Abstract
Electroplating manufacturing processes release industrial effluents that comprise severe levels of heavy metals into the environment. This study investigated the utilization of nanoparticles of zero-valent iron (nZVI) for the treatment of electroplating wastewater industry containing multiple heavy metal ions. In batch experiments using Cu2+ as a single solute, the optimum operating condition was pH 7.3, nZVI dosage 1.6 g/L, time 36 min, temperature 30 °C, and agitation speed 180 rpm, achieving almost 100% Cu2+ removal efficiency. The adsorption mechanisms were illustrated using scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDX), and Fourier transform infrared (FTIR) spectrometer. Moreover, continuous-feed experiments were performed to treat real electroplating wastewater industry via adsorption and sedimentation processes. The system attained removal efficiencies of 91.3% total suspended solids (TSS), 68.3% chemical oxygen demand (COD), 94.2% nitrogen (N), 98.5% phosphorus (P), 66.7% Cr6+, 91.5% Pb2+, 83.3% Ag+, 80.8% Cu2+, 17.4% Ni2+, 47.1% Mn2+, 54.6% Zn2+, 94.7% Fe3+, 100.0% Al3+, and 42.1% Co2+. The removal mechanisms included reduction of Men+ to Me(n-x)+/Me0 by the Fe0 core, adsorption to the oxide shell as Me(OH)x and Me-Fe-OOH, oxidation of Men+ to Me(n+z)+, specific surface bonding, and sequential steps of electron transfer and precipitation. The total cost, including amortized and operating expenses for scaling up the adsorption system, was 4.45$ per m3 of electroplating wastewater.

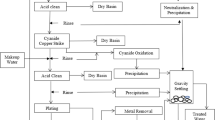
Graphical abstract




Similar content being viewed by others
References
Anastopoulos I, Mittal A, Usman M, Mittal J, Yu G, Núñez-Delgado A, Kornaros M (2018) A review on halloysite-based adsorbents to remove pollutants in water and wastewater. J Mol Liq 269:855–868. https://doi.org/10.1016/j.molliq.2018.08.104
APHA (1998) APHA, AWWA, WPCF: Standard Methods for the Examination of Water and Wastewater, 20th edn. American Public Health Association, Washington, DC
Barreto-Rodrigues M, Silva F, Paiva T (2009) Optimization of Brazilian TNT industry wastewater treatment using combined zero-valent iron and Fenton processes. J Hazard Mater 168:1065–1069. https://doi.org/10.1016/j.jhazmat.2009.02.172
Christoforidis AK, Orfanidis S, Papageorgiou SK, Lazaridou AN, Favvas EP, Mitropoulos AC (2015) Study of Cu(II) removal by Cystoseira crinitophylla biomass in batch and continuous flow biosorption. Chem Eng J 277:334–340. https://doi.org/10.1016/j.cej.2015.04.138
Fan M, Hu J, Cao R, Xiong K, Wei X (2017) Modeling and prediction of copper removal from aqueous solutions by nZVI/rGO magnetic nanocomposites using ANN-GA and ANN-PSO. Sci Rep 7:18040. https://doi.org/10.1038/s41598-017-18223-y
Fawzy M, Nasr M, Adel S, Helmi S (2018) Regression model, artificial neural network, and cost estimation for biosorption of Ni (II)-ions from aqueous solutions by Potamogeton pectinatus. Int J Phytoremediation 20:321–329. https://doi.org/10.1080/15226514.2017.1381941
Fawzy M, Nasr M, Abdel-Rahman AM, Hosny G, Raheem Odhafa B (2019) Techno-economic and environmental approaches of Cd2+ adsorption by olive leaves (Olea europaea L.) waste. Int J Phytoremediation:1–10. https://doi.org/10.1080/15226514.2019.1612848
Freundlich H (1906) Over the adsorption in solution. J Phys Chem 57:385–470
Fu F, Dionysiou D, Liu H (2014) The use of zero-valent iron for groundwater remediation and wastewater treatment: a review. J Hazard Mater 267:194–205. https://doi.org/10.1016/j.jhazmat.2013.12.062
