Abstract
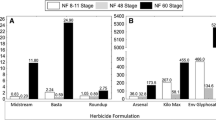
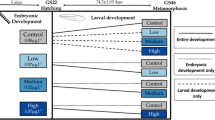
Environmental contamination, especially due to the increasing use of pesticides, is suggested to be one out of six main reasons for the global amphibian decline. Adverse effects of glyphosate-based herbicides on amphibians have been already discussed in several studies with different conclusions, especially regarding sublethal effects at environmentally relevant concentrations. Therefore, we studied the acute toxic effects (mortality, growth, and morphological changes) of the commonly used glyphosate-based herbicide formulation Roundup® UltraMax on early aquatic developmental stages of two anuran species with different larval types (obligate vs. facultative filtrating suspension feeders), the African clawed frog (Xenopus laevis) and the Mediterranean painted frog (Discoglossus pictus). While X. laevis is an established anuran model organism in amphibian toxicological studies, we aim to establish D. pictus as another model for species with facultative filtrating larvae. A special focus of the present study lies on malformations in X. laevis embryos, which were investigated using histological preparations. In general, embryos and larvae of X. laevis reacted more sensitive concerning lethal effects compared to early developmental stages of D. pictus. It was suggested, that especially the different morphology of their filter apparatus and the higher volume of water pumped through the buccopharynx of X. laevis larvae lead to higher exposure to the formulation. The test substance induced similar lethal effects in D. pictus larvae as it does in the teleost standard test organism used in pesticide approval, the rainbow trout (Oncorhynchus mykiss), whereas embryos of both species are apparently more tolerant and, conversely, X. laevis larvae about two times more sensitive. In both species, early larvae always reacted significantly more sensitive than embryos. Exposure to the test substance increased malformation rates in embryos of both species in a concentration-dependent manner, but not at environmentally relevant concentrations. However, the assumed field safety, based on calculated surface water concentrations of the active ingredient (glyphosate), should be validated with realistic field data and buffer strips have to be urgently regarded to any aquatic amphibian habitat.





Similar content being viewed by others
References
ASTM (1998) Standard guide for conducting the frog embryo teratogenesis assay-Xenopus (FETAX)—E1439. ASTM International, West Conshohocken
ASTM (2002) Standard guide for conducting acute toxicity tests on test materials with fishes, macroinvertebrates, and amphibians—E729. ASTM International, West Conshohocken
Attademo AM, Peltzer PM, Lajmanovich RC, Cabagna-Zenklusen MC, Junges CM, Basso A (2014) Biological endpoints, enzyme activities, and blood cell parameters in two anuran tadpole species in rice agroecosystems of mideastern Argentina. Environ Monitor Assess 186:635–649. doi:10.1007/s10661-013-3404-z
Bantle JA, Dumont JN, Finch RA, Linder G, Fort DJ (1998) Atlas of abnormalities: a guide for the performance of FETAX. Oklahoma State University Press, Stillwater
Bantle JA, Finch RA, Fort DJ, Stover EL, Hull M, Kumsher-King M, Gaudet-Hull AM (1999) Phase III interlaboratory study of FETAX. Part 3. FETAX validation using 12 compounds with and without an exogenous metabolic activation system. J Appl Toxicol 19:447–472
Battaglin WA, Rice KC, Focazio MJ, Salmons S, Barry RX (2009) The occurrence of GLY, atrazine, and other pesticides in vernal pools and adjacent streams in Washington, DC, Maryland, Iowa ,and Wyoming, 2005–2006. Environ Monit Assess 155:281–307. doi:10.1007/s10661-008-0435-y
Berger G, Graef F, Pfeffer H (2013) Glyphosate applications on arable fields considerably coincide with migrating amphibians. Sci Rep 3:2622. doi:10.1038/srep02622
