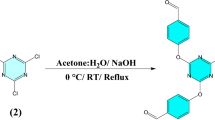
Abstract
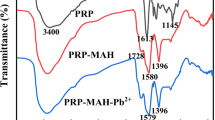
In this study, a novel N-rich triazine-based porous organic polymer (NR-POP) was synthesized via Schiff-base condensation. The structure of the synthesized porous polymer was identified using FT-IR, XRD, SEM, EDS, TEM, TGA, and BET analyses. The adsorption efficiency of this polymer was investigated for the removal of lead and cadmium ions pollutants. The adsorption processes of Pb(II) and Cd(II) metal ions by this polymer adsorbent were exothermic and matched by the Langmuir isotherm with a high correlation coefficient (R2 = 0.9904, 0.9778), the maximum adsorption capacity (833.33, 178.57 mg g−1), and the pseudo-second-order kinetic model. Furthermore, NR-POP showed an excellent adsorption selectivity for Pb(II) compared to Cd(II).
Graphical Abstract














Similar content being viewed by others
Data availability
Research data are available in article and its supplementary information.
References
Ahmad R, Mirza A (2018) Facile one pot green synthesis of Chitosan-Iron oxide (CS-Fe2O3) nanocomposite: Removal of Pb(II) and Cd(II) from synthetic and industrial wastewater. J Clean Prod 186:342–352. https://doi.org/10.1016/j.jclepro.2018.03.075
Ahmadijokani F, Tajahmadi S, Bahi A, Molavi H, Rezakazemi M, Ko F, Aminabhavi TM, Arjmand M (2021) Ethylenediamine-functionalized Zr-based MOF for efficient removal of heavy metal ions from water. Chemosphere 264:128466. https://doi.org/10.1016/j.chemosphere.2020.128466
Ain QU, Zhang H, Yaseen M, Rasheed U, Liu K, Subhan S, Tong Z (2020) Facile fabrication of hydroxyapatite-magnetite-bentonite composite for efficient adsorption of Pb(II), Cd(II), and crystal violet from aqueous solution. J Clean Prod 247:119088. https://doi.org/10.1016/j.jclepro.2019.119088
Awual MR, Islam A, Hasan MM, Rahman MM, Asiri AM, Khaleque MA, Chanmiya Sheikh M (2019) Introducing an alternate conjugated material for enhanced lead(II) capturing from wastewater. J Clean Prod 224:920–929. https://doi.org/10.1016/j.jclepro.2019.03.241
Azizian S (2004) Kinetic Models of Sorption: A Theoretical Analysis. J Colloid Interface Sci 276:47–52. https://doi.org/10.1016/j.jcis.2004.03.048
Bijad M, Hojjati-Najafabadi A, Asari-Bami H, Habibzadeh S, Amini I, Fazeli F (2021) An overview of modified sensors with focus on electrochemical sensing of sulfite in food samples. Eurasian Chem Commun 3:116–138. https://doi.org/10.22034/ecc.2021.268819.1122
Cao H, Yang P, Ye T, Yuan M, Yu J, Wu X, Yin F, Li Y, Xu F (2021) Recognizing adsorption of Cd(II) by a novel core-shell mesoporous ion-imprinted polymer: characterization, binding mechanism and practical application. Chemosphere 278:130369. https://doi.org/10.1016/j.chemosphere.2021.130369
Chen G, Shah KJ, Shi L, Chiang P-C (2017) Removal of Cd(II) and Pb(II) ions from aqueous solutions by synthetic mineral adsorbent: Performance and mechanisms. Appl Surf Sci 409:296–305. https://doi.org/10.1016/j.apsusc.2017.03.022
Cheng N, Wang B, Wu P, Lee X, Xing Y, Chen M, Gao B (2021) Adsorption of emerging contaminants from water and wastewater by modified biochar: a review. Environ Pollut 273:116448. https://doi.org/10.1016/j.envpol.2021.116448
