An insight into thermodynamics of adsorptive removal of fluoride by calcined Ca–Al–(NO3) layered double hydroxide. RSC Adv 5:105889–105900. https://doi.org/10.1039/C5RA20538G
Gomaa H, Shenashen MA, Elbaz A et al (2021) Mesoscopic engineering materials for visual detection and selective removal of copper ions from drinking and waste water sources. J Hazard Mater 406:124314. https://doi.org/10.1016/j.jhazmat.2020.124314
Hong Y-K, Kim J-W, Kim H-S et al (2021) Bottom ash modification via sintering process for its use as a potential heavy metal adsorbent: sorption kinetics and mechanism. Materials 14:3060. https://doi.org/10.3390/ma14113060
Huang Q, Chen Y, Yu H et al (2018) Magnetic graphene oxide/MgAl-layered double hydroxide nanocomposite: one-pot solvothermal synthesis, adsorption performance and mechanisms for Pb2+, Cd2+, and Cu2+. Chem Eng J 341:1–9. https://doi.org/10.1016/j.cej.2018.01.156
Jiang D, Yu Q, Huang C et al (2019) Preparation of mesoporous spherical magnesium hydroxide particles via the static self-assembled method. J Mol Struct 1175:858–864. https://doi.org/10.1016/j.molstruc.2018.08.058
Jin Z, Jia Y, Zhang K-S et al (2016) Effective removal of fluoride by porous MgO nanoplates and its adsorption mechanism. J Alloys Compd 675:292–300. https://doi.org/10.1016/j.jallcom.2016.03.118
Jung-König J, Feldmann C (2017) Microemulsion-made magnesium carbonate hollow nanospheres. Z Für Anorg Allg Chem 643:1491–1496. https://doi.org/10.1002/zaac.201700156
Kamran Haghighi H, Irannajad M, MohammadJafari A (2021) Thermodynamic and kinetic studies of heavy metal adsorption by modified nano-zeolite. Geosystem Eng 24:101–113. https://doi.org/10.1080/12269328.2021.1873197
Khairy M, El-Safty SA, MohamedA S, Elshehy EA (2013) Hierarchical inorganic–organic multi-shell nanospheres for intervention and treatment of lead-contaminated blood. Nanoscale 5:7920. https://doi.org/10.1039/c3nr02403b
Khalil U, Shakoor MB, Ali S et al (2021) Selective removal of hexavalent chromium from wastewater by rice husk: kinetic, isotherm and spectroscopic investigation. Water 13:263. https://doi.org/10.3390/w13030263
Koliehova A, Trokhymenko H, Melnychuk S, Gomelya M (2019) Treatment of wastewater containing a mixture of heavy metal ions (copper-zinc, copper-nickel) using ion-exchange methods. J Ecol Eng 20:146–151. https://doi.org/10.12911/22998993/112746
Liang S, Li Y, Qu G et al (2021) Preparation of recyclable materials for removing heavy metal ions in aqueous solution and wastewater applications. J Chem Technol Biotechnol 96:3330–3341. https://doi.org/10.1002/jctb.6885
Lu S, Cui W, Wang R et al (2022) Biomimetic mineralization and characterization of hierarchically structured hydrated magnesium carbonates: the effects of sodium alginate. J CO2 Util 56:101848. https://doi.org/10.1016/j.jcou.2021.101848
Luo G, Han Z, Xiong J et al (2021) Heavy metal pollution and ecological risk assessment of tailings in the Qinglong Dachang antimony mine, China. Environ Sci Pollut Res 28:33491–33504. https://doi.org/10.1007/s11356-021-12987-7
Marín-Alzate N, Tobón JI, Bertolotti B et al (2021) Evaluation of the properties of diatomaceous earth in relation to their performance in the removal of heavy metals from contaminated effluents. Water Air Soil Pollut 232:122. https://doi.org/10.1007/s11270-021-05045-y
Mnasri-Ghnimi S, Frini-Srasra N (2019) Removal of heavy metals from aqueous solutions by adsorption using single and mixed pillared clays. Appl Clay Sci 179:105151. https://doi.org/10.1016/j.clay.2019.105151
Mulungulungu GA, Mao T, Han K (2021) Efficient removal of high-concentration copper ions from wastewater via 2D g-C3N4 photocatalytic membrane filtration. Colloids Surf Physicochem Eng Asp 623:126714. https://doi.org/10.1016/j.colsurfa.2021.126714
Niu M, Li G, Cao L et al (2020a) Preparation of sulphate aluminate cement amended bentonite and its use in heavy metal adsorption. J Clean Prod 256:120700. https://doi.org/10.1016/j.jclepro.2020.120700
Niu Y, Jiang X, Wang K et al (2020b) Meta analysis of heavy metal pollution and sources in surface sediments of Lake Taihu, China. Sci Total Environ 700:134509. https://doi.org/10.1016/j.scitotenv.2019.134509