Böck P (1989) Romeis Mikroskopische Technik. Urban und Schwarzenberg, München
Böll S, Schmidt BR, Veith M, Wagner N, Rödder D, Weimann C, Kirschey T, Lötters S (2013) Anuran amphibians as indicators of changes in aquatic and terrestrial ecosystems following GM crop cultivation: a monitoring guideline. BioRisk 8:39–51. doi:10.3897/biorisk.8.3251
Bosch J, Andreone F, Tejedo M, Donaire-Barroso D, Lizana M, Martínez-Solano I, Salvador A, García-París M, Recuero Gil E, Slimani T, El Mouden EH, Joger U, Geniez P, Corti C (2009) Discoglossus pictus. The IUCN red list of threatened species 2009: e.T55270 A11285021
Brown CD, van Beinum W (2009) Pesticide transport via sub-surface drains in Europe. Environ Pollut 157:3314–3324. doi:10.1016/j.envpol.2009.06.029
Brühl CA, Pieper S, Weber B (2011) Amphibians at risk? Susceptibility of terrestrial amphibian life stages to pesticides. Environ Toxicol Chem 30:2465–2472. doi:10.1002/etc.650
Brühl CA, Schmidt T, Pieper S, Alscher A (2013) Terrestrial pesticide exposure of amphibians: an underestimated cause of global decrease? Sci Rep 3:1135. doi:10.1038/srep01135
BVL (2010) Glyphosate – Comments from Germany on the paper by Paganelli, A.et al. (2010) “Glyphosate-based herbicides produce teratogenic effects on vertebrates by impairing retinoic acid signaling.” Bundesamt für Verbraucherschutz und Lebensmittelsicherheit, Braunschweig
Collins JP, Storfer A (2003) Global amphibian declines: sorting the hypotheses. Divers Distrib 9:89–98. doi:10.1046/j.1472-4642.2003.00012.x
Costa RN, Nomura F (2016) Measuring the impacts of roundup original® on fluctuating asymmetry and mortality in a Neotropical tadpole. Hydrobiol 765:85–96. doi:10.1007/s10750-015-2404-0
Davidson C, Shaffer HB, Jennings MR (2002) Spatial tests of the pesticide drift, habitat destruction, UV-B, and climate-change hypotheses for California amphibian declines. Conserv Biol 16:1588–1601. doi:10.1046/j.1523-1739.2002.01030.x
Dill GM, Sammons RD, Feng PCC, Kohn F, Kretzmer K, Mehrsheikh A, Bleeke M, Honegger JL, Farmer D, Wright D, Haupfear EA (2010) Glyphosate: discovery, development, applications, and properties. In: Nandula VK (ed) Glyphosate resistance in crops and weeds. John Wiley & Sons, Hoboken, pp. 1–33
Duke SO, Powles SB (2008) Glyphosate: a once-in-a-century herbicide. Pest Manag Sci 64:319–325. doi:10.1002/ps.1518
ECB (2000) IUCLID dataset glyphosate. European Commission, Brussels
Edginton AN, Sheridan PM, Stephenson GR, Thompson DG, Boermans HJ (2004) Comparative effects of pH and vision herbicide on two life stages of four anuran amphibian species. Environ Toxicol Chem 23:815–822
Edwards WM, Triplett GB Jr, Kramer RM (1980) A watershed study of glyphosate transport in runoff. J Environ Qual 9:661–665
Fort DJ, Rogers RL (2005) Enhanced frog embryo teratogenesis assay: Xenopus model using Xenopus tropicalis. In: Ostrander GK (ed) Techniques in aquatic toxicology, vol 2. CRC Press, Boca Raton, pp. 39–54
Giesy JP, Dobson S, Solomon KR (2000) Ecotoxicological risk assessment for roundup herbicide. Rev Environ Contam Toxicol 167:35–120. doi:10.1007/978-1-4612-1156-3_2
Gosner KL (1960) A simple table for staging anuran embryos and larvae with notes on identification. Herpetologica 16:183–190
Greulich K, Pflugmacher S (2003) Differences in susceptibility of various life stages of amphibians to pesticide exposure. Aquat Toxicol 65:329–336
Houlahan JE, Findlay CS, Schmidt BR, Meyer AH, Kuzmin SL (2000) Quantitative evidence for global amphibian population declines. Nature 404:752–755. doi:10.1038/35008052
Howe CM, Berrill M, Pauli BD, Helbing CC, Werry K, Veldhoen N (2004) Toxicity of glyphosate-based pesticides to four north American frog species. Environ Toxicol Chem 23:1928–1938
Jones DK, Hammond JI, Relyea RA (2010) Roundup and amphibians: the importance of concentration, application time, and stratification. Environ Toxicol Chem 29:2016–2025. doi:10.1002/etc.240
Kotthaus A (1933) Die Entwicklung des Primordial-Craniums von Xenopus laevis bis zur Metamorphose. Z Wiss Zool 144:510–572
Lajmanovich RC, Sandoval MT, Peltzer PM (2003) Induction of mortality and malformation in Scinax nasicus tadpoles exposed to glyphosate formulations. Bull Environ Contam Tox 70:612–618. doi:10.1007/s00128-003-0029-x
Lajmanovich RC, Attademo AM, Peltzer PM, Junges CM, Cabagna MC (2011) Toxicity of four herbicide formulations with glyphosate on Rhinella arenarum (Anura: Bufonidae) tadpoles: B-esterases and glutathione S-transferase inhibitors. Arch Environ Contam Toxicol 60:681–689. doi:10.1007/s00244-010-9578-2
Lajmanovich RC, Attademo AM, Simoniello MF, Poletta GL, Junges CM, Peltzer PM, Grenón P, Cabagna-Zenklusen MC (2015) Harmful effects of the dermal intake of commercial formulations containing chlorpyrifos, 2,4-D, and glyphosate on the common toad Rhinella arenarum (Anura: Bufonidae). Water Air Soil Poll 226:427. doi:10.1007/s11270-015-2695-9
Lenkowski JR, Sanchez-Bravo G, McLaughlin KA (2010) Low concentrations of atrazine, glyphosate, 2: 4-dichlorophenoxyacetic acid, and triadimefon exposures have diverse effects on Xenopus laevis organ morphogenesis. J Environ Sci 22:1305–1308. doi:10.1016/S1001-0742(09)60254-0
Mann RM, Bidwell JR (1999) The toxicity of glyphosate and several glyphosate formulations to four species of southwestern Australian frogs. Arch Environ Contam Toxicol 36:193–199. doi:10.1007/s002449900460
Mann RM, Bidwell JR (2001) The acute toxicity of agricultural surfactants to the tadpoles of four Australian and two exotic frogs. Environ Pollut 114:195–205. doi:10.1016/S0269-7491(00)00216-5
Mann RM, Hyne RV, Choung CB, Wilson SP (2009) Amphibians and agricultural chemicals: review of the risks in a complex environment. Environ Pollut 157:2903–2927. doi:10.1016/j.envpol.2009.05.015
McComb BC, Curtis L, Chambers CL, Newton M, Bentson K (2008) Acute toxic hazard evaluations of glyphosate herbicide on terrestrial vertebrates of the Oregon coast range. Environ Sci Pollut Res 15:266–272
McCoy KA, Bortnick LJ, Campbell CM, Hamlin HJ, Guillette LJ Jr, St. Mary CM (2008) Agriculture alters gonadal form and function in the toad Bufo marinus. Environ Health Perspect 11:1526–1532. doi:10.1289/ehp.11536
Nieuwkoop PD, Faber J (1956) Normal table of Xenopus laevis (Daudin). North Holland Publishers, Amsterdam
OECD (2009) Test No. 231: amphibian metamorphosis assay. In: OECD guidelines for testing of chemicals. Section 2: effects on biotic systems. OECD Publishing, Paris
Orton GL (1953) The systematics of vertebrate larvae. Syst Zool 2:63–75. doi:10.2307/sysbio/2.2.63
Paganelli A, Gnazzo V, Acosta H, Lopez SL, Carrasco AE (2010) Glyphosate-based herbicides produce teratogenic effects on vertebrates by impairing retinoic acid signaling. Chem Res Toxicol 23:1586–1595. doi:10.1021/tx1001749
Payton ME, Greenstone MH, Schenker N (2003) Overlapping confidence intervals or SE intervals: what do they mean in terms of statistical significance? J Insect Sci 3:34
Peltzer PM, Lajmanovich RC, Attademo AM, Junges CM, Cabagna-Zenklusen MC, Repetti MR, Sigrist ME, Beldoménico H (2013) Effect of exposure to contaminated pond sediments on survival, development, and enzyme and blood biomarkers in veined treefrog (Trachycephalus typhonius) tadpoles. Ecotoxicol Environ Saf 98:142–151. doi:10.1016/j.ecoenv.2013.09.010
Perkins PJ, Boermans HJ, Stephenson GR (2000) Toxicity of glyphosate and triclopyr using the frog embryo teratogenesis assay-Xenopus. Environ Toxicol Chem 19:940–945. doi:10.1002/etc.5620190422
Puglis HJ, Boone MD (2011) Effects of technical-grade active ingredient vs. commercial formulation of seven pesticides in the presence or absence of UV radiation on survival of green frog tadpoles. Arch Environ Contam Toxicol 60:145–155. doi:10.1007/s00244-010-9528-z
Quaranta A, Bellantuono V, Cassano G, Lippe C (2009) Why amphibians are more sensitive than mammals to xenobiotics. PLoS One 4:e7699. doi:10.1371/journal.pone.0007699
Relyea RA (2005a) The lethal impact of Roundup® and predatory stress on six species of North American tadpoles. Arch Environ Contam Tox 48:351–357. doi:10.1007/s00244-004-0086-0
Relyea RA (2005b) The lethal impact of Roundup® on aquatic and terrestrial amphibians. Ecol Appl 15:1118–1124. doi:10.1890/04-1291
Relyea RA (2006) The impact of insecticides and herbicides on the biodiversity and productivity of aquatic communities: response. Ecol Appl 16:2027–2034. doi:10.1890/1051-0761(2006)016[2027:TIOIAH]2.0.CO;2
Schmidt BR (2004) Pesticides, mortality and population growth rate. Trends Ecol Evol 19:459–460. doi:10.1016/j.tree.2004.06.006
Seale DB, Wassersug RJ (1979) Suspension feeding dynamics of anuran larvae related to their functional morphology. Oecologia 39:259–272. doi:10.1007/BF00345438
Smalling KL, Fellers GM, Kleeman PM, Kuivila KM (2013) Accumulation of pesticides in pacific chorus frogs (Pseudacris regilla) from California's sierra Nevada Mountains, USA. Environ Toxicol Chem 32:2026–2034. doi:10.1002/etc.2308
Smalling KL, Reeves R, Muths E, Vandever M, Battaglin WA, Hladik ML, Pierce CL (2015) Pesticide concentrations in frog tissue and wetland habitats in a landscape dominated by agriculture. Sci Total Environ 502:80–90. doi:10.1016/j.scitotenv.2014.08.114
Soloneski S, de Arcaute CR, Larramendy ML (2016) Genotoxic effect of a binary mixture of dicamba- and glyphosate-based commercial herbicide formulations on Rhinella arenarum (Hensel, 1867) (Anura, Bufonidae) late-stage larvae. Environ Sci Pollut Res 23:17811–17821. doi:10.1007/s11356-016-6992-7
Stuart SN, Hoffmann M, Chanson JS, Cox NA, Berridge RJ, Ramani P, Young B (2008) Threatened amphibians of the world. Lynx Editions, Barcelona
Takahashi M (2007) Oviposition site selection: pesticide avoidance by gray treefrogs. Environ Toxicol Chem 26:1476–1480. doi:10.1897/06-511R.1
Taylor WR, Van Dyke GC (1985) Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium 9:107–119
Thompson DG, Wojtaszek BF, Staznik B, Chartrand DT, Stephenson GR (2004) Chemical and biomonitoring to assess potential acute effects of vision herbicide on native amphibian larvae in forest wetlands. Environ Toxicol Chem 23:843–849. doi:10.1897/02-280
Thompson DG, Solomon KR, Wojtaszek BF, Edginton AN, Stephenson GR (2006) Letters to the editor: the impact of insecticides and herbicides onthe biodiversity and productivity of aquatic communities. Ecol Appl 16:2022–2027
Van Meter RJ, Glinski DA, Henderson WM, Garrison AW, Cyterski M, Purucker ST (2015) Pesticide uptake across the amphibian dermis through soil and overspray exposures. Arch Environ Contam Toxicol 69:545–556. doi:10.1007/s00244-015-0183-2
Vences M, de Pous P, Nicolas V, Díaz-Rodríguez J, Donaire D, Hugemann K, Hauswaldt JS, Amat F, Barnestein JAM, Bogaerts S, Bouazza A, Carranza S, Galán P, González de la Vega JP, Joger U, Lansari A, El Mouden EH, Ohler A, Sanuy D, Slimani T, Tejedo M (2014) New insights on phylogeography and distribution of painted frogs (Discoglossus) in northern Africa and the Iberian Peninsula. Amph-Rep 35:305–320. doi:10.1163/15685381-00002954
Viertel B (1990) Suspension feeding of anuran larvae at low concentrations of Chlorella algae (Amphibia, Anura). Oecologia 85:167–177. doi:10.1007/BF00319398
Viertel B (1992) Functional response of suspension feeding anuran larvae to different particle sizes at low concentrations. Hydrobiol 234:151–173. doi:10.1007/BF00014247
Vonesh RJ, Buck JC (2007) Pesticide alters oviposition site selection by gray treefrogs. Oecologia 154:219–226. doi:10.1007/s00442-007-0811-2
Wagner N, Lötters S (2013a) Effects of water contamination on site selection by amphibians: experiences from an arena approach with European frogs and newts. Arch Environ Contam Toxicol 65:98–104. doi:10.1007/s00244-013-9873-9
Wagner N, Lötters S (2013b) Possible correlation of the worldwide amphibian decline and the increasing use of glyphosate in the agrarian industry. Federal Agency for Nature Conservation, Bonn
Wagner N, Reichenbecher W, Teichmann H, Tappeser B, Lötters S (2013) Questions concerning the potential impact of glyphosate-based herbicides on amphibians. Environ Toxicol Chem 32:1688–1700. doi:10.1002/etc.2268
Wagner N, Rödder D, Brühl CA, Veith M, Lenhardt PP, Lötters S (2014a) Evaluating the risk of pesticide exposure for amphibian species listed in Annex II of the European Union Habitats Directive. Biol Conserv 176:64–70. doi:10.1016/j.biocon.2014.05.014
Wagner N, Züghart W, Mingo V, Lötters S (2014b) Are deformation rates of anuran developmental stages suitable indicators for environmental pollution? Possibilities and limitations. Ecol Indic 45:394–401. doi:10.1016/j.ecolind.2014.04.039
Wagner N, Lötters S, Veith M, Viertel B (2015a) Effects of an environmentally relevant temporal application scheme of low herbicide concentrations on larvae of two anuran species. Chemosphere 135:175–181. doi:10.1016/j.chemosphere.2015.04.028
Wagner N, Lötters S, Veith M, Viertel B (2015b) Acute toxic effects of the herbicide formulation and the active ingredient used in cycloxydim-tolerant maize cultivation on embryos and larvae of the African clawed frog, Xenopus laevis. Bull Environ Contam Toxicol 94:412–418. doi:10.1007/s00128-015-1474-z
Wagner N, Lötters S, Veith M, Viertel B (2015c) Acute toxic effects of the herbicide formulation Focus Ultra® on embryos and larvae of the Moroccan painted frog, Discoglossus scovazzi. Arch Environ Contam Toxicol 69:535–544. doi:10.1007/s00244-015-0176-1
Wells KD (2007) The ecology and behavior of amphibians. The University of Chicago Press, Chicago, London, pp. 1–1148. doi:10.1007/978-0-387-47796-1_3
Weltje L, Simpson P, Gross M, Crane M, Wheeler JR (2013) Comparative acute and chronic sensitivity of fish and amphibians: a critical review of data. Environ Toxicol Chem 32:984–994. doi:10.1002/etc.2149
Williams BK, Semlitsch RD (2010) Larval responses of three Midwestern anurans to chronic, low-dose exposures of four herbicides. Arch Environ Contam Toxicol 58:819–827. doi:10.1007/s00244-009-9390-z
Wilson JG, Warkany J (1965) Teratology: principles and techniques. The University of Chicago Press, Chicago
Weir SM, Yu S, Salice CJ (2012) Acute toxicity of herbicide formulations and chronic toxicity of technical-grade trifluralin to larval green frogs (Lithobates clamitans). Environ Toxicol Chem 31:2029–2034. doi:10.1002/etc.1910
Yu S, Wakes MR, Cai Q, Maul JD, Cobb BP (2013) Lethal and sublethal effects of three insecticides on two developmental stages of Xenopus laevis and comparison with other amphibians. Environ Toxicol Chem 32:2056–2064. doi:10.1002/etc.2280
Acknowledgments
Permissions were obtained with courtesy by the Landesuntersuchungsamt (Koblenz, Germany), the Veterinary Office Trier-Saarburg, and the Nature Conservation Authority Trier, Germany. N. W. is grateful for the financial support from the “Graduiertenkolleg 1319 – Verbesserung von Normsetzung und Normanwendung im integrierten Umweltschutz durch rechts und naturwissenschaftliche Kooperation” at Trier University, which was funded by the German Research Foundation. We thank Frank Pasmans from Gent University for providing eggs from captivity-bred D. pictus and Katja Flebel (Jena) for preparing the histological sections.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Wagner, N., Müller, H. & Viertel, B. Effects of a commonly used glyphosate-based herbicide formulation on early developmental stages of two anuran species. Environ Sci Pollut Res 24, 1495–1508 (2017). https://doi.org/10.1007/s11356-016-7927-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-7927-z