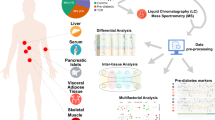
Abstract
Introduction
Diabetes mellitus is a serious metabolic disorder causing multiple organ damage in human. However, the lipidomic profiles in different organs and their associations are rarely studied in either diabetic patients or animals.
Objectives
To evaluate and compare the characteristics of lipid species in serum and multiple tissues in a diabetic mouse model.
Methods
Semi-quantitative profiling analyses of intact and oxidized lipids were performed in serum and multiple tissues from a diabetic mouse model fed a high fat diet and treated with streptozotocin by using LC/HRMS and MS/MS. The total content of each lipid class, and the tissue-specific lipid species in all tissue samples were determined and compared by multivariate analyses.
Results
The diabetic mouse model displayed characteristic differences in serum and multiple organs: the brain and heart showed the largest reduction in cardiolipin, while the kidney had more alterations in triacylglycerol. Interestingly, the lipidomic differences also existed between different regions of the same organ: cardiolipin species with highly polyunsaturated fatty acyls decreased only in atrium but not in ventricle, while renal cortex showed longer fatty acyl chains for both increased and decreased triacylglycerol species than renal medulla. Importantly, diabetes caused an accumulation of lipid hydroperoxides, suggesting that oxidative stress was induced in all organs except for the brain during the development of diabetes.
Conclusions
These findings provided novel insight into the organ-specific relationship between diabetes and lipid metabolism, which might be useful for evaluating not only diabetic tissue injury but also the effectiveness of diabetic treatments.





Similar content being viewed by others
References
Ahmad, M., Ukkalam, M., & Ahmad, I. (2005). Cardiolipin molecules and methods of synthesis. Waukegan: NeoPharm, Inc.
Auberval, N., Dal, S., Bietiger, W., Pinget, M., Jeandidier, N., Maillard-Pedracini, E., et al. (2014). Metabolic and oxidative stress markers in Wistar rats after 2 months on a high-fat diet. Diabetology & Metabolic Syndrome, 6(1), 130. https://doi.org/10.1186/1758-5996-6-130.
Benjamini, Y., & Hochberg, Y. (1995). Controlling the false discovery rate: A practical and powerful approach to multiple testing. Journal of the Royal Statistical Society: Series B (Methodological), 57(1), 289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Bertoni, A. G., Krop, J. S., Anderson, G. F., & Brancati, F. L. (2002). Diabetes-related morbidity and mortality in a national sample of U.S. elders. Diabetes Care, 25(3), 471–475. https://doi.org/10.2337/diacare.25.3.471
Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37(8), 911–917. https://doi.org/10.1139/o59-099
Boretti, F. S., Burla, B., Deuel, J., Gao, L., Wenk, M. R., Liesegang, A., & Sieber-Ruckstuhl, N. S. (2020). Serum lipidome analysis of healthy beagle dogs receiving different diets. Metabolomics, 16(1), 1. https://doi.org/10.1007/s11306-019-1621-3.
Chen, Z., Wu, Y., Ma, Y.-S., Kobayashi, Y., Zhao, Y.-Y., Miura, Y., et al. (2017). Profiling of cardiolipins and their hydroperoxides in HepG2 cells by LC/MS. Analytical and Bioanalytical Chemistry, 409(24), 5735–5745. https://doi.org/10.1007/s00216-017-0515-3.
Chen, Z., Zang, L., Wu, Y., Nakayama, H., Shimada, Y., Shrestha, R., et al. (2018). Lipidomic profiling on oxidized phospholipids in type 2 diabetes mellitus model zebrafish. Analytical Sciences, 34(10), 1201–1208. https://doi.org/10.2116/analsci.18P281
Chicco, A. J., & Sparagna, G. C. (2007). Role of cardiolipin alterations in mitochondrial dysfunction and disease. American Journal of Physiology-Cell Physiology, 292(1), C33–C44. https://doi.org/10.1152/ajpcell.00243.2006.
