Abstract
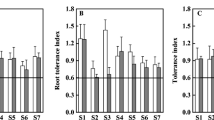
The potential use of a metal-tolerant sunflower mutant line for biomonitoring Cu phytoavailability, Cu-induced soil phytotoxicity, and Cu phytoextraction was assessed on a Cu-contaminated soil series (13–1020 mg Cu kg−1) obtained by fading a sandy topsoil from a wood preservation site with a similar uncontaminated soil. Morphological and functional plant responses as well as shoot, leaf, and root ionomes were measured after a 1-month pot experiment. Hypocotyl length, shoot and root dry weight (DW) yields, and leaf area gradually decreased as soil Cu exposure rose. Their dose-response curves (DRC) plotted against indicators of Cu exposure were generally well fitted by sigmoidal curves. The half-maximal effective concentration (EC50) of morphological parameters ranged between 203 and 333 mg Cu kg−1 soil, corresponding to 290–430 μg Cu L−1 in the soil pore water, and 20 ± 5 mg Cu kg−1 DW in the shoots. The EC10 for shoot Cu concentration (13–15 mg Cu kg−1 DW) coincided to 166 mg Cu kg−1 soil. Total chlorophyll content and total antioxidant capacity (TAC) were early biomarkers (EC10: 23 and 51 mg Cu kg−1 soil). Their DRC displayed a biphasic response. Photosynthetic pigment contents, e.g., carotenoids, correlated with TAC. Ionome was changed in Cu-stressed roots, shoots, and leaves. Shoot Cu removal peaked roughly at 280 μg Cu L−1 in the soil pore water.


Similar content being viewed by others
Abbreviations
- ABTS:
-
2,2′-Azinobis (3-ethylbenzothiazoline 6-sulfonate)
- AOA:
-
Antioxidant activity
- APX:
-
Ascorbate peroxidase
- AsA:
-
Ascorbate
- Carot:
-
Carotenoid content
- CEC:
-
Cation exchange capacity
- Chl a :
-
Chlorophyll a content
- Chl b :
-
Chlorophyll b content
- ChlTOT:
-
Total chlorophyll content
- CuSH:
-
Shoot Cu concentration
- CuSPW:
-
Total Cu concentration in the soil pore water
- CuTOT:
-
Total soil Cu
- DHAR:
-
Dehydroascorbate reductase
- DMA:
-
Dimethylamine
- DMF:
-
N,N-dimethylformamide
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl
- DRC:
-
Dose-response curve
- DW SH:
-
Shoot dry weight yield
- DW RT:
-
Root dry weight yield
- EC:
-
Effective concentration
- EL:
-
Epicotyl length
- FW:
-
Fresh weight
- FRAP:
-
Ferric-reducing antioxidant potential
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- GSSG:
-
Oxidized glutathione
- HL:
-
Hypocotyl length
- IRT:
-
Iron-regulated transporter
- MDHAR:
-
Monodehydroascorbate reductase
- NA:
-
Nicotianamine
- OM:
-
Organic matter
- ORAC:
-
Oxygen radical absorption capacity
- ROS:
-
Reactive oxygen species
- SL:
-
Stem length
- SOD:
-
Superoxide dismutase
- S/R:
-
Shoot dry weight yield: root dry weight yield ratio
- TAC:
-
Total antioxidant capacity
- TE:
-
Trace element
- TEAC:
-
Trolox equivalent antioxidant capacity
- TF:
-
Translocation factor
- TLA:
-
Total leaf area
- ZIP:
-
Zinc- and iron-regulated transporter
References
Ali NA, Bernal MP, Ater M (2002) Tolerance and bioaccumulation of copper in Phragmites australis and Zea mays. Plant Soil 239:103–111. https://doi.org/10.1023/A:1014995321560
Batty C, Baker AJM, Wheeler BD, Curtis CD (2000) The effect of pH and plaque on the uptake of Cu and Mn in Phragmites australis (Cav.) Trin ex. Steudel. Ann Bot 86:647–653. https://doi.org/10.1006/anbo.2000.1191
Bes C, Mench M, Aulen M, Gasté H, Taberly J (2010) Spatial variation of plant communities and shoot Cu concentrations of plant species at a timber treatment site. Plant Soil 330:267–280. https://doi.org/10.1007/s11104-009-0198-4