Claverie M, Garcia J, Prevost T, Brendlé J, Limousy L (2019) Inorganic and hybrid (organic–inorganic) lamellar materials for heavy metals and radionuclides capture in energy wastes management—a review. Materials 12:1399. https://doi.org/10.3390/ma12091399
Deng J-H, Zhang X-R, Zeng G-M, Gong J-L, Niu Q-Y, Liang J (2013) Simultaneous removal of Cd(II) and ionic dyes from aqueous solution using magnetic graphene oxide nanocomposite as an adsorbent. Chem Eng J 226:189–200. https://doi.org/10.1016/j.cej.2013.04.045
Dou H, Xu M, Wang B, Zhang Z, Wen G, Zheng Y, Luo D, Zhao L, Yu A, Zhang L, Jiang Z, Chen Z (2020) Microporous framework membranes for precise molecule/ion separations.https://doi.org/10.1039/D0CS00552E
Ebrahimpour E, Kazemi A (2023) Mercury(II) and lead(II) ions removal using a novel thiol-rich hydrogel adsorbent; PHPAm/Fe3O4@SiO2-SH polymer nanocomposite. Environ Sci Pollut Res 30:13605–13623. https://doi.org/10.1007/s11356-022-23055-z
Fang Q, Zhuang Z, Gu S, Kaspar RB, Zheng J, Wang J, Qiu S, Yan Y (2014) Designed synthesis of large-pore crystalline polyimide covalent organic frameworks. Nat Commun 5:4503. https://doi.org/10.1038/ncomms5503
Gallareta-Olivares G, Rivas-Sanchez A, Cruz-Cruz A, Hussain SM, González-González RB, Cárdenas-Alcaide MF, Iqbal HMN, Parra-Saldívar R (2023) Metal-doped carbon dots as robust nanomaterials for the monitoring and degradation of water pollutants. Chemosphere 312:137190. https://doi.org/10.1016/j.chemosphere.2022.137190
Guo W, Xia T, Pei M, Du Y, Wang L (2019) Bentonite Modified by Allylamine Polymer for Adsorption of Amido Black 10B. Polymers 11:502. https://doi.org/10.3390/polym11030502
Hassan M, Deb AK, Qi F, Liu Y, Du J, Fahy A, Ahsan MA, Parikh SJ, Naidu R (2021) Magnetically separable mesoporous alginate polymer beads assist adequate removal of aqueous methylene blue over broad solution pH. J Clean Prod 319:128694. https://doi.org/10.1016/j.jclepro.2021.128694
He J, Li Y, Wang C, Zhang K, Lin D, Kong L, Liu J (2017) Rapid adsorption of Pb, Cu and Cd from aqueous solutions by β-cyclodextrin polymers. Appl Surf Sci 426:29–39. https://doi.org/10.1016/j.apsusc.2017.07.103
Ho YS, Mckay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Hojjati-Najafabadi A, Mansoorianfar M, Liang T, Shahin K, Karimi-Maleh H (2022a) A review on magnetic sensors for monitoring of hazardous pollutants in water resources. Sci Total Environ 824:153844. https://doi.org/10.1016/j.scitotenv.2022.153844
Hojjati-Najafabadi A, Mansoorianfar M, Liang T, Shahin K, Wen Y, Bahrami A, Karaman C, Zare N, Karimi-Maleh H, Vasseghian Y (2022b) Magnetic-MXene-based nanocomposites for water and wastewater treatment: a review. J Water Process Eng 47:102696. https://doi.org/10.1016/j.jwpe.2022.102696
Huang N, Zhai L, Xu H, Jiang D (2017) Stable covalent organic frameworks for exceptional mercury removal from aqueous solutions. J Am Chem Soc 139:2428–2434. https://doi.org/10.1021/jacs.6b12328
Hussain MS, Musharraf SG, Bhanger MI, Malik MI (2020) Salicylaldehyde derivative of nano-chitosan as an efficient adsorbent for lead(II), copper(II), and cadmium(II) ions. Int J Biol Macromol 147:643–652. https://doi.org/10.1016/j.ijbiomac.2020.01.091