Choi, J., Yin, T., Shinozaki, K., Lampe, J. W., Stevens, J. F., Becker, L. B., & Kim, J. (2018). Comprehensive analysis of phospholipids in the brain, heart, kidney, and liver: Brain phospholipids are least enriched with polyunsaturated fatty acids. Molecular and Cellular Biochemistry, 442(1–2), 187–201. https://doi.org/10.1007/s11010-017-3203-x
Das, U. (2002). Is type 2 diabetes mellitus a disorder of the brain? Nutrition (Burbank, Los Angeles County, California), 18(7–8), 667–672. https://doi.org/10.1016/S0899-9007(02)00746-3
Das, U. N. (1994). Beneficial effect of eicosapentaenoic and docosahexaenoic acids in the management of systemic lupus erythematosus and its relationship to the cytokine network. Prostaglandins, Leukotrienes and Essential Fatty Acids, 51(3), 207–213. https://doi.org/10.1016/0952-3278(94)90136-8.
Davì, G., Falco, A., & Patrono, C. (2005). Lipid peroxidation in diabetes mellitus. Antioxidants & Redox Signaling, 7(1–2), 256–268. https://doi.org/10.1089/ars.2005.7.256
Evans, J. L., Goldfine, I. D., Maddux, B. A., & Grodsky, G. M. (2002). Oxidative stress and stress-activated signaling pathways: A unifying hypothesis of type 2 diabetes. Endocrine Reviews, 23(5), 599–622. https://doi.org/10.1210/er.2001-0039
Fatani, S. H., Babakr, A. T., NourEldin, E. M., & Almarzouki, A. A. (2016). Lipid peroxidation is associated with poor control of type-2 diabetes mellitus. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 10(2), S64–S67. https://doi.org/10.1016/j.dsx.2016.01.028.
Folch, J., Lees, M., Stanley, G. H. S., & Sloane Stanley, G. H. (1957). A simple method for the isolation and purification of total lipides from animal tissues. The Journal of Biological Chemistry, 226(1), 497–509.
Fritsche, K. L. (2008). Too much linoleic acid promotes inflammation—doesn’t it? Prostaglandins, Leukotrienes and Essential Fatty Acids, 79(3–5), 173–175. https://doi.org/10.1016/j.plefa.2008.09.019
Fukushima, A., & Lopaschuk, G. D. (2016). Cardiac fatty acid oxidation in heart failure associated with obesity and diabetes. Biochimica et Biophysica Acta (BBA)—Molecular and Cell Biology of Lipids, 1861(10), 1525–1534. https://doi.org/10.1016/j.bbalip.2016.03.020
Giugliano, D., Ceriello, A., & Paolisso, G. (1996). Oxidative stress and diabetic vascular complications. Diabetes Care, 19(3), 257–267. https://doi.org/10.2337/diacare.19.3.257
Gómez-Marcos, M. A., Recio-Rodríguez, J. I., Rodríguez-Sánchez, E., Castaño-Sánchez, Y., de Cabo-Laso, A., Sánchez-Salgado, B., et al. (2010). Central blood pressure and pulse wave velocity: relationship to target organ damage and cardiovascular morbidity-mortality in diabetic patients or metabolic syndrome. An observational prospective study. LOD-DIABETES study protocol. BMC Public Health, 10(1), 143. https://doi.org/10.1186/1471-2458-10-143
Greifenhagen, U., Frolov, A., Blüher, M., & Hoffmann, R. (2016). Site-specific analysis of advanced glycation end products in plasma proteins of type 2 diabetes mellitus patients. Analytical and Bioanalytical Chemistry, 408(20), 5557–5566. https://doi.org/10.1007/s00216-016-9651-4.
Grove, K. J., Lareau, N. M., Voziyan, P. A., Zeng, F., Harris, R. C., Hudson, B. G., & Caprioli, R. M. (2018). Imaging mass spectrometry reveals direct albumin fragmentation within the diabetic kidney. Kidney International, 94(2), 292–302. https://doi.org/10.1016/j.kint.2018.01.040.
Ha, C. Y., Kim, J. Y., Paik, J. K., Kim, O. Y., Paik, Y.-H., Lee, E. J., & Lee, J. H. (2012). The association of specific metabolites of lipid metabolism with markers of oxidative stress, inflammation and arterial stiffness in men with newly diagnosed type 2 diabetes. Clinical Endocrinology, 76(5), 674–682. https://doi.org/10.1111/j.1365-2265.2011.04244.x.
Han, X., Yang, J., Yang, K., Zhongdan, Z., Abendschein, D. R., & Gross, R. W. (2007). Alterations in myocardial cardiolipin content and composition occur at the very earliest stages of diabetes: A shotgun lipidomics study. Biochemistry, 46(21), 6417–6428. https://doi.org/10.1021/bi7004015.
Hayasaka, T., Fuda, H., Hui, S.-P., & Chiba, H. (2016). Imaging mass spectrometry reveals a decrease of cardiolipin in the kidney of NASH model mice. Analytical Sciences, 32(4), 473–476. https://doi.org/10.2116/analsci.32.473
Hopps, E., Noto, D., Caimi, G., & Averna, M. R. (2010). A novel component of the metabolic syndrome: The oxidative stress. Nutrition, Metabolism and Cardiovascular Diseases, 20(1), 72–77. https://doi.org/10.1016/j.numecd.2009.06.002.
Hui, S.-P., Chiba, H., Jin, S., Nagasaka, H., & Kurosawa, T. (2010). Analyses for phosphatidylcholine hydroperoxides by LC/MS. Journal of Chromatography B, 878(20), 1677–1682. https://doi.org/10.1016/j.jchromb.2010.04.011.
Hui, S.-P., Murai, T., Yoshimura, T., Chiba, H., & Kurosawa, T. (2003). Simple chemical syntheses of TAG monohydroperoxides. Lipids, 38(12), 1287–1292. https://doi.org/10.1007/s11745-003-1191-9.
International Diabetes Federation. (2017). IDF Diabetes Atlas (8th ed.). Brussels: International Diabetes Federation. http://www.diabetesatlas.org.
Klein, J. P., & Waxman, S. G. (2003). The brain in diabetes: molecular changes in neurons and their implications for end-organ damage. The Lancet Neurology, 2(9), 548–554. https://doi.org/10.1016/S1474-4422(03)00503-9.
Lee, D.-K., Yoon, M. H., Kang, Y. P., Yu, J., Park, J. H., Lee, J., & Kwon, S. W. (2013). Comparison of primary and secondary metabolites for suitability to discriminate the origins of Schisandra chinensis by GC/MS and LC/MS. Food Chemistry, 141(4), 3931–3937. https://doi.org/10.1016/j.foodchem.2013.06.064.
Makino, A., Skelton, M. M., Zou, A., Roman, R. J., & Cowley, A. W. (2002). Increased renal medullary oxidative stress produces hypertension. Hypertension, 39(2), 667–672. https://doi.org/10.1161/hy0202.103469
Mastrocola, R., Restivo, F., Vercellinatto, I., Danni, O., Brignardello, E., Aragno, M., & Boccuzzi, G. (2005). Oxidative and nitrosative stress in brain mitochondria of diabetic rats. Journal of Endocrinology, 187(1), 37–44. https://doi.org/10.1677/joe.1.06269.
Mazumder, P. K., O’Neill, B. T., Roberts, M. W., Buchanan, J., Yun, U. J., Cooksey, R. C., et al. (2004). Impaired cardiac efficiency and increased fatty acid oxidation in insulin-resistant ob/ob mouse hearts. Diabetes, 53(9), 2366–2374. https://doi.org/10.2337/diabetes.53.9.2366
Mora-Ortiz, M., Nuñez Ramos, P., Oregioni, A., & Claus, S. P. (2019). NMR metabolomics identifies over 60 biomarkers associated with Type II Diabetes impairment in db/db mice. Metabolomics, 15(6), 1–16. https://doi.org/10.1007/s11306-019-1548-8.
Moreira, P. I., Santos, M. S., Seiça, R., & Oliveira, C. R. (2007). Brain mitochondrial dysfunction as a link between Alzheimer’s disease and diabetes. Journal of the Neurological Sciences, 257(1–2), 206–214. https://doi.org/10.1016/j.jns.2007.01.017.
Mori, T., Ogawa, S., Cowely, A. W., & Ito, S. (2012). Role of renal medullary oxidative and/or carbonyl stress in salt-sensitive hypertension and diabetes. Clinical and Experimental Pharmacology and Physiology, 39(1), 125–131. https://doi.org/10.1111/j.1440-1681.2011.05653.x.
Muriach, M., Flores-Bellver, M., Romero, F. J., & Barcia, J. M. (2014). Diabetes and the brain: Oxidative stress, inflammation, and autophagy. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2014/102158
Nagashima, T., Oikawa, S., Hirayama, Y., Tokita, Y., Sekikawa, A., Ishigaki, Y., et al. (2002). Increase of serum phosphatidylcholine hydroperoxide dependent on glycemic control in type 2 diabetic patients. Diabetes Research and Clinical Practice, 56(1), 19–25. https://doi.org/10.1016/S0168-8227(01)00353-9.