Panuccio MR, Sorgonà A, Rizzo M, Cacco G (2009) Cadmium adsorption on vermiculite, zeolite and pumice: batch experimental studies. J Environ Manage 90:364–374. https://doi.org/10.1016/j.jenvman.2007.10.005
Quyen V thi, Pham T-H, Kim J et al (2021) Biosorbent derived from coffee husk for efficient removal of toxic heavy metals from wastewater. Chemosphere 284:131312. https://doi.org/10.1016/j.chemosphere.2021.131312
Rahmani-Sani A, Singh P, Raizada P et al (2020) Use of chicken feather and eggshell to synthesize a novel magnetized activated carbon for sorption of heavy metal ions. Bioresour Technol 297:122452. https://doi.org/10.1016/j.biortech.2019.122452
Rheinheimer V, Unluer C, Liu J et al (2017) XPS Study on the stability and transformation of hydrate and carbonate phases within MgO systems. Materials 10:75. https://doi.org/10.3390/ma10010075
Rojas R (2014) Copper, lead and cadmium removal by Ca Al layered double hydroxides. Appl Clay Sci 87:254–259. https://doi.org/10.1016/j.clay.2013.11.015
Sarikaya M, Turan MD, Aydogmus R et al (2017) Extraction of Meso-pores Amorphous SiO2 from Van Pumice. Curr Phys Chem 7:301–304. https://doi.org/10.2174/1877946807666170808122455
Shahwan T, Zünbül B, Eroğlu AE, Yılmaz S (2005) Effect of magnesium carbonate on the uptake of aqueous zinc and lead ions by natural kaolinite and clinoptilolite. Appl Clay Sci 30:209–218. https://doi.org/10.1016/j.clay.2005.05.002
Shenashen MA, El-Safty SA, Elshehy EA (2014) Monolithic scaffolds for highly selective ion sensing/removal of Co( ii ), Cu( ii ), and Cd( ii ) ions in water. Analyst 139:6393–6405. https://doi.org/10.1039/C4AN00980K
Shrestha R, Ban S, Devkota S et al (2021) Technological trends in heavy metals removal from industrial wastewater: a review. J Environ Chem Eng 9:105688. https://doi.org/10.1016/j.jece.2021.105688
Shukla AK, Alam J, Alhoshan M et al (2018) Removal of heavy metal ions using a carboxylated graphene oxide-incorporated polyphenylsulfone nanofiltration membrane. Environ Sci Water Res Technol 4:438–448. https://doi.org/10.1039/C7EW00506G
Soliman A-AE, Shenashen MA, El-Sewify IM et al (2017) Mesoporous organic-inorganic core-shell necklace cages for potentially capturing Cd 2+ ions from water sources. ChemistrySelect 2:6135–6142. https://doi.org/10.1002/slct.201701247
Song H-J, Kim S, Cho Y (2020) Removal of heavy metals using sorbents derived from bark. J Porous Mater 27:319–328. https://doi.org/10.1007/s10934-019-00845-4
Sudagar AJ, Andrejkovičová S, Rocha F et al (2021) Combined influence of low-grade metakaolins and natural zeolite on compressive strength and heavy metal adsorption of geopolymers. Minerals 11:486. https://doi.org/10.3390/min11050486
Tural B, Ertaş E, Güzel M, Tural S (2021) Effect of structural differences of pumice on synthesis of pumice-supported nFe0: removal of Cr (VI) from water. Appl Water Sci 11:128. https://doi.org/10.1007/s13201-021-01458-6
Wahab N, Saeed M, Ibrahim M et al (2019) Synthesis, Characterization, and Applications of Silk/Bentonite Clay Composite for Heavy Metal Removal From Aqueous Solution. Front Chem 7:654. https://doi.org/10.3389/fchem.2019.00654
Wang F, Jin L, Guo C et al (2021) Enhanced heavy metals sorption by modified biochars derived from pig manure. Sci Total Environ 786:147595. https://doi.org/10.1016/j.scitotenv.2021.147595
Wang J, Guo X (2020) Adsorption kinetic models: physical meanings, applications, and solving methods. J Hazard Mater 390:122156. https://doi.org/10.1016/j.jhazmat.2020.122156
Wang K, Zhao J, Li H et al (2016a) Removal of cadmium (II) from aqueous solution by granular activated carbon supported magnesium hydroxide. J Taiwan Inst Chem Eng 61:287–291. https://doi.org/10.1016/j.jtice.2016.01.006
Wang S-Y, Li G-M, Xu W et al (2016b) Facile preparation and application of magnesium hydroxide assembly spheres. Res Chem Intermed 42:2661–2668. https://doi.org/10.1007/s11164-015-2173-4
Wu Y, Pang H, Liu Y et al (2019) Environmental remediation of heavy metal ions by novel-nanomaterials: a review. Environ Pollut 246:608–620. https://doi.org/10.1016/j.envpol.2018.12.076
Yang Y, Zhu H, Xu X et al (2021) Construction of a novel lanthanum carbonate-grafted ZSM-5 zeolite for effective highly selective phosphate removal from wastewater. Microporous Mesoporous Mater 324:111289. https://doi.org/10.1016/j.micromeso.2021.111289