Gar Alalm M, Nasr M (2018) Artificial intelligence, regression model, and cost estimation for removal of chlorothalonil pesticide by activated carbon prepared from casuarina charcoal. Sustain Environ Res 28:101–110. https://doi.org/10.1016/j.serj.2018.01.003
Greenland S, Senn S, Rothman K, Carlin J, Poole C, Goodman S, Altman D (2016) Statistical tests, P values, confidence intervals, and power: a guide to misinterpretations. Eur J Epidemiol 31:337–350. https://doi.org/10.1007/s10654-016-0149-3
Gupta SK, Nayunigari MK, Misra R, Ansari FA, Dionysiou DD, Maity A, Bux F (2016) Synthesis and performance evaluation of a new polymeric composite for the treatment of textile wastewater. Ind Eng Chem Res 55:13–20. https://doi.org/10.1021/acs.iecr.5b03714
Hamdy A, Mostafa M, Nasr M (2018a) Regression analysis and artificial intelligence for removal of methylene blue from aqueous solutions using nanoscale zero-valent iron. Int J Environ Sci Technol 16:357–372. https://doi.org/10.1007/s13762-018-1677-z
Hamdy A, Mostafa M, Nasr M (2018b) Zero-valent iron nanoparticles for methylene blue removal from aqueous solutions and textile wastewater treatment, with cost estimation. Water Sci Technol 78:367–378. https://doi.org/10.2166/wst.2018.306
Ho Y, Mckay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Javanbakht V, Alavi S, Zilouei H (2014) Mechanisms of heavy metal removal using microorganisms as biosorbent. Water Sci Technol 69:1775–1787. https://doi.org/10.2166/wst.2013.718
Karabelli D, Üzüm Ç, Shahwan T, Eroğlu AE, Scott TB, Hallam KR, Lieberwirth I (2008) Batch removal of aqueous Cu2+ ions using nanoparticles of zero-valent iron: a study of the capacity and mechanism of uptake. Ind Eng Chem Res 47:4758–4764. https://doi.org/10.1021/ie800081s
Krstić V, Urošević T, Pešovski B (2018) A review on adsorbents for treatment of water and wastewaters containing copper ions. Chem Eng Sci 192:273–287. https://doi.org/10.1016/j.ces.2018.07.022
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403. https://doi.org/10.1021/ja02242a004
Largitte L, Pasquier R (2016) A review of the kinetics adsorption models and their application to the adsorption of lead by an activated carbon. Chem Eng Res Des 109:495–504. https://doi.org/10.1016/j.cherd.2016.02.006
Li S, Wang W, Liang F, Zhang W (2017) Heavy metal removal using nanoscale zero-valent iron (nZVI): theory and application. J Hazard Mater 322:163–171. https://doi.org/10.1016/j.jhazmat.2016.01.032
Mahmoud A, Mostafa M, Nasr M (2018) Regression model, artificial intelligence, and cost estimation for phosphate adsorption using encapsulated nanoscale zero-valent iron. Sep Sci Technol 54:13–26. https://doi.org/10.1080/01496395.2018.1504799
Majlesi M, Hashempour Y (2017) Removal of 4-chlorophenol from aqueous solution by granular activated carbon/nanoscale zero valent iron based on response surface modeling. Arch Environ Prot 43:13–25. https://doi.org/10.1515/aep-2017-0035
Park S, Cho H, Ryu C, Park Y-K (2016) Removal of copper(II) in aqueous solution using pyrolytic biochars derived from red macroalga Porphyra tenera. J Ind Eng Chem 36:314–319. https://doi.org/10.1016/j.jiec.2016.02.021
Park J-A, Kang J-K, Lee S-C, Kim S-B (2017) Electrospun poly (acrylic acid)/poly (vinyl alcohol) nanofibrous adsorbents for cu (II) removal from industrial plating wastewater. RSC Adv 7:18075–18084. https://doi.org/10.1039/C7RA01362K