Boojar MMA, Goodarzi F (2007) The copper tolerance strategies and the role of antioxidative enzymes in three plant species grown on copper mine. Chemosphere 67:2138–2147. https://doi.org/10.1016/j.chemosphere.2006.12.071
Burkhead JL, Reynolds KAG, Abdel-Ghany SE, Cohu CM, Pilon M (2009) Copper homeostasis. New Phytol 182:799–816. https://doi.org/10.1111/j.1469-8137.2009.02846.x
Chaignon V, Sanchez-Neira I, Herrmann P, Jaillard B, Hinsinger P (2003) Copper bioavailability and extractability as related to chemical properties of contaminated soils from a vine-growing area. Environ Pollut 123:229–238. https://doi.org/10.1016/S0269-7491(02)00374-3
Cicatelli A, Guarino F, Castiglione S (2017) Reclamation of Cr-contaminated or Cu-contaminated agricultural soils using sunflower and chelants. Environ Sci Pollut Res 24:10131–10138. https://doi.org/10.1007/s11356-017-8655-8
Cuypers A, Vangronsveld J, Clijsters H (2000) Biphasic effect of copper on the ascorbate-glutathione pathway in primary leaves of Phaseolus vulgaris seedlings during the early stages of metal assimilation. Physiol Plant 110:512–517. https://doi.org/10.1111/j.1399-3054.2000.1100413.x
Cuypers A, Vangronsveld J, Clijsters H (2002) Peroxidases in roots and primary leaves of Phaseolus vulgaris. Copper and zinc phytotoxicity: a comparison. J Plant Physiol 159:869–876. https://doi.org/10.1078/0176-1617-00676
Dudonne S, Vitrac X, Coutiere P, Woillez M, Merillon J-M (2009) Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J Agric Food Chem 57:1768–1774. https://doi.org/10.1021/jf803011r
Feigl G, Kumar D, Lehotai N, Pető A, Molnár Á, Rácz É, Ördög A, Erdei L, Kolbert Z, Laskay G (2015) Comparing the effects of excess copper in the leaves of Brassica juncea (L. Czern) and Brassica napus (L.) seedlings: growth inhibition, oxidative stress and photosynthetic damage. Acta Biol Hung 66:205–221. https://doi.org/10.1556/018.66.2015.2.7
Gerola AP, Tsubone TM, Santana A, de Oliveira HP, Hioka N, Caetano W (2011) Properties of chlorophyll and derivatives in homogeneous and microheterogeneous systems. J Phys Chem B 115:7364–7373. https://doi.org/10.1021/jp201278b
Gruber BR, Davies LRR, Kruger EL, McManus PS (2009) Effects of copper-based fungicides on foliar gas exchange in tart cherry. Plant Dis 93:512–518. https://doi.org/10.1094/PDIS-93-5-0512
Guo W, Nazim H, Liang Z, Yang D (2016) Magnesium deficiency in plants: an urgent problem. The Crop Journal 4:83–91. https://doi.org/10.1016/j.cj.2015.11.003
Hänsch R, Mendel RR (2009) Physiological functions of mineral micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, Cl). Curr Opin Plant Biol 12:259–266. https://doi.org/10.1016/j.pbi.2009.05.006
Hartley-Whitaker J, Ainsworth G, Meharg AA (2001) Copper- and arsenate-induced oxidative stress in Holcus lanatus L. clones with differential sensitivity. Plant Cell Environ 24:713–722. https://doi.org/10.1046/j.0016-8025.2001.00721.x
Herzig R, Nehnevajova E, Pfistner C, Schwitzguébel J-P, Ricci A, Keller C (2014) Feasibility of labile Zn phytoextraction using enhanced tobacco and sunflower: results of five- and one-year field-scale experiments in Switzerland. Int J Phytoremediation 16:735–754. https://doi.org/10.1080/15226514.2013.856846
Hewitt E (1966) Sand and water culture methods used in the study of plant nutrition. The Eastern press Ltd, London
Hossain PP, Teixeira da Silva JA, Fujita M (2012) Molecular mechanism of heavy metal toxicity and tolerance in plants: central role of glutathione in detoxification of reactive oxygen species and methylglyoxal and in heavy metal chelation. J Bot 2012, article ID 872875, 37 pages. https://doi.org/10.1155/2012/872875