Huy DH, Seelen E, Liem-Nguyen V (2020) Removal mechanisms of cadmium and lead ions in contaminated water by stainless steel slag obtained from scrap metal recycling. J Water Process Eng 36:101369. https://doi.org/10.1016/j.jwpe.2020.101369
Issakhov A, Alimbek A, Abylkassymova A (2023) Numerical modeling of water pollution by products of chemical reactions from the activities of industrial facilities at variable and constant temperatures of the environment. J Contam Hydrol 252:104116. https://doi.org/10.1016/j.jconhyd.2022.104116
Jiao S, Deng L, Zhang X, Zhang Y, Liu K, Li S, Wang L, Ma D (2021) Evaluation of an Ionic Porous Organic Polymer for Water Remediation. ACS Appl Mater Interfaces 13:39404–39413. https://doi.org/10.1021/acsami.1c10464
Jing W, Zhang F, Chen H (2022) Comparative tribological performance and erosion resistance of epoxy resin composite coatings reinforced with aramid fiber and carbon fiber. Colloids Surf A 648:129354. https://doi.org/10.1016/j.colsurfa.2022.129354
Karrouchi K, Brandán SA, Sert Y, El-marzouqi H, Radi S, Ferbinteanu M, Faouzi MEA, Garcia Y, Ansar Mh (2020) Synthesis, X-ray structure, vibrational spectroscopy, DFT, biological evaluation and molecular docking studies of (E)-N’-(4-(dimethylamino)benzylidene)-5-methyl-1H-pyrazole-3-carbohydrazide. J Mol Struct 1219:128541. https://doi.org/10.1016/j.molstruc.2020.128541
Khan FSA, Mubarak NM, Khalid M, Walvekar R, Abdullah EC, Mazari SA, Nizamuddin S, Karri RR (2020) Magnetic nanoadsorbentsʼ potential route for heavy metals removal-a review. Environ Sci Pollut Res Int 27:24342–24356. https://doi.org/10.1007/s11356-020-08711-6
Kumar PS, Gayathri R, Rathi BS (2021) A review on adsorptive separation of toxic metals from aquatic system using biochar produced from agro-waste. Chemosphere 285:131438. https://doi.org/10.1016/j.chemosphere.2021.131438
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403. https://doi.org/10.1021/ja02242a004
Lee J-SM, Cooper AI (2020) Advances in conjugated microporous polymers. Chem Rev 120:2171–2214. https://doi.org/10.1021/acs.chemrev.9b00399
Li X, Wang S, Liu Y, Jiang L, Song B, Li M, Zeng G, Tan X, Cai X, Ding Y (2017) Adsorption of Cu(II), Pb(II), and Cd(II) ions from acidic aqueous solutions by diethylenetriaminepentaacetic acid-modified magnetic graphene oxide. J Chem Eng Data 62:407–416. https://doi.org/10.1021/acs.jced.6b00746
Li J, Tian T, Jia Y, Xu N, Yang S, Zhang C, Gao S, Shen W, Wang Z (2023) Adsorption performance and optimization by response surface methodology on tetracycline using Fe-doped ZIF-8-loaded multi-walled carbon nanotubes. Environ Sci Pollut Res 30:4123–4136. https://doi.org/10.1007/s11356-022-22524-9
Liu X, Xu Q, Li Z, Pan B, Ndagijimana P, Wang Y (2022) Simultaneous removal of cationic heavy metals and arsenic from drinking water by an activated carbon supported nanoscale zero-valent iron and nanosilver composite. Colloids Surf A 650:129581. https://doi.org/10.1016/j.colsurfa.2022.129581
Lu Q, Ma Y, Li H, Guan X, Yusran Y, Xue M, Fang Q, Yan Y, Qiu S, Valtchev V (2018) Postsynthetic functionalization of three-dimensional covalent organic frameworks for selective extraction of lanthanide ions. Angew Chem Int Ed Engl 57:6042–6048. https://doi.org/10.1002/anie.201712246