Peng W (2015) Amino-functionalized adsorbent prepared by means of Cu(II) imprinted method and its selective removal of copper from aqueous solutions. J Hazard Mater 294:9–16. https://doi.org/10.1016/j.jhazmat.2015.03.046
Prema P (2011) Color removal efficiency of dyes using nanozerovalent iron treatment. Toxicol Environ Chem 93:1908–1917. https://doi.org/10.1080/02772248.2011.606613
Santos-Juanes Jordá L, Ballesteros Martín MM, Ortega Gómez E, Cabrera Reina A, Román Sánchez IM, Casas López JL, Sánchez Pérez JA (2011) Economic evaluation of the photo-Fenton process. Mineralization level and reaction time: the keys for increasing plant efficiency. J Hazard Mater 186:1924–1929. https://doi.org/10.1016/j.jhazmat.2010.12.100
Shrivastava A, Gupta VB (2011) Methods for the determination of limit of detection and limit of quantitation of the analytical methods. Chron Young Sci 2:21–25 http://www.cysonline.org/text.asp?2011/2/1/21/79345
Tan K, Hameed B (2017) Insight into the adsorption kinetics models for the removal of contaminants from aqueous solutions. J Taiwan Inst Chem E 74:25–48. https://doi.org/10.1016/j.jtice.2017.01.024
USEPA (2009) In: May (ed) United State EPA 816-F-09-004. United States Environmental Protection Agency (USEPA), Washington, D.C. https://www.epa.gov/dwstandardsregulations. Accessed Mar 2019
Weber W, Morris J (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div 89:31–60 https://cedb.asce.org/CEDBsearch/record.jsp?dockey=0013042
World Health Organization (2011) Guidelines for Drinking-water Quality (forth ed.), WHO, Switzerland
Yang L (2016) In situ NH2-functionalized graphene oxide/SiO2 composites to improve Cu(II) removal from ammoniacal solutions. Chem Eng J 306:77–85. https://doi.org/10.1016/j.cej.2016.07.035
Zhang Y (2015) Structural evolution of nanoscale zero-valent iron (nZVI) in anoxic Co2+ solution: interactional performance and mechanism. Sci Rep 5:13966. https://doi.org/10.1038/srep13966
Zou Y (2016) Environmental remediation and application of nanoscale zero-valent iron and its composites for the removal of heavy metal ions: a review. Environ Sci Technol 50:7290–7304. https://doi.org/10.1021/acs.est.6b01897
Zou Z, Shi Z, Deng L (2017) Highly efficient removal of Cu(II) from aqueous solution using a novel magnetic EDTA functionalized CoFe2O4. RSC Adv 7:5195–5205 https://pubs.rsc.org/en/content/articlelanding/2017/ra/c6ra26821h#!divAbstract. Accessed January 2019
Acknowledgments
This research was supported by the Egyptian Housing and Building National Research Center (HBRC), Environmental Engineering Program, Zewail City of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
Zero-valent Fe nanoparticles were synthesized to treat electroplating wastewater
Total Cu2+ adsorption at pH 7.3, nZVI dosage 1.6 g/L, mixing 180 rpm, 36 min, and 30 °C
Real electroplating wastewater was continuously fed to adsorption/sedimentation units
Pollutants removal mechanisms were reduction, oxidation, complexation, and bonding
Total cost for scaling up adsorption system = 4.45$/m3 of electroplating wastewater
Electronic supplementary material
ESM 1
(DOCX 3328 kb)
Rights and permissions
About this article
Cite this article
Hamdy, A., Mostafa, M.K. & Nasr, M. Techno-economic estimation of electroplating wastewater treatment using zero-valent iron nanoparticles: batch optimization, continuous feed, and scaling up studies. Environ Sci Pollut Res 26, 25372–25385 (2019). https://doi.org/10.1007/s11356-019-05850-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-05850-3