Palm, F., Cederberg, J., Hansell, P., Liss, P., & Carlsson, P.-O. (2003). Reactive oxygen species cause diabetes-induced decrease in renal oxygen tension. Diabetologia, 46(8), 1153–1160. https://doi.org/10.1007/s00125-003-1155-z.
Papatheodorou, K., Banach, M., Edmonds, M., Papanas, N., & Papazoglou, D. (2015). Complications of diabetes. Journal of Diabetes Research, 2015(4), 1–5. https://doi.org/10.1155/2015/189525
Sas, K. M., Lin, J., Rajendiran, T. M., Soni, T., Nair, V., Hinder, L. M., et al. (2018). Shared and distinct lipid-lipid interactions in plasma and affected tissues in a diabetic mouse model. Journal of Lipid Research, 59(2), 173–183. https://doi.org/10.1194/jlr.M077222.
Shannon, P., Markiel, A., Ozier, O., Baliga, N. S., Wang, J. T., Ramage, D., et al. (2003). Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Research, 13(11), 2498–2504. https://doi.org/10.1101/gr.1239303
Shrestha, R., Chen, Z., Miura, Y., Yamamoto, Y., Sakurai, T., Chiba, H., et al. (2019). Identification of molecular species of phosphatidylcholine hydroperoxides in native and copper-oxidized triglyceride-rich lipoproteins in humans. Annals of Clinical Biochemistry: International Journal of Laboratory Medicine. https://doi.org/10.1177/0004563219880932.
Söderberg, M., Edlund, C., Kristensson, K., & Dallner, G. (1991). Fatty acid composition of brain phospholipids in aging and in Alzheimer’s disease. Lipids, 26(6), 421–425. https://doi.org/10.1007/BF02536067.
Sud, M., Fahy, E., Cotter, D., Brown, A., Dennis, E. A., Glass, C. K., et al. (2007). LMSD: LIPID MAPS structure database. Nucleic Acids Research, 35(Database), D527–D532. https://doi.org/10.1093/nar/gkl838.
Thomas, M. C., Brownlee, M., Susztak, K., Sharma, K., Jandeleit-Dahm, K. A. M., Zoungas, S., et al. (2015). Diabetic kidney disease. Nature Reviews Disease Primers, 1(1), 15018. https://doi.org/10.1038/nrdp.2015.18.
Tsukui, T., Chen, Z., Fuda, H., Furukawa, T., Oura, K., Sakurai, T., et al. (2019). Novel fluorescence-based method to characterize the antioxidative effects of food metabolites on lipid droplets in cultured hepatocytes. Journal of Agricultural and Food Chemistry, 67(35), 9934–9941. https://doi.org/10.1021/acs.jafc.9b02081
Wu, Y., Chen, Z., Darwish, W. S., Terada, K., Chiba, H., & Hui, S.-P. (2019). Choline and ethanolamine plasmalogens prevent lead-induced cytotoxicity and lipid oxidation in HepG2 cells. Journal of Agricultural and Food Chemistry, 67(27), 7716–7725. https://doi.org/10.1021/acs.jafc.9b02485
Yu, T., Sungelo, M., Goldberg, I., Wang, H., & Eckel, R. (2017). Streptozotocin-treated high fat fed mice: A new type 2 diabetes model used to study canagliflozin-induced alterations in lipids and lipoproteins. Hormone and Metabolic Research, 49(05), 400–406. https://doi.org/10.1055/s-0042-110934
Acknowledgements
This study was supported by (1) Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 19K16531 to Zhen Chen, and (2) National Institutes of Health Grant 1R15HL137130-01A1 to Qiangrong Liang.
Author information
Authors and Affiliations
Contributions
Conceptualization: S-PH; Methodology: ZC; Formal analysis and investigation: ZC, QL, YW, ZG, CL; Writing - original draft preparation: ZC, QL; Writing - review and editing: HC; Funding acquisition: ZC, QL; Resources: SK, JP, CL, FC, YZ; Supervision: HC, S-PH.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the Public Health Service Guide for Care and Use of Laboratory Animals in the New York Institute of Technology College of Osteopathic Medicine.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, Z., Liang, Q., Wu, Y. et al. Comprehensive lipidomic profiling in serum and multiple tissues from a mouse model of diabetes. Metabolomics 16, 115 (2020). https://doi.org/10.1007/s11306-020-01732-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-020-01732-9