Hötzer B, Ivanov R, Bauer P, Jung G (2012) Investigation of copper homeostasis in plant cells by fluorescence lifetime imaging microscopy. Plant Signal Behav 7:521–523. https://doi.org/10.4161/psb.19561
INRA LAS (2014) Sols: méthodes applicables aux sols. Available at https://www6npcinrafr/las/Methodes-d-analyse/Sols Access on December 26, 2017
ISO (2012) Soil quality - Determination of the effects of pollutants on soil flora, Part 2: Effects of chemicals on the emergence and growth of higher plants, ISO 11269–2:2012, Geneva. https://www.iso.org/standard/51382.html
Japenga J, Koopmans GF, Song J, Romkens PFAM (2007) A feasibility test to estimate the duration of phytoextraction of heavy metals from polluted soils. Int J Phytoremediation 9:115–132. https://doi.org/10.1080/15226510701232773
Jones S, Bardos P, Kidd PS, Mench M, de Leij F, Hutchings T, Cundy A, Joyce C, Soja G, Friesl-Hanl W, Herzig R, Menger P (2016) Biochar and compost amendments enhance copper immobilisation and support plant growth in a contaminated soil. J Environ Manag 171:101–112. https://doi.org/10.1016/j.jenvman.2016.01.024
Kabata-Pendias A, Pendias H (2011) Trace elements in soils and plants, 4th edn. Boca Raton CRC Press, Boca Raton
Kato M, Shimizu S (1987) Chlorophyll metabolism in higher-plants. VII. Chlorophyll degradation in senescing tobacco leaves—phenolic-dependent peroxidative degradation. Can J Bot - Rev Can Bot 65:729–735. https://doi.org/10.1139/b87-097
Kidd P, Mench M, Álvarez-López V, Bert V, Dimitriou I, Friesl-Hanl W, Herzig R, Janssen JO, Kolbas A, Müller I, Neu S, Renella G, Ruttens A, Vangronsveld J, Puschenreiter M (2015) Agronomic practices for improving gentle remediation of trace-element-contaminated soils. Int J Phytoremediation 17:1005–1037. https://doi.org/10.1080/15226514.2014.1003788
Kinraide TB, Pedler JF, Parker DR (2004) Relative effectiveness of calcium and magnesium in the alleviation of rhizotoxicity in wheat induced by copper, zinc, aluminum, sodium, and low pH. Plant Soil 259:201–208. https://doi.org/10.1023/B:PLSO.0000020972.18777.99
Knezevic SZ, Streibig JC, Ritz C (2007) Utilizing R software package for dose-response studies: the concept and data analysis. Weed Technol 21:840–848. https://doi.org/10.1614/WT-06-161.1
Kolbas A, Mench M, Herzig R, Nehnevajova E, Bes CM (2011) Copper phytoextraction in tandem with oilseed production using commercial cultivars and mutant lines of sunflower. Int J Phytoremediation 13(Suppl 1):55–76. https://doi.org/10.1080/15226514.2011.568536
Kolbas A, Mench M, Marchand L, Herzig R, Nehnevajova E (2014) Phenotypic seedling responses of a metal-tolerant mutant line of sunflower growing on a Cu-contaminated soil series: potential uses for biomonitoring of Cu exposure and phytoremediation. Plant Soil 376:377–397. https://doi.org/10.1007/s11104-013-1974-8
Kopittke PM, Asher CJ, Blamey FPC, Menzies NW (2009) Toxic effects of Cu(2+) on growth, nutrition, root morphology, and distribution of Cu in roots of Sabi grass. Sci Total Environ 407:4616–4621. https://doi.org/10.1016/j.scitotenv.2009.04.041
Korpe DA, Aras S (2011) Evaluation of copper-induced stress on eggplant (Solanum melongena L.) seedlings at the molecular and population levels by use of various biomarkers. Mutat Res Genet Toxicol Environ Mutagen 719:29–34. https://doi.org/10.1016/j.mrgentox.2010.10.003
