Abstract
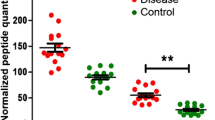
Advanced glycation end products (AGEs) are posttranslational modifications formed non-enzymatically from the reaction of carbohydrates and their degradation products with proteins. Accumulation of AGEs is associated with the progression of severe diabetic complications, for example, and elevated tissue levels of AGEs might even predict these pathologies. As AGE formation is often site-specific, mapping of these modification sites may reveal more sensitive and specific markers than the global tissue level. Here, 42 AGE modifications were identified in a bottom-up proteomic approach by tandem mass spectrometry, which corresponded to 36 sites in 22 high to medium abundant proteins in individual plasma samples obtained from type 2 diabetes mellitus (T2DM) patients with long disease duration (>10 years). Major modifications were glarg (11 modification sites) and carboxymethylation (5) of arginine and formylation (8), acetylation (7), and carboxymethylation (7) of lysine residues. Relative quantification of these sites in plasma samples obtained from normoglycemic individuals (n = 47) and patients with T2DM being newly diagnosed (n = 47) or of medium (2–5 years, n = 20) and long disease duration (>10 years, n = 20) did not reveal any significant differences.



Similar content being viewed by others
References
Ulrich P, Cerami A. Protein glycation, diabetes, and aging. Recent Prog Horm Res. 2001;56:1. Vol 56.
Schalkwijk CG, Stehouwer CDA, van Hinsbergh VWM. Fructose-mediated non-enzymatic glycation: sweet coupling or bad modification. Diabetes Metab Res Rev. 2004;20:369–82.
Wolff SP, Dean RT. Glucose autoxidation and protein modification—the potential role of autoxidative glycosylation in diabetes. Biochem J. 1987;245:243.
Niyatishrikhodaee F, Shibamoto T. Gas-chromatographic analysis of glyoxal and methylglyoxal formed from lipids and related-compounds upon ultraviolet-irradiation. J Agric Food Chem. 1993;41:227–30.
Ray M, Ray S. Aminoacetone oxidase from goat liver—formation of methylglyoxal from aminoacetone. J Biol Chem. 1987;262:5974.
Ahmed MU, Thorpe SR, Baynes JW. Identification of N-epsilon-carboxymethyllysine as a degradation product of fructoselysine in glycated protein. J Biol Chem. 1986;261:4889–94.
Akira K, Hashimoto T. Use of C-13 labeling and NMR spectroscopy for the investigation of degradation pathways of Amadori compounds. Biol Pharm Bull. 2005;28:344.
Smuda M, Henning C, Raghavan CT, Johar K, Vasavada AR, Nagaraj RH, et al. Comprehensive analysis of maillard protein modifications in human lenses: effect of age and cataract. Biochemistry. 2015;54:2500–7.
Schleicher ED, Wagner E, Nerlich AG. Increased accumulation of the glycoxidation product N-epsilon(carboxymethyl)lysine in human tissues in diabetes and aging. J Clin Invest. 1997;99:457–68.
Ahmed N, Thornalley PJ. Advanced glycation endproducts: what is their relevance to diabetic complications? Diabetes Obes Metab. 2007;9:233–45.
Flores-Morales P, Diema C, Vilaseca M, Estelrich J, Luque FJ, Gutierrez-Oliva S, et al. Enhanced reactivity of Lys182 explains the limited efficacy of biogenic amines in preventing the inactivation of glucose-6-phosphate dehydrogenase by methylglyoxal. Bioorg Med Chem. 2011;19:1613–22.
Bron AJ, Sparrow J, Brown NAP, Harding JJ, Blakytny R. The lens in diabetes. Eye. 1993;7:260–75.
Sell DR, Monnier VM. Molecular basis of arterial stiffening: role of glycation—a mini-review. Gerontology. 2012;58:227–37.
Basta G, Schmidt AM, De Caterina R. Advanced glycation end products and vascular inflammation: implications for accelerated atherosclerosis in diabetes. Cardiovasc Res. 2004;63:582.
Mitsuhashi T, Vlassara H, Founds HW, Li YM. Standardizing the immunological measurement of advanced glycation endproducts using normal human serum. J Immunol Methods. 1997;207:79–88.
Horiuchi S, Araki N, Morino Y. Immunochemical approach to characterize advanced glycation end-products of the maillard reaction—evidence for the presence of a common structure. J Biol Chem. 1991;266:7329–32.
Hasenkopf K, Ronner B, Hiller H, Pischetsrieder M. Analysis of glycated and ascorbylated proteins by gas chromatography-mass spectrometry. J Agric Food Chem. 2002;50:5697–703.
Ahmed N, Thornalley PJ. Chromatographic assay of glycation adducts in human serum albumin glycated in vitro by derivatization with 6-aminoquinolyl-N-hydroxysuccinimidyl-carbamate and intrinsic fluorescence. Biochem J. 2002;364:15.
Babaei-Jadidi R, Karachalias N, Ahmed N, Battah S, Thornalley PJ. Prevention of incipient diabetic nephropathy by high-dose thiamine and benfotiamine. Diabetes. 2003;52:2110–20.
Venkatraman J, Aggarwal K, Balaram P. Helical peptide models for protein glycation: proximity effects in catalysis of the Amadori rearrangement. Chem Biol. 2001;8:611.
Frolov A, Blueher M, Hoffmann R. Glycation sites of human plasma proteins are affected to different extents by hyperglycemic conditions in type 2 diabetes mellitus. Anal Bioanal Chem. 2014;406:5755–63.
Zhang Q, Monroe ME, Schepmoes AA, Clauss TRW, Gritsenko MA, Meng D, et al. Comprehensive identification of glycated peptides and their glycation motifs in plasma and erythrocytes of control and diabetic subjects. J Proteome Res. 2011;10:3076.
Greifenhagen U, Frolov A, Hoffmann R. Oxidative degradation of N (epsilon)-fructosylamine-substituted peptides in heated aqueous systems. Amino Acids. 2015;47:1065–76.
Distler L, Georgieva A, Kenkel I, Huppert J, Pischetsrieder M. Structure- and concentration-specific assessment of the physiological reactivity of alpha-dicarbonyl glucose degradation products in peritoneal dialysis fluids. Chem Res Toxicol. 2014;27:1421–30.
Bertoletti L, Regazzoni L, Altomare A, Colombo R, Colzani M, Vistoli G, et al. Advanced glycation end products of beta(2)-microglobulin in uremic patients as determined by high resolution mass spectrometry. J Pharm Biomed Anal. 2014;91:193–201.
Wilmarth PA, Tanner S, Dasari S, Nagalla SR, Riviere MA, Bafna V, et al. Age-related changes in human crystallins determined from comparative analysis of post-translational modifications in young and aged lens: does deamidation contribute to crystallin insolubility? J Proteome Res. 2006;5:2554–66.
Greifenhagen U, Frolov A, Blueher M, Hoffmann R. Plasma proteins modified by advanced glycation endproducts (AGEs) reveal site-specific susceptibilities to glycemic control in patients with type 2 diabetes. J Biol Chem. 2016;291:9610–6.
Gavin JR, Alberti KGMM, Davidson MB, DeFronzo RA, Drash A, Gabbe SG, et al. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2000;23:S4–19.
Kloeting N, Fasshauer M, Dietrich A, Kovacs P, Schoen MR, Kern M, et al. Insulin-sensitive obesity. Am J Physