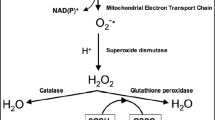
Abstract
We examined the time-course stress responses in the liver of rainbow trout exposed to cuprous chloride (CuCl) and cupric chloride (CuCl2). The treatment groups received a single intraperitoneal injection of CuCl or CuCl2 (both at a dose of 0.01 and 0.05 mg/kg); the control group received only the physiologic solution vehicle. Liver tissue samples were analyzed for total copper, superoxide dismutase, catalase, glutathione peroxidases, glutathione reductase, glutathione S-transferase, glyoxalases, and lactate dehydrogenase at 3, 6, and 9 days post-injection. Total glutathione, metallothionein, and malondialdehyde levels were also measured. The time course of metal accumulation differed between the groups; no dose-response relationship for metal load was found. Both copper species elicited significant changes in oxidative stress markers and in metal trapping. Copper underwent adaptive shifts in glutathione and metallothionein concentrations. The defense strategy primarily versus CuCl2 first involved glutathione, with a peak in metallothionein levels at day 6 for CuCl2 (at both doses) and for CuCl (0.05 mg/kg). Early stimulation of lipid peroxidation was noted after treatment with the higher copper dose and at day 9 after treatment with the lower dose of both CuCl and CuCl2. Antioxidant enzyme activity was impaired due to a more or a less severe oxidative stress condition in relation to the copper species and exposure time. Copper dynamics, in terms of metal accumulation and homeostatic regulation, is noticeably complex. The present findings may advance our understanding of the effects of both copper species on the antioxidant response of rainbow trout.






Similar content being viewed by others
References
Ahmad I, Oliveira M, Pacheco M, Santos MA (2005) Anguilla anguilla L. oxidative stress biomarkers responses to copper exposure with or without naphthoflavone pre-exposure. Chemosphere 61:267–275
Akerboom TPM, Sies H (1981) Assay of glutathione disulfide and glutathione mixed disulfide in biological samples. Meth Enzymol 71:373–382
Al Kaddissi S, Legeay A, Elia AC, Gonzalez P, Floriani M, Cavalie I, Massabuau JC, Gilbin R, Simon O (2014) Mitochondrial gene expression, antioxidant responses, and histopathology after cadmium exposure. Environ Toxicol 29(8):893907
Aliaga ME, López-Alarcón C, Bridi R, Speisky H (2016) Redox-implications associated with the formation of complexes between copper ions and reduced or oxidized glutathione. J Inorg Biochem 154:78–88
Antognelli C, Romani R, Baldracchini F, De Santis A, Andreani G, Talesa V (2003) Different activity of glyoxalase system enzymes in specimens of Sparus auratus exposed to sublethal copper concentrations. Chem Biol Interact 142:297–305
Arellano JM, Storch V, Sarasquete C (1999) Histological changes and copper accumulation in liver and gills of the Senegales sole, Solea senegalensis. Ecotox Environ Saf 44(1):62–72
Atli G, Alptekin Ö, Tükel S, Canli M (2006) Response of catalase activity to Ag+, Cd2+, Cr6+, Cu2+ and Zn2+ in five tissues of freshwater fish Oreochromis niloticus. Comp Biochem Physiol C 143:218–224
Banci L, Bertini I, Ciofi-Baffoni S, Kozyreva T, Zovo K, Paluma P (2010) Affinity gradients drive copper to cellular destinations. Nature 465:645–648
Bisswanger H (2011) Practical Enzymology. Second Edition. 1–360 pp. Wiley-VCH Verlag GmbH and Co. KGaA. ISBN: 978–3–527-31957-2.
Bopp SK, Abicht HK, Knauer K (2008) Copper-induced oxidative stress in rainbow trout gill cells. Aquat Toxicol 86:197–204
Bunton TE, Baksi SM, George SG, Frazier JM (1987) Abnormal hepatic copper storage in a teleost fish (Morone americana). Vet Pathol 24:515–524
Chung PM, Cappel RE, Gilbert HF (1991) Inhibition of glutathione disulfide reductase by glutathione. Arch Biochem Biophys 288:48–53
Cirillo T, Cocchieri RA, Fasano E, Lucisano A, Tafuri S, Ferrante MC, Carpenè E, Andreani G, Isani G (2012) Cadmium accumulation and antioxidant responses in Sparus aurata exposed to waterborne cadmium. Arch Environ Contam Toxicol 62:118–126
Cotou E, Henry M, Zeri C, Rigos G, Torreblanca A, Catsiki VA (2012) Short-term exposure of the European sea bass Dicentrarchus labrax to copper-based antifouling treated nets: copper bioavailability and biomarkers responses. Chemosphere 89:1091–1097
Cozzari M, Elia AC, Pacini N, Smith BD, Boyle D, Rainbow PS, Khan FR (2015) Bioaccumulation and oxidative stress responses measured in the estuarine ragworm (Nereis diversicolor) exposed to dissolved, nano and bulksized silver. Environ Poll 198:32–40
Craig PM, Wood CM, McClelland GB (2007) Oxidative stress response and gene expression with acute copper exposure in zebrafish (Danio rerio). Am J Physiol Regul Integr Comp Physiol 293:1882–1892
Dautremepuits C, Betoulle S, Vernet G (2002) Antioxidant response modulated by copper in healthy or parasitized carp (Cyprinus carpio L.) by Ptychobothrium sp. (Cestoda). Bioch Biophys Acta 1573:4–8
Diamantino TC, Almeida E, Soares A, Guilhermino L (2000) Lactate dehydrogenase activity as an effect criterion in toxicity tests with Daphnia magna straus. Chemosphere 45:553–560
Doyotte A, Cossu C, Jacquin MC, Babut M, Vasseur P (1997) Antioxidant enzymes, glutathione and lipid peroxidation as relevant biomarkers of experimental or field exposure in the gills and the digestive gland of the freshwater bivalve Unio tumidus. Environ Toxicol 39:93–110
Elia AC, Galarini R, Taticchi MI, Dörr AJM, Mantilacci L (2003) Antioxidant responses and bioaccumulation in Ictalurus melas under mercury exposure. Ecotox Environ Saf 55:162–167
Elia AC, Dörr AJM, Galarini R (2007) Comparison of organochlorine pesticides, PCBs and heavy metal contamination and of detoxifying response in tissues of Amerius melas from Corbara, Alviano and Trasimeno Lakes, Italy. Bull Environ Contam Toxicol 78:463–468
Elia AC, Dörr AJM, Abete MC, Prearo M (2010) Seasonal variability of detoxificant response and heavy metal accumulation in tissues of both sexes in Tinca tinca (L.) from Lake Trasimeno. Rev Fish Biol Fish 20:425–434
Elia AC, Pacini N, Fioravanti ML, Dörr AJM, Zaccaroni A, Parmeggiani AM, Gustinelli A, Mordenti O, Abete MC, Prearo M (2016) Detoxifying markers assessment of florfenicol in liver of rainbow trout Oncorhynchus mykiss. J Aquat Anim Health 28:258–265
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Eroglu A, Dogan Z, Kanak EG, Atli G, Canli M (2015) Effects of heavy metals (Cd, Cu, Cr, Pb, Zn) on fish glutathione metabolism. Environ Sci Pollut Res 22:3229–3237
Eyckmans M, Celis N, Horemans N, Blust R, De Boeck G (2011) Exposure to waterborne copper reveals differences in oxidative stress response in three freshwater fish species. Aquat Toxicol 103:112–120
Freedman JH, Ciriolo MR, Peisach J (1989) The role of glutathione in copper metabolism and toxicity. J Biol Chem 264:5598–5605
Fridovich I (1989) Superoxide dismutases. J Biol Chem 264:7761–7764
Fukuan D, Gangchun X, Yan L, Zhijuan N, Pao X (2016) Glyoxalase 1 gene of Coilia nasus: molecular characterization and differential expression during transport stress. Fish Sci 82:719–728
Georgopoulos PG, Roy A, Yonone-Lioy MJ, Opiekun RE, Lioy PJ (2001) Environmental copper: its dynamics and human exposure issues. J Toxicol Environ Health B 4:341–394
Gomes CM, Wittung-Stafshede P (2016) 1. Metal ions, protein folding, and conformational states. Protein folding and metal ions: mechanisms, biology and disease, 1. Protein folding and metal ions. Mechanisms, biology and disease. Ed. Gomes CM. CRC Press, Wittung-Stafshede P, p 287
Greenwald RA (1985) Handbook of methods for oxygen radicals research. CRC Press, Boca Raton, FL
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Hodgson EK, Fridovich I (1975) The interaction of bovine erythrocyte superoxide dismutase with hydrogen peroxide: inactivation of the enzyme. Biochemistry 14:5294–5299
Hoyle I, Shaw BJ, Handy RD (2007) Dietary copper exposure in the African walking catfish, Clarias gariepinus: transient osmoregulatory disturbances and oxidative stress. Aquat Toxicol 83:62–72
Jiménez I, Aracena P, Letelier ME, Navarro P, Speisky H (2002) Chronic exposure of HepG2 cells to excess copper results in depletion of glutathione and induction of metallothionein. Toxicol in Vitro 16:167–175
Kalapos MP (1999) Methylglyoxal in living organisms: chemistry, biochemistry, toxicology and biological implications. Toxicol Lett 110:145–175
Lange A, Ausseil O, Segner H (2002) Alterations of tissue glutathione levels and metallothionein mRNA in rainbow trout during single and combined exposure to cadmium and zinc. Comp Biochem Physiol C: Toxicol Pharmacol 131:231–243
Lavanya S, Ramesh M, Kavitha C, Malarvizhi A (2011) Hematological, biochemical and ionoregulatory responses of Indian major carp Catla catla during chronic sublethal exposure to inorganic arsenic. Chemosphere 82:977–985
Lawrence RA, Burk RF (1976) Glutathione peroxidase activity in selenium-deficient rat liver. Biochem Biophys Res Comm 71:592–598
Linder MC, Hazegh-Azam M (1996) Copper biochemistry and molecular biology. Am J Clin Nutr 63:797S–811S
Liu H, Wang W, Zhang JF, Wang XR (2006) Effects of copper and its ethylenediaminetetraacetate complex on the antioxidant defenses of the goldfish, Carassius auratus. Ecotox Environ Safety 65:350–354
Liu J, Xiaoting S, Meng Q, Luqung Z, Lian C, Yan X (2015) Copper-induced hydrogen peroxide upregulation of metallothionein gene, OsMT2c, from Oryza sativa L. confers copper tolerance in Arabidopsis thaliana. J Hazard Mater 294:99–108
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lushchak VI (2016) Contaminant-induced oxidative stress in fish: a mechanistic approach. Fish Physiol Biochem 42:711–747
Macomber L, Imlay JA (2009) The iron-sulfur clusters of dehydratases are primary intracellular targets of copper toxicity. PNAS 106:8344–8349
Mahboob S (2013) Environmental pollution of heavy metals as a cause of oxidative stress in fish: a review. Life Sci J 10(10s):336–347
Mansouri B, Maleki A, Johari SA, Shahmoradi B, Mohammadi E, Shahsavari S, Davari B (2016) Copper bioaccumulation and depuration in common carp (Cyprinus carpio) following co-exposure to TiO2 and CuO nanoparticles. Arch Environ Contam Toxicol 71(4):541–552
McCord JM, Fridovich I (1969) Superoxide dismutase: an enzymatic function for erythrocuprein (hemocuprein). J Biol Chem 244:6049–6055
McGeer JC, Szebedinszky C, McDonald DG, Wood CM (2000) Effects of chronic sublethal exposure to waterborne Cu, Cd or Zn in rainbow trout 2: tissue specific metal accumulation. Aquat Toxicol 50:245–256
Monteiro SM, Dos Santos N, Calejo M, Fontainhas-Fernandes A, Sousa M (2009) Copper toxicity in gills of the teleost fish, Oreochromis niloticus: effects in apoptosis induction and cell proliferation. Aquat Toxicol 94:219–228
Murakami K, Tsubouchi R, Fukayama M, Yoshino M (2014) Copper-dependent inhibition and oxidative inactivation with affinity cleavage of yeast glutathione reductase. Biometals 27:551–558
Norton SJ, Elia AC, Chyan MK, Gillis G, Frenzel C, Principato GB (1993) Inhibitors and inhibition studies of mammalian glyoxalase II activity. Biochem Soc Trans 21:545–549
Nunes B, Brandão F, Sérgio T, Rodrigues S, Gonçalves F, Correia AT (2014) Effects of environmentally relevant concentrations of metallic compounds on the flatfish Scophthalmus maximus: biomarkers of neurotoxicity, oxidative stress and metabolism. Environ Sci Pollut Res 21:7501–7511
Olsson P-E, Haux C (1985) Rainbow trout metallothionein. Inorg Chim Acta 107:67–71
Olsson PE, Kling P, Erkell LJ, Kille P (1995) Structural and functional analysis of the rainbow trout (Oncorhyncus mykiss) metallothionein-A gene. Eur J Biochem 230:344–349
Pacini N, Prearo M, Abete MC, Brizio P, Dörr AJM, Reimschuessel R, Andersen W, Gasco L, Righetti M, Elia AC (2013) Antioxidant responses and renal crystal formation in rainbow trout treated with melamine administered individually or in combination with cyanuric acid. J Toxicol Environ Health A 76(8):491–508
Pandey S, Parvez S, Ahamd Ansar R, Ali M, Kaur M, Hayat F, Ahmad F, Raisuddin S (2008) Effects of exposure to multiple trace metals on biochemical, histological and ultrastructural features of gills of a freshwater fish, Channa punctata Bloch. Chem Biol Interact 174:183–192
Paris-Palacios S, Risbourg SB, Vernet G (2000) Biochemical and (ultra) structural hepatic perturbations of Brachydanio rerio (Teleostei, Cyprinidae) exposed to two sublethal concentrations of copper sulfate. Aquat Toxicol 50:109–124
Pedrajas JR, Peinado J, Lopez-Barea J (1995) Oxidative stress in fish exposed to model xenobiotics: oxidatively modified forms of Cu, Zn-superoxide dismutase as potential biomarkers. Chem Biol Interact 98:267–282
Pham AN, Xing G, Miller CJ, Waite TD (2013) Fenton-like copper redox chemistry revisited: hydrogen peroxide and superoxide mediation of copper-catalyzed oxidant production. J Catalys 301:54–64
Radi AAR, Matkovics B (1988) Effects of metal ions on the antioxidant enzyme activities, protein contents and lipid peroxidation of carp tissues. Comp Biochem Physiol C: Comp Pharmacol 90:69–72
Roméo M, Cosson RP, Gnassia-Barelli M, Risso C, Stien X, Lafaurie M (1997) Metallothionein determination in the liver of the sea bass Dicentrarchus labrax treated with copper and B (a) P. Mar Environ Res 44:275–284
Roméo M, Bennani N, Gnassia-Barelli M, Lafaurie M, Girard JP (2000) Cadmium and copper display different responses towards oxidative stress in the kidney of the sea bass Dicentrarchus labrax. Aquat Toxicol 48:185–194
Ruttkay-Nedecky B, Nejdl L, Gumulec J, Zitka O, Masarik M, Eckschlager T, Stiborova M, Adam V, Kizek R (2013) The role of metallothionein in oxidative stress. Int J Mol Sci 14:6044–6066
Sanchez W, Palluel O, Meunier L, Coquery M, Porcher JM, Aït-Aïssa S (2005) Copper-induced oxidative stress in three-spined stickleback: relationship with hepatic metal levels. Environ Toxicol Pharmacol 19:177–183
Shaw JP, Large AT, Donkin P, Evans SV, Staff FJ, Livingstone DR, Chipman JK (2004) Seasonal variation in cytochrome P450 in immunopositive protein levels, lipid peroxidation and genetic toxicity in digestive gland of the mussel Mytilus edulis. Aquat Toxicol 67:325–333
Simonato JD, Mela M, Doria HB, Guiloski IC, Randi MAF, Carvalho PSM, Meletti PC, Silva de Assis HC, Bianchini A, Martinez CBR (2016) Biomarkers of waterborne copper exposure in the Neotropical fish Prochilodus lineatus. Aquat Toxicol 170:31–41
Speisky H, Gómez M, Burgos-Bravo F, López-Alarcón C, Jullian C, Olea-Azar C, Aliaga ME (2009) Generation of superoxide radicals by copper-glutathione complexes: redox-consequences associated with their interaction with reduced glutathione. Bioorganic Med Chem 17:1803–1810
Srikanth K, Pereira E, Duarte AC, Ahmad I (2013) Glutathione and its dependent enzymes’ modulatory responses to toxic metals and metalloids in fish—a review. Environ Sci Pollut Res 20:2133–2149
Tóth L, Juhász M, Varga T, Csikkel-Szolnoki A, Nemcsók J (1996) Some effect of CuSO4 on carp. J Environ Sci Health B 31:627–635
Varanka Z, Rojik I, Varanka I, Nemcsók J, Ábrahám M (2001) Biochemical and morphological changes in carp (Cyprinus carpio L.) liver following exposure to copper sulfate and tannic acid. Comp Biochem Physiol 128C:467–478
Viarengo A, Ponzano E, Dondero F, Fabbri R (1997) A simple spectrophotometric method for metallothionein evaluation in marine organisms: an application to Mediterranean and Antarctic molluscs. Mar Environ Res 44:69–84
Vutukuru SS, Chintada S, Radha Madhavi K, Venkateswara Rao J, Anjaneyulu Y (2006) Acute effects of copper on superoxide dismutase, catalase and lipid peroxidation in the freshwater teleost fish, Esomus danricus. Fish Physiol Biochem 32:221–229
Yang Y, Maret W, Vallee BL (2001) Differential fluorescence labeling of cysteinyl clusters uncovers high tissue levels of thionein. Proc Natl Acad Sci U S A 98:5556–5559
Acknowledgments
This study was financially supported by “Ricerca Corrente” IZS PLV 10/09 RC, State Veterinary Institute, Turin (Italy). The authors would like to thank Prof. Laura Gasco of the Department of Agricultural, Forest, and Food Sciences (DISAFA), University of Turin (Italy), for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The procedures for the care and management of the fish were conducted in accordance with EC Council Directive 86/609 EEC (Council of the European Communities 1986) implemented by Italian Law (D. Lgs 116/1992) and approved by the Italian Ministry of Health (DGSAF 0024175-P-17/12/2013, D. Lgs. 306/2013-B).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Cinta Porte
Rights and permissions
About this article
Cite this article
Elia, A.C., Magara, G., Righetti, M. et al. Oxidative stress and related biomarkers in cupric and cuprous chloride-treated rainbow trout. Environ Sci Pollut Res 24, 10205–10219 (2017). https://doi.org/10.1007/s11356-017-8651-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-8651-z