Küpper H, Kroneck PMH (2005) Heavy metal uptake by plants and cyanobacteria. In: Sigel A, Sigel H, Sigel RKO (eds) Metal ions in biological systems. Marcel Dekker, New York, pp 97–142
Küpper H, Setlik I, Setlikova E, Ferimazova N, Spiller M, Küpper FC (2003) Copper-induced inhibition of photosynthesis: limiting steps of in vivo copper chlorophyll formation in Scenedesmus quadricauda. Funct Plant Biol 30:1187–1196. https://doi.org/10.1071/FP03129
Lagriffoul A, Mocquot B, Mench M, Vangronsveld J (1998) Cadmium toxicity effects on growth, mineral and chlorophyll contents, and activities of stress related enzymes in young maize plants (Zea mays L.) Plant Soil 200:241–250. https://doi.org/10.1023/A:100434690
Lequeux H, Hermans C, Lutts S, Verbruggen N (2010) Response to copper excess in Arabidopsis thaliana: impact on the root system architecture, hormone distribution, lignin accumulation and mineral profile. Plant Physiol Biochem 48:673–682. https://doi.org/10.1016/j.plaphy.2010.05.005
Lin JX, Jiang WS, Liu DH (2003) Accumulation of copper by roots, hypocotyls, cotyledons and leaves of sunflower (Helianthus annuus L.) Bioresour Technol 86:151–155. https://doi.org/10.1016/S0960-8524(02)00152-9
Liotenberg S, Steunou AS, Durand A, Bourbon ML, Bollivar D, Hansson M, Astier C, Ouchane S (2015) Oxygen-dependent copper toxicity: targets in the chlorophyll biosynthesis pathway identified in the copper efflux ATPase CopA deficient mutant. Environ Microbiol 17:1963–1976. https://doi.org/10.1111/1462-2920.12733
Luo ZB, He J, Polle A, Rennenberg H (2016) Heavy metal accumulation and signal transduction in herbaceous and woody plants: paving the way for enhancing phytoremediation efficiency. Biotechnol Adv 34:1131–1148. https://doi.org/10.1016/j.biotechadv.2016.07.003
Lyubenova L, Nehnevajova E, Herzig R, Schroeder P (2009) Response of antioxidant enzymes in Nicotiana tabacum clones during phytoextraction of heavy metals. Environ Sci Pollut Res Int 16:573–581. https://doi.org/10.1007/s11356-009-0175-8
MacDonald-Wicks LK, Wood LG, Garg ML (2006) Methodology for the determination of biological antioxidant capacity in vitro: a review. J Sci Food Agric 86:2046–2056. https://doi.org/10.1002/jsfa
Macnicol RD, Beckett PHT (1985) Critical tissue concentrations of potentially toxic elements. Plant Soil 85:107–129. https://doi.org/10.1007/BF02197805
Madejon P, Murillo JM, Maranon T, Cabrera F, Soriano MA (2003) Trace element and nutrient accumulation in sunflower plants two years after the Aznalcollar mine spill. Sci Total Environ 307:239–257. https://doi.org/10.1016/S0048-9697(02)00609-5
Marchand L, Lamy P, Bert V, Quintela-Sabaris C, Mench M (2016) Potential of Ranunculus acris L. for biomonitoring trace element contamination of riverbank soils: photosystem II activity and phenotypic responses for two soil series. Environ Sci Pollut Res 23:3104–3119. https://doi.org/10.1007/s11356-015-4646-9
Markert B, Oehlmann J, Roth M (1997) Biomonitoring von Schwermetallen – eine kritische Bestandsaufnahme (biomonitoring of heavy metals—a critical review). Z Ökologie u Naturschutz 6:1–8
Marschner H (2011) Mineral nutrition of higher plants, 3rd edn. Academic press, London
Meers E, Ruttens A, Geebelen W, Vangronsveld J, Samson R, Vanbroekhoven K, Vandegehuchte M, Diels L, Tack FMG (2006) Potential use of the plant antioxidant network for environmental exposure assessment of heavy metals in soils. Environ Monit Assess 120:243–267. https://doi.org/10.1007/s10661-005-9059-7