Metin AÜ, Doğan D, Can M (2020) Novel magnetic gel beads based on ionically crosslinked sodium alginate and polyanetholesulfonic acid: Synthesis and application for adsorption of cationic dyes. Mater Chem Phys 256:123659. https://doi.org/10.1016/j.matchemphys.2020.123659
Mohamed MG, El-Mahdy AFM, Kotp MG, Kuo S-W (2022) Advances in porous organic polymers: syntheses, structures, and diverse applications. Mater Adv 3:707–733. https://doi.org/10.1039/D1MA00771H
Pawar RR, Lalhmunsiama, Ingole PG, Lee S-M (2020) Use of activated bentonite-alginate composite beads for efficient removal of toxic Cu2+ and Pb2+ ions from aquatic environment. Int J Biol Macromol 164:3145–3154. https://doi.org/10.1016/j.ijbiomac.2020.08.130
Quesada HB, de Araújo TP, Vareschini DT, de Barros M, Gomes RG, Bergamasco R (2020) Chitosan, alginate and other macromolecules as activated carbon immobilizing agents: A review on composite adsorbents for the removal of water contaminants. Int J Biol Macromol 164:2535–2549. https://doi.org/10.1016/j.ijbiomac.2020.08.118
Ravi S, Kim S-Y, Bae Y-S (2022) Novel benzylphosphate-based covalent porous organic polymers for the effective capture of rare earth elements from aqueous solutions. J Hazard Mater 424:127356. https://doi.org/10.1016/j.jhazmat.2021.127356
Sagaama A, Issaoui N, Bardak F, Al-Dossary O, Kazachenko AS, Karrouchi K, Atac A, Wojcik MJ (2022) Non covalent interactions analysis and spectroscopic characterization combined with molecular docking study of N′-(4-Methoxybenzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide. J King Saud Univ Sci 34:101778. https://doi.org/10.1016/j.jksus.2021.101778
Sen A, Dutta S, Dam GK, Samanta P, Let S, Sharma S, Shirolkar MM, Ghosh SK (2021) Imidazolium-functionalized chemically robust ionic porous organic polymers (iPOPs) toward toxic oxo-pollutants capture from water. Chemistry 27:13442–13449. https://doi.org/10.1002/chem.202102399
Shahraki S, Delarami HS, Khosravi F (2019) Synthesis and characterization of an adsorptive Schiff base-chitosan nanocomposite for removal of Pb(II) ion from aqueous media. Int J Biol Macromol 139:577–586. https://doi.org/10.1016/j.ijbiomac.2019.07.223
Skorjanc T, Shetty D, Trabolsi A (2021) Pollutant removal with organic macrocycle-based covalent organic polymers and frameworks. Chem 7:882–918. https://doi.org/10.1016/j.chempr.2021.01.002
Subodh, Prakash K, Masram DT (2021) Silver nanoparticles immobilized covalent organic microspheres for hydrogenation of nitroaromatics with intriguing catalytic activity. ACS Appl Polym Mater 3:310–318. https://doi.org/10.1021/acsapm.0c01111
Sun Q, Dai Z, Meng X, Xiao F-S (2015) Porous polymer catalysts with hierarchical structures. Chem Soc Rev 44:6018–6034. https://doi.org/10.1039/C5CS00198F
Teng W, Bai N, Chen Z, Shi J, Fan J, Zhang W-x (2018) Hierarchically porous carbon derived from metal-organic frameworks for separation of aromatic pollutants. Chem Eng J 346:388–396. https://doi.org/10.1016/j.cej.2018.04.051
Thaçi BS, Gashi ST (2019) Reverse osmosis removal of heavy metals from wastewater effluents using biowaste materials pretreatment. Pol J Environ Stud 28:337–341. https://doi.org/10.15244/pjoes/81268
Tran HN, You SJ, Hosseini-Bandegharaei A, Chao HP (2017) Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: a critical review. Water Res 120:88–116. https://doi.org/10.1016/j.watres.2017.04.014
Tran CV, Quang DV, Nguyen Thi HP, Truong TN, La DD (2020) Effective Removal of Pb(II) from Aqueous Media by a New Design of Cu-Mg Binary Ferrite. ACS Omega 5:7298–7306. https://doi.org/10.1021/acsomega.9b04126
Upadhyay U, Sreedhar I, Singh SA, Patel CM, Anitha KL (2021) Recent advances in heavy metal removal by chitosan based adsorbents. Carbohydr Polym 251:117000. https://doi.org/10.1016/j.carbpol.2020.117000
Valenzuela F, Quintana G, Briso A, Ide V, Basualto C, Gaete J, Montes G (2021) Cu(II), Cd(II), Pb(II) and As(V) adsorption from aqueous solutions using magnetic iron-modified calcium silicate hydrate: Adsorption kinetic analysis. J Water Process Eng 40:101951. https://doi.org/10.1016/j.jwpe.2021.101951
Vo TS, Hossain MM, Jeong HM, Kim K (2020) Heavy metal removal applications using adsorptive membranes. Nano Convergence 7:36. https://doi.org/10.1186/s40580-020-00245-4
Wang T-X, Liang H-P, Anito DA, Ding X, Han B-H (2020) Emerging applications of porous organic polymers in visible-light photocatalysis. J Mater Chem A 8:7003–7034. https://doi.org/10.1039/D0TA00364F
Wang J, Sharaf F, Kanwal A (2023a) Nitrate pollution and its solutions with special emphasis on electrochemical reduction removal. Environ Sci Pollut Res 30:9290–9310. https://doi.org/10.1007/s11356-022-24450-2
Wang S, Wang H, Wang S, Fu L, Zhang L (2023b) Novel magnetic covalent organic framework for the selective and effective removal of hazardous metal Pb(II) from solution: synthesis and adsorption characteristics. Sep Purif Technol 307:122783. https://doi.org/10.1016/j.seppur.2022.122783
Wei W, Han X, Zhang M, Zhang Y, Zhang Y, Zheng C (2020) Macromolecular humic acid modified nano-hydroxyapatite for simultaneous removal of Cu(II) and methylene blue from aqueous solution: experimental design and adsorption study. Int J Biol Macromol 150:849–860. https://doi.org/10.1016/j.ijbiomac.2020.02.137
Wu R, Zhao K, Lv W, Xu J, Hu J, Liu H, Wang H (2021) Solid-phase synthesis of bi-functionalized porous organic polymer for simultaneous removal of Hg(II) and Pb(II). Microporous Mesoporous Mater 316:110942. https://doi.org/10.1016/j.micromeso.2021.110942
Xiao X, Yu Y, Sun Y, Zheng X, Chen A (2021) Heavy metal removal from aqueous solutions by chitosan-based magnetic composite flocculants. J Environ Sci (china) 108:22–32. https://doi.org/10.1016/j.jes.2021.02.014
Xiong T, Yuan X, Cao X, Wang H, Jiang L, Wu Z, Liu Y (2020) Mechanistic insights into heavy metals affinity in magnetic MnO2@Fe3O4/poly(m-phenylenediamine) core−shell adsorbent. Ecotoxicol Environ Saf 192:110326. https://doi.org/10.1016/j.ecoenv.2020.110326
Yadav S, Asthana A, Chakraborty R, Jain B, Singh AK, Carabineiro SAC, Susan M (2020) Cationic dye removal using novel magnetic/activated charcoal/β-cyclodextrin/alginate polymer nanocomposite. Nanomaterials (Basel) 10. https://doi.org/10.3390/nano10010170
Yang Z, Yang T, Yang Y, Yi X, Hao X, Xie T, Liao CJ (2021) The behavior and mechanism of the adsorption of Pb(II) and Cd(II) by a porous double network porous hydrogel derived from peanut shells. Mater Today Commun 27:102449. https://doi.org/10.1016/j.mtcomm.2021.102449
Yuan Y, Zhu G (2019) Porous aromatic frameworks as a platform for multifunctional applications. ACS Cent Sci 5:409–418. https://doi.org/10.1021/acscentsci.9b00047
Yuvaraja G, Pang Y, Chen D-Y, Kong L-J, Mehmood S, Subbaiah MV, Rao DS, Mouli Pavuluri C, Wen J-C, Reddy GM (2019) Modification of chitosan macromolecule and its mechanism for the removal of Pb(II) ions from aqueous environment. Int J Biol Macromol 136:177–188. https://doi.org/10.1016/j.ijbiomac.2019.06.016
Zhang T, Xing G, Chen W, Chen L (2020) Porous organic polymers: a promising platform for efficient photocatalysis. Mater Chem Front 4:332–353. https://doi.org/10.1039/C9QM00633H
Zhang H, Lin H, Li Q, Cheng C, Shen H, Zhang Z, Zhang Z, Wang H (2021) Removal of refractory organics in wastewater by coagulation/flocculation with green chlorine-free coagulants. Sci Total Environ 787:147654. https://doi.org/10.1016/j.scitotenv.2021.147654
Zhang X, Song Z, Dou Y, Xue Y, Ji Y, Tang Y, Hu M (2021) Removal difference of Cr(VI) by modified zeolites coated with MgAl and ZnAl-layered double hydroxides: efficiency, factors and mechanism. Colloids Surf A 621:126583. https://doi.org/10.1016/j.colsurfa.2021.126583
Zhang X, Zheng J, Jin P, Xu D, Yuan S, Zhao R, Depuydt S, Gao Y, Xu Z-L, Van der Bruggen B (2022) A PEI/TMC membrane modified with an ionic liquid with enhanced permeability and antibacterial properties for the removal of heavy metal ions. J Hazard Mater 435:129010. https://doi.org/10.1016/j.jhazmat.2022.129010
Zhang Y, Wang X, Li Y, Wang H, Shi Y, Li Y, Zhang Y (2022) Improving nanoplastic removal by coagulation: impact mechanism of particle size and water chemical conditions. J Hazard Mater 425:127962. https://doi.org/10.1016/j.jhazmat.2021.127962
Zhao J, Yan G, Hu Z, Zhang X, Shi J, Jiang X (2021) Triazine-based porous organic polymers with enhanced electronegativity as multifunctional separator coatings in lithium–sulfur batteries. Nanoscale 13:12028–12037. https://doi.org/10.1039/D1NR02980K
Zhou T, Xia F, Deng Y, Zhao Y (2018) Removal of Pb(II) from aqueous solutions using waste textiles/poly(acrylic acid) composite synthesized by radical polymerization technique. J Environ Sci 67:368–377. https://doi.org/10.1016/j.jes.2017.04.010
Acknowledgements
We gratefully acknowledge the financial support of this work by the research council of Arak University.
Author information
Authors and Affiliations
Contributions
Design of proposal was presented by Akbar Mobinikhaledi and Javad Ghanbari. The experimental section was done by Javad Ghanbari. The interpretation of data, preparing of the manuscript and its editing were performed by Akbar Mobinikhaledi and Javad Ghanbari.
Corresponding author
Ethics declarations
Ethical approval
Authors have completed the manuscript with ethical standard.
Consent to participate
Not applicable.
Consent to publish
Authors are satisfied to publish their research work in Environmental Science and Pollution Research.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghanbari, J., Mobinikhaledi, A. Synthesis and characterization of a novel N-rich porous organic polymer and its application as an efficient porous adsorbent for the removal of Pb(II) and Cd(II) ions from aqueous solutions. Environ Sci Pollut Res 30, 68919–68933 (2023). https://doi.org/10.1007/s11356-023-27274-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-27274-w