Abstract
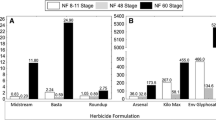
For regulatory and scientific purposes, there is a need to understand the sensitivity of a wider variety of wild species of amphibians and the sensitivities within their life stages to chemicals of widespread use such as herbicides. We investigated the acute toxic effects of the herbicide formulation Focus Ultra [with the active ingredient (a.i.) cycloxydim plus solvent naphtha and sodium dioctylsulphosuccinate as added substances] on embryos and early stage larvae of the Moroccan painted frog (Discoglossus scovazzi). Different clinical signs (twitching, convulsion, and narcosis) occurred at 40 and 80 mg/L in embryos (4 and 8 mg a.i./L) and narcotic effects (total immobilization or irregular escape responses) at 10, 15, and 20 mg/L in larvae (1, 1.5, and 2 mg a.i./L). Growth inhibition (total length), starting at 20 mg/L in embryos and 2.5 mg/L in larvae (2 and 0.25 mg a.i./L, respectively) was understood as sign of toxicity (retardation) and not as sign of teratogenicity. However, the connection to teratogenesis remained unclear though total length reduction occurred at concentrations <20 % of the 96-h LC50 value and at a minimum concentration that inhibits growth of only 17 % of the 96-h LC50 value. Starting at 20 mg/L, mortality in embryos significantly increased and at 15 mg/L in early larvae (2 and 1.5 mg a.i./L, respectively). Mortality of larvae was enhanced during the first 24 h of exposure to 15 and 20 mg/L (1.5 and 2 mg a.i./L). Morphology of the embryos remained unobtrusive. In contrary, axial malformations significantly increased in the early larvae starting at 10 mg/L (1 mg a.i./L), a concentration free of lethal effects. In all considered end points, larvae were significantly more sensitive than embryos, probably because of developmental and physiological properties or different exposure and bioavailability of the compound. Focus Ultra induced comparable lethal and immobilization effects in D. scovazzi as it does to standard test organisms in pesticide approval. However, to validate the apparent safety in the field, which is based on calculated surface water concentrations of the a.i., more data on real contamination levels is necessary (e.g., peak concentrations, concentrations of added substances). Furthermore, sufficient buffer strips between the farmland and amphibian ponds must be considered, and the effects of the substance on terrestrial life stages have not been assessed yet.


Similar content being viewed by others
References
American Society for Testing and Materials (1998) Standard guide for conducting the frog embryo teratogenesis assay-Xenopus (FETAX). E1439-98. ASTM International, West Conshohocken
American Society for Testing and Materials (2002) Standard guide for conducting acute toxicity tests on test materials with fishes, macroinvertebrates, and amphibians. E 729-96. ASTM International, West Conshohocken
Bantle JA, Dumont JN, Finch RA, Linder G, Fort DJ (1998) Atlas of abnormalities: a guide for the performance of FETAX. Oklahoma State University Press, Stillwater
Bantle JA, Finch RA, Fort DJ, Stover EL, Hull M, Kumsher-King M et al (1999) Phase III interlaboratory study of FETAX. Part 3. FETAX validation using 12 compounds with and without an exogenous metabolic activation system. J Appl Toxicol 19:447–472
Beketov MA, Kefford BJ, Schäfer RB, Liess M (2013) Pesticides reduce regional biodiversity of stream invertebrates. Proc Natl Acad Sci USA 110:11039–11043
Bellantuono V, Cassano G, Lippe C (2014) Pesticides alter ion transport across frog (Pelophylax kl. esculentus) skin. Chem Ecol 30:602–610
Berger G, Graef F, Pfeffer H (2013) Glyphosate applications on arable fields considerably coincide with migrating amphibians. Sci Rep 3:2622
Bernabò I, Sperone E, Tripepi S, Brunelli E (2011) Toxicity of chlorpyrifos to larval Rana dalmatina: acute and chronic effects on survival, development, growth and gill apparatus. Arch Environ Contam Toxicol 61:704–718
Bernal MH, Solomon KR, Carrasquilla G (2009) Toxicity of formulated glyphosate (Glyphos) and Cosmo-Flux to larval and juvenile Colombian frogs. 2. Field and laboratory microcosm acute toxicity. J Toxicol Environ Health A 72:966–973