Mench M, Lepp N, Bert V, Schwitzguébel J-P, Gawronski SW, Schroeder P, Vangronsveld J (2010) Successes and limitations of phytotechnologies at field scale: outcomes, assessment and outlook from COST action 859. J Soils Sediments 10:1039–1070. https://doi.org/10.1007/s11368-010-0190-x
Mendoza-Soto AB, Sanchez F, Hernandez G (2012) MicroRNAs as regulators in plant metal toxicity response. Front Plant Sci 3:105. https://doi.org/10.3389/fpls.2012.00105
Mijovilovich A, Leitenmaier B, Meyer-Klaucke W, Kroneck PMH, Goetz B, Küpper H (2009) Complexation and toxicity of copper in higher plants. II. Different mechanisms for copper versus cadmium detoxification in the copper-sensitive cadmium/zinc hyperaccumulator Thlaspi caerulescens (Ganges ecotype). Plant Physiol 151:715–731. https://doi.org/10.1104/pp.109.144675
Mocquot B, Vangronsveld J, Clijsters H, Mench M (1996) Copper toxicity in young maize (Zea mays L.) plants: effects on growth, mineral and chlorophyll contents, and enzyme activities. Plant Soil 182:287–300. https://doi.org/10.1007/BF00029060
Moreno-Jimenez E, Beesley L, Lepp NW, Dickinson NM, Hartley W, Clemente R (2011) Field sampling of soil pore water to evaluate trace element mobility and associated environmental risk. Environ Pollut 159:3078–3085. https://doi.org/10.1016/j.envpol.2011.04.004
Nehnevajova E, Herzig R, Bourigault C, Bangerter S, Schwitzguébel J-P (2009) Stability of enhanced yield and metal uptake by sunflower mutants for improved phytoremediation. Int J Phytoremediation 11:329–346. https://doi.org/10.1080/15226510802565394
Nehnevajova E, Lyubenova L, Herzig R, Schroeder P, Schwitzguébel J-P, Schmuelling T (2012) Metal accumulation and response of antioxidant enzymes in seedlings and adult sunflower mutants with improved metal removal traits on a metal-contaminated soil. Environ Exp Bot 76:39–48. https://doi.org/10.1016/j.envexpbot.2011.10.005
Oustrière N, Marchand L, Lottier N, Motelica M, Mench M (2017) Long-term Cu stabilization and biomass yields of Giant reed and poplar after adding a biochar, alone or with iron grit, into a contaminated soil from a wood preservation site. Sci Total Environ 579:620–627. https://doi.org/10.1016/j.scitotenv.2016.11.048
Ozgen M, Reese RN, Tulio AZ, Scheerens JC, Miller AR (2006) Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2′-diphenyl-1-picrylhydrazyl (DPPH) methods. J Agric Food Chem 54:1151–1157. https://doi.org/10.1021/jf051960d
Palmer CM, Guerinot ML (2009) Facing the challenges of Cu, Fe and Zn homeostasis in plants. Nat Chem Biol 5:333–340. https://doi.org/10.1038/nchembio.166
Panou-Filotheou H, Bosabalidis AM, Karataglis S (2001) Effects of copper toxicity on leaves of oregano (Origanum vulgare subsp hirtum). Ann Botany 88:207–214. https://doi.org/10.1006/anbo.2001.1441
Pätsikkä E, Kairavuo M, Sersen F, Aro EM, Tyystjärvi E (2002) Excess copper predisposes photosystem II to photoinhibition in vivo by outcompeting iron and causing decrease in leaf chlorophyll. Plant Physiol 129:1359–1367. https://doi.org/10.1104/pp.004788
Peñarrubia L, Romero P, Carrió-Seguí A, Andrés-Bordería A, Moreno J, Sanz A (2015) Temporal aspects of copper homeostasis and its crosstalk with hormones. Front Plant Sci 6:255. https://doi.org/10.3389/fpls.2015.00255
Perry JJP, Shin DS, Getzoff ED, Tainer JA (2010) The structural biochemistry of the superoxide dismutases. Biochim Biophys Acta 1804:245–262. https://doi.org/10.1016/j.bbapap.2009.11.004
Pilon M (2011) Moving copper in plants. New Phytol 192:305–307. https://doi.org/10.1111/j.1469-8137.2011.03869.x