Yavuz M, Gode F, Pehlivan E et al (2008) An economic removal of Cu2+ and Cr3+ on the new adsorbents: pumice and polyacrylonitrile/pumice composite. Chem Eng J 137:453–461. https://doi.org/10.1016/j.cej.2007.04.030
Yuan L, Shen J, Chen Z, Guan X (2016) Synergistic role of pumice surface composition in hydroxyl radical initiation in the catalytic ozonation process. Ozone Sci Eng 38:42–47. https://doi.org/10.1080/01919512.2015.1074535
Yurak V, Apakashev R, Dushin A et al (2021) Testing of natural sorbents for the assessment of heavy metal ions’ adsorption. Appl Sci 11:3723. https://doi.org/10.3390/app11083723
Zamora-Ledezma C, Negrete-Bolagay D, Figueroa F et al (2021) Heavy metal water pollution: a fresh look about hazards, novel and conventional remediation methods. Environ Technol Innov 22:101504. https://doi.org/10.1016/j.eti.2021.101504
Zha F, Chen S, Kang B et al (2022) Synergistic solidification of lead-contaminated soil by magnesium oxide and microorganisms. Chemosphere 308:136422. https://doi.org/10.1016/j.chemosphere.2022.136422
Zhang K, Wu S, Wang X et al (2015) Wide pH range for fluoride removal from water by MHS-MgO/MgCO3 adsorbent: kinetic, thermodynamic and mechanism studies. J Colloid Interface Sci 446:194–202. https://doi.org/10.1016/j.jcis.2015.01.049
Zhang R, Richardson JJ, Masters AF et al (2018a) Effective removal of toxic heavy metal ions from aqueous solution by CaCO3 microparticles. Water Air Soil Pollut 229:136. https://doi.org/10.1007/s11270-018-3787-0
Zhang Y, Xu G-S, Xu M-D et al (2021) Preparation of MgO porous nanoplates modified pumice and its adsorption performance on fluoride removal. J Alloys Compd 884:160953. https://doi.org/10.1016/j.jallcom.2021.160953
Zhang Z, Lu Y, Li H et al (2018b) Assessment of heavy metal contamination, distribution and source identification in the sediments from the Zijiang River, China. Sci Total Environ 645:235–243. https://doi.org/10.1016/j.scitotenv.2018.07.026
Zhou C, Song X, Wang Y et al (2022) The sorption and short-term immobilization of lead and cadmium by nano-hydroxyapatite/biochar in aqueous solution and soil. Chemosphere 286:131810. https://doi.org/10.1016/j.chemosphere.2021.131810
Zhou D, Wang H, Guo S (2021) Preparation of cellulose/chitin blend materials and influence of their properties on sorption of heavy metals. Sustainability 13:6460. https://doi.org/10.3390/su13116460
Zhu D, He Y, Zhang B et al (2021a) Simultaneous removal of multiple heavy metals from wastewater by novel plateau laterite ceramic in batch and fixed-bed studies. J Environ Chem Eng 9:105792. https://doi.org/10.1016/j.jece.2021.105792
Zhu H, Li L, Chen W et al (2021b) Controllable synthesis of coral-like hierarchical porous magnesium hydroxide with various surface area and pore volume for lead and cadmium ion adsorption. J Hazard Mater 416:125922. https://doi.org/10.1016/j.jhazmat.2021.125922
Funding
This work was supported in part by the project of National Key Research and development (No. 2019YFC0408504), Natural Science Foundation of Anhui Province (No. 2008085MB44), Natural Science Research Project of Anhui Educational Committee (No. 2022AH040046), and Quality engineering project of colleges and universities in Anhui Province (No. 2020kfkc263).
Author information
Authors and Affiliations
Contributions
Conceptualization: Yong Zhang and Zhen Jin; methodology: Yong Zhang, Yi-Fan Xiao, Guang-Song Xu, and Min-Da Xu; formal analysis and investigation: Yi-Fan Xiao, Guang-Song Xu, Min-Da Xu, De-Cai Wang, and Jia-Qi Liu; writing—original draft preparation: Yong Zhang, Yi-Fan Xiao, Zhen Jin, Guang-Song Xu, Min-Da Xu, and De-Cai Wang; writing—review and editing: Yi-Fan Xiao, Min-Da Xu, De-Cai Wang, and Zhen Jin; funding acquisition: Yong Zhang and Zhen Jin; resources: Min-Da Xu, De-Cai Wang, and Li–Li Yang; visualization: Yi-Fan Xiao, Jia-Qi Liu, and Li–Li Yang; validation: Jia-Qi Liu and Li–Li Yang; supervision: Yong Zhang and Zhen Jin.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Xiao, YF., Xu, GS. et al. Preparation of basic magnesium carbonate nanosheets modified pumice and its adsorption of heavy metals. Environ Sci Pollut Res 30, 111137–111151 (2023). https://doi.org/10.1007/s11356-023-30023-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30023-8