Böll S, Schmidt BR, Veith M, Wagner N, Rödder D, Weimann C et al (2013) Anuran amphibians as indicators of changes in aquatic and terrestrial ecosystems following GM crop cultivation: a monitoring guideline. Biorisk 8:39–51
Boone MD, Cowman D, Davidson C, Hayes T, Hopkins W, Relyea RA, et al. (2007) Evaluating the role of environmental contamination in amphibian population declines. In: Gascon C, Collins JP, Moore RD, Church DR, McKay JE, Mendelson JR III (eds) Amphibian conservation action plan. IUCN/SSC Amphibian Specialist Group, Gland, pp 32–36
Boutilier RG, Stiffler DF, Toews DP (1992) Exchange of respiratory gases, ions, and water in amphibious and aquatic amphibians. In: Feder ME, Burggren WW (eds) Environmental physiology of the amphibians. The University of Chicago Press, Chicago, pp 81–124
Brown CD, van Beinum W (2009) Pesticide transport by way of subsurface drains in Europe. Environ Pollut 157:3314–3324
Brühl CA, Pieper S, Weber B (2011) Amphibians at risk? Susceptibility of terrestrial amphibian life stages to pesticides. Environ Toxicol Chem 30:2465–2472
Brühl CA, Schmidt T, Pieper S, Alscher A (2013) Terrestrial pesticide exposure of amphibians: an underestimated cause of global decrease? Sci Rep 3:1135
Burton JD, Gronwald JW, Somers DA, Gengenbach BG, Wyse DL (1989) Inhibition of corn acetyl-CoA carboxylase by cyclohexanedione and aryloxyphenoxypropionate herbicides. Pestic Biochem Physiol 34:76–85
Collins JP, Storfer A (2003) Global amphibian decreases: sorting the hypotheses. Divers Distrib 9:89–98
Davidson C (2004) Declining downwind: amphibian population decreases in California and historical pesticide use. Ecol Appl 14:1892–1902
Duke SO, Powles SB (2008) Glyphosate: a once-in-a-century herbicide. Pest Manag Sci 64:319–325
Edge CB, Gahl MK, Thompson DG, Houlahan JE (2013) Labortaory and field exposure of two species of juvenile amphibians to a glyphosate-based herbicide and Batrachochytrium dendrobatidis. Sci Total Environ 44:145–152
Edge CB, Thompson DG, Hao C, Houlahan JE (2014) The response of amphibian larvae to exposure to a glyphosate-based herbicide (Roundup WeatherMax) and nutrient enrichment in an ecosystem experiment. Ecotoxicol Environ Saf 109:124–132
Edginton AN, Sheridan PM, Stephenson GR, Thompson DG, Boermans HJ (2004) Comparative effects of pH and vision herbicide on two life stages of four anuran amphibian species. Environ Toxicol Chem 23:815–822
Edwards WM, Triplett GB Jr, Kramer RM (1980) A watershed study of glyphosate transport in runoff. J Environ Qual 9:661–665
Erickson RJ, McKim JM (1990) A simple flow-limited model for gas exchange of organic chemicals at fish gills. Environ Toxicol Chem 9:159–165
European Food Safety Authority (2010) Conclusion on the peer review of the pesticide risk assessment of the active substance cycloxydim. EFSA J 8:1669
Evans DH (1987) The fish gill: site of action and model for toxic effects of environmental pollutants. Environ Health Perspect 71:47–58
Evans DH, Piermarini PM, Potts WTW (1999) Ionic transport in the fish gill epithelium. J Experiment Zool 283:641–652
Evans DH, Piermarini PM, Choe KP (2006) The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation and excretion of nitrogeneous waste. Physiol Rev 85:97–177
Feder ME, Burggren WW (1985) Cutaneous gas exchange in vertebrates: design, patterns, control, and implications. Biol Rev 60:1–45
Federal Ministry of Food, Agriculture and Consumer Protection (2013) National action plan on sustainable use of plant protection products. Available at: http://www.nap-pflanzenschutz.de/fileadmin/SITE_MASTER/content/Dokumente/Grundlagen/NAP_2013/NAP_2013_En.pdf
Fenwick JC (1989) Calcium exchange across fish gills. In: Pang PKT, Schreibman MP (eds) Vertebrate endocrinology: fundamentals and biomedical implications. Academic, San Diego, pp 319–338
Fort DJ, Rogers RL (2005) Enhanced frog embryo teratogenesis assay: Xenopus model using Xenopus tropicalis. In: Ostrander GK (ed) Techniques in aquatic toxicology, vol 2. CRC Press, Boca Raton, pp 39–54
Fromhage L, Vences M, Veith M (2004) Testing alternative vicariance scenarios in western mediterranean discoglossid frogs. Mol Phylogenet Evol 31:308–322
Fuentes L, Moore LJ, Rodgers JH Jr, Bowerman WW, Yarrow GK, Chao WY (2011) Comparative toxicity of two glyphosate formulations (original formulation of Roundup and Roundup WeatherMAX) to six North American larval anurans. Environ Toxicol Chem 30:2756–2761
Geiger F, Bengtsson J, Berendse F, Weisser WW, Emmerson M, Morales MB et al (2010) Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland. Basic Appl Ecol 11:97–105
Gerhardt J (1999) Warkany lecture: signaling pathways in development. Teratology 60:226–239
Gilbert SF (2010) Developmental biology, 9th edn. Sinauer Associates Inc., Sunderland
Gosner KL (1960) A simple table for staging anuran embryos and larvae with notes on identification. Herpetologica 16:183–190
Gradwell N (1972a) Comments on gill irrigation in Rana fuscigula. Herpetology 28:123–125
Gradwell N (1972b) Gill irrigation in Rana catesbeiana, part I. On the anatomical basis. Can J Zool 50:481–499
Gradwell N (1972c) Gill irrigation in Rana catesbeiana, part II. On the musculoskeletal mechanism. Can J Zool 50:501–521
Gradwell N (1975) The bearing of filter feeding on the water pumping mechanism of Xenopus tadpoles (Anura: Pipidae). Acta Zool 56:119–128
Houlahan JE, Findlay CS, Schmidt BR, Meyer AH, Kuzmin SL (2000) Quantitative evidence for global amphibian population decreases. Nature 404:752–755
Howe CM, Gillis R, Mowbray RC (1998) Effect of chemical synergy and larval stage on the toxicity of atrazine and alachlor to amphibian larvae. Environ Toxicol Chem 17:519–525
Howe CM, Berrill M, Pauli BD, Helbing CC, Werry K, Veldhoen N (2004) Toxicity of glyphosate-based pesticides to four North American frog species. Environ Toxicol Chem 23:1928–1938
Landis DA, Gardiner MM, van der Werf W, Swinton SM (2008) Increasing corn for biofuel production reduces biocontrol services in agricultural landscapes. Proc Natl Acad Sci USA 105:20552–20557
Lenhardt PP, Brühl CA, Berger G (2014) Temporal coincidence of amphibian migration and pesticide applications on arable fields in spring. Basic Appl Ecol 16:54–63
Malaj E, von der Ohe PC, Grote M, Kühne R, Mondy CP, Usseglio-Polatera P et al (2014) Organic chemicals jeopardize the health of freshwater ecosystems on the continental scale. Proc Natl Acad Sci USA 111:9549–9554
Mann RM, Bidwell JR (1999) The toxicity of glyphosate and several glyphosate formulations to four species of Southwestern Australian frogs. Arch Environ Contam Toxicol 36:193–199
Mann RM, Bidwell JR (2000) Application of the FETAX protocol to assess the developmental toxicity of nonylphenol ethoxylate to Xenopus laevis and two Australian frogs. Aquat Toxicol 51:19–29
Mann RM, Bidwell JR (2001) The acute toxicity of agricultural surfactants to the tadpoles of four Australian and two exotic frogs. Environ Pollut 114:195–205
Mann RM, Bidwell JR, Tyler MJ (2003) Toxicity of herbicide formulations to frogs and the implications for product registration: a case study from Western Australia. Appl Herpetol 1:13–22
Mann RM, Hyne RV, Choung CB, Wilson SP (2009) Amphibians and agricultural chemicals: review of the risks in a complex environment. Environ Pollut 157:2903–2927
McComb BC, Curtis L, Chambers CL, Newton M, Bentson K (2008) Acute toxic hazard evaluations of glyphosate herbicide on terrestrial vertebrates of the Oregon coast range. Environ Sci Pollut Res 15:266–272
Organisation for Economic Co-operation and Development (OECD) (2009) Test no. 231: Amphibian metamorphosis assay. In: OECD Guidelines for Testing of Chemicals. Section 2: Effects on biotic systems. OECD Publishing, Paris
Payton ME, Greenstone MH, Schenker N (2003) Overlapping confidence intervals or SE intervals: what do they mean in terms of statistical significance? J Insect Sci 3:34
Puglis HJ, Boone MD (2011) Effects of technical-grade active ingredient vs. commercial formulation of seven pesticides in the presence or absence of UV radiation on survival of green frog tadpoles. Arch Environ Contam Toxicol 60:145–155
Quaranta A, Bellantuono V, Cassano G, Lippe C (2009) Why amphibians are more sensitive than mammals to xenobiotics. PLoS One 4:e7699
Relyea RA (2005) The lethal impact of Roundup on aquatic and terrestrial amphibians. Ecol Appl 15:1118–1124
Relyea RA (2011) Amphibians are not ready for Roundup. In: Elliott JE, Bishop CA, Morrissey CA (eds) Wildlife ecotoxicology, vol 3., Emerging topics in ecotoxicologySpringer, New York, pp 267–300
Relyea RA, Jones DK (2009) Toxicity of Roundup Original Max to 13 species of larval amphibians. Environ Toxicol Chem 28:2004–2008
Roelants K, Haas A, Bossuyt F (2011) Anuran radiations and the evolution of tadpole morphospace. Proc Natl Acad Sci USA 108:8731–8736
Salvador A, Donaire-Barroso D, Slimani T, El Mouden EH, Geniez P, Perez-Mellado V, et al. (2009) Discoglossus scovazzi. The IUCN red list of threatened species. Version 2014.3. Available at: http://www.iucnredlist.org/details/55272/0
Schmidt BR (2004) Pesticides, mortality and population growth rate. Trends Ecol Evol 19:459–460
Stuart SN, Hoffmann M, Chanson JS, Cox NA, Berridge RJ, Ramani P et al (2008) Threatened amphibians of the world. Lynx Edicions, Barcelona
Thompson DG, Wojtaszek BF, Staznik B, Chartrand DT, Stephenson GR (2004) Chemical and biomonitoring to assess potential acute effects of Vision herbicide on native amphibian larvae in forest wetlands. Environ Toxicol Chem 23:843–849
Ultsch GR, Bradford DF, Freda J (1999) Physiology, coping with the environment. In: Altig R, McDiarmid RW (eds) Tadpoles, the biology of anuran larvae. The University of Chicago Press, Chicago, pp 189–214
Vancetovic J, Vidakovic M, Babic M, Radojcic DB, Bozinovic S, Stevanovic M (2009) The effect of cycloxydim tolerant maize (CTM) alleles on grain yield and agronomic traits of maize single cross hybrid. Maydica 54:91–95
Viertel B (1990) Suspension feeding of anuran larvae at low concentrations of Chlorella algae (Amphibia, Anura). Oecologia 85:167–177
Viertel B (1992) Functional response of suspension feeding anuran larvae to different particle sizes at low concentrations. Hydrobiology 234:151–173
Wagner N, Reichenbecher W, Teichmann H, Tappeser B, Lötters S (2013) Questions concerning the potential impact of glyphosate-based herbicides on amphibians. Environ Toxicol Chem 32:1688–1700
Wagner N, Rödder D, Brühl CA, Veith M, Lenhardt PP, Lötters S (2014a) Evaluating the risk of pesticide exposure for amphibian species listed in Annex II of the European Union Habitats Directive. Biol Conserv 176:64–70
Wagner N, Züghart W, Mingo V, Lötters S (2014b) Are deformation rates of anuran developmental stages suitable indicators for environmental pollution? Possibilities and limitations. Ecol Indic 45:394–401
Wagner N, Lötters S, Veith M, Viertel B (2015) Acute toxic effects of the herbicide formulation and the active ingredient used in cycloxydim-tolerant maize cultivation on embryos and larvae of the African clawed frog, Xenopus laevis. Bull Environ Contam Toxicol 94:412–418
Wassersug R, Hoff K (1979) A comparative study of the buccal pumping mechanism of tadpoles. Biol J Linn Soc 12:225–259
Wassersug R, Hoff K (1982) Developmental changes in the orientation of the anuran jaw suspension. Evol Biol 15:223–246
Williams BK, Semlitsch RD (2010) Larval responses of three Midwestern anurans to chronic, low-dose exposures of four herbicides. Arch Environ Contam Toxicol 58:819–827
Weir SM, Yu S, Salice CJ (2012) Acute toxicity of herbicide formulations and chronic toxicity of technical-grade trifluralin to larval green frogs (Lithobates clamitans). Environ Toxicol Chem 31:2029–2034
Wilson JG, Warkany J (1965) Teratology: principles and techniques. The University of Chicago Press, Chicago
Yu S, Wakes MR, Cai Q, Maul JD, Cobb BP (2013) Lethal and sublethal effects of three insecticides on two developmental stages of Xenopus laevis and comparison with other amphibians. Environ Toxicol Chem 32:2056–2064
Acknowledgments
Permissions were obtained with courtesy by the Landesuntersuchungsamt (Koblenz, Germany), the Veterinary Office, and the Nature Conservation Authority Trier-Saarburg (Trier, Germany). N. W. is grateful for financial support from the ‘Graduiertenkolleg, 1319—Verbesserung von Normsetzung und “Normanwendung im integrierten Umweltschutz durch rechts und naturwissenschaftliche Kooperation” at Trier University, which was funded by the German Research Foundation. Reinhard Bierl (Department of Hydrology, Trier University) performed the water analysis. We thank Frank Pasmans from Gent University for providing eggs from captivity-bred D. scovazzi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wagner, N., Lötters, S., Veith, M. et al. Acute Toxic Effects of the Herbicide Formulation Focus® Ultra on Embryos and Larvae of the Moroccan Painted Frog, Discoglossus scovazzi . Arch Environ Contam Toxicol 69, 535–544 (2015). https://doi.org/10.1007/s00244-015-0176-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-015-0176-1