Printz B, Lutts S, Hausman J-F, Sergeant K (2016) Copper trafficking in plants and its implication on cell wall dynamics. Front Plant Sci 7:601. https://doi.org/10.3389/fpls.2016.00601
Prior RL, Wu XL, Schaich K (2005) Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J Agric Food Chem 53:4290–4302. https://doi.org/10.1021/jf0502698
Qi XM, Li PJ, Liu W, Xie LJ (2006) Multiple biomarkers response in maize (Zea mays L.) during exposure to copper. J Environ Sci (China) 18:1182–1188. https://doi.org/10.1016/S1001-0742(06)60059-4
Ravet K, Pilon M (2013) Copper and iron homeostasis in plants: the challenges of oxidative stress. Antioxid Redox Signal 19:919–932. https://doi.org/10.1089/ars.2012.5084
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26(9–10):1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Rivelli AR, De Maria S, Puschenreiter M, Gherbin P (2012) Accumulation of cadmium, zinc, and copper by Helianthus annuus L.: impact on plant growth and uptake of nutritional elements. Int J Phytoremediation 14:320–334. https://doi.org/10.1080/15226514.2011.620649
Rizwan M, Ali S, Rizvi H, Rinklebe J, Tsang DCW, Meers E, Ok YS, Ishaque W (2016) Phytomanagement of heavy metals in contaminated soils using sunflower: a review. Crit Rev Env Sci Technol 46:1498–1528. https://doi.org/10.1080/10643389.2016.1248199
Saglam A, Yetissin F, Demiralay M, Terzi R (2016) Copper stress and responses in plants. In: Parvaiz A (ed) Plant metal interaction, emerging remediation techniques. Elsevier, Amsterdam, pp 21–40. https://doi.org/10.1016/B978-0-12-803158-2.00002-3
Sanchez-Pardo B, Fernandez-Pascual M, Zornoza P (2014) Copper microlocalisation and changes in leaf morphology, chloroplast ultrastructure and antioxidative response in white lupin and soybean grown in copper excess. J Plant Res 127:119–129. https://doi.org/10.1007/s10265-013-0583-1
Sharma SS, Dietz KJ (2009) The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci 14:43–50. https://doi.org/10.1016/j.tplants.2008.10.007
Sheldon AR, Menzies NW (2005) The effect of copper toxicity on the growth and root morphology of Rhodes grass (Chloris gayana Knuth.) in resin buffered solution culture. Plant Soil 278:341–349. https://doi.org/10.1007/s11104-005-8815-3
Shi J, Yuan X, Chen X, Wu B, Huang Y, Chen Y (2011) Copper uptake and its effect on metal distribution in root growth zones of Commelina communis revealed by SRXRF. Biol Trace Elem Res 141:294–304. https://doi.org/10.1007/s12011-010-8710-5
Sinclair SA, Krämer U (2012) The zinc homeostasis network of land plants. Biochim Biophys Acta Mol Cell Res 1823:1553–1567. https://doi.org/10.1016/j.bbamcr.2012.05.016
Singh S, Saxena R, Pandey K, Bhatt K, Sinha S (2004) Response of antioxidants in sunflower (Helianthus annuus L.) grown on different amendments of tannery sludge: its metal accumulation potential. Chemosphere 57:1663–1673. https://doi.org/10.1016/j.chemosphere.2004.07.049
Sun BY, Kan SH, Zhang YZ, Deng SH, Wu J, Yuan H, Qi H, Yang G, Li L, Zhang XH, Xiao H, Wang YJ, Peng H, Li YW (2010) Certain antioxidant enzymes and lipid peroxidation of radish (Raphanus sativus L.) as early warning biomarkers of soil copper exposure. J Hazard Mater 183:833–838. https://doi.org/10.1016/j.jhazmat.2010.07.102
Terres Inovia (2017) http://www.terresinovia.fr/tournesol/cultiver-du-tournesol/atouts-points-cles/stades-reperes/
Thomas G, Stärk HJ, Wellenreuther G, Dickinson BC, Küpper H (2013) Effects of nanomolar copper on water plants—comparison of biochemical and biophysical mechanisms of deficiency and sublethal toxicity under environmentally relevant conditions. Aquat Toxicol 140-141:27–36. https://doi.org/10.1016/j.aquatox.2013.05.008
Thounaojam TC, Panda P, Mazumdar P, Kumar D, Sharma GD, Sahoo L, Panda SK (2012) Excess copper induced oxidative stress and response of antioxidants in rice. Plant Physiol Biochem 53:33–39. https://doi.org/10.1016/j.plaphy.2012.01.006
Vamerali T, Bandiera M, Mosca G (2010) Field crops for phytoremediation of metal-contaminated land. A review. Environ Chem Lett 8:1–17. https://doi.org/10.1007/s10311-009-0268-0
Vangronsveld J, Clijsters H (1994) Toxic effects of metals. In: Farago M (ed) Plants and the chemical elements. VCH Verlagsgesellshaft, Weinheim, pp 149–177
Verdejo J, Ginocchio R, Sauvé S, Salgado E, Neaman A (2015) Thresholds of copper phytotoxicity in field-collected agricultural soils exposed to copper mining activities in Chile. Ecotoxicol Environ Saf 122:171–177. https://doi.org/10.1016/j.ecoenv.2015.07.026
Verdejo J, Ginocchio R, Sauvé S, Mondaca P, Neaman A (2016) Thresholds of copper toxicity to lettuce in field-collected agricultural soils exposed to copper mining activities in Chile. J Soil Sci Plant Nutr 16:154–158. https://doi.org/10.4067/S0718-95162016005000011
Verdoni N, Mench M, Cassagne C, Bessoule JJ (2001) Fatty acid composition of tomato leaves as biomarkers of metal-contaminated soils. Environ Toxicol Chem 20:382–388. https://doi.org/10.1002/etc.5620200220
Vulkan R, Zhao FJ, Barbosa-Jefferson V, Preston S, Paton GI, Tipping E, McGrath SP (2000) Copper speciation and impacts on bacterial biosensors in the pore water of copper-contaminated soils. Environ Sci Technol 34:5115–5121. https://doi.org/10.1021/es0000910
Yruela I (2009) Copper in plants: acquisition, transport and interactions. Funct Plant Biol 36:409–430. https://doi.org/10.1071/FP08288
Yuan M, Li X, Xiao J, Wang S (2011) Molecular and functional analyses of COPT/Ctr-type copper transporter-like gene family in rice. BMC Plant Biol 11:69. https://doi.org/10.1186/1471-2229-11-69
Acknowledgements
This work was financially supported by ADEME, Department of Urban Brownfields and Polluted Sites, Angers, France, the European Commission under the Seventh Framework Programme for Research (FP7-KBBE-266124, GREENLAND), the French National Research Agency (PHYTOCHEM ANR-13-CDII-0005-01), and the ERA-Net FACCE SURPLUS (project INTENSE; http://faccesurplus.org/research-projects/intense/). This study has been carried out in the frameworks of the Cluster of Excellence Labex COTE and the INRA Ecotox. Authors give special thanks to Dr. Jean-Paul Maalouf for his technical assistance. Dr. A. Kolbas is grateful to Galina Brutcova for English editing.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Highlights
• Leaf area, hypocotyl length, root and shoot dry weight yields correlated with total Cu concentration in the soil and the soil pore water.
• Total chlorophyll content and antioxidant capacity in the second leaf pair earlier sensed Cu excess than morphological parameters.
• Shoot Cu removal peaked at 280 μg Cu L−1 in the soil pore water.
Electronic supplementary material
ESM 1
(DOCX 130 kb)
Rights and permissions
About this article
Cite this article
Kolbas, A., Kolbas, N., Marchand, L. et al. Morphological and functional responses of a metal-tolerant sunflower mutant line to a copper-contaminated soil series. Environ Sci Pollut Res 25, 16686–16701 (2018). https://doi.org/10.1007/s11356-018-1837-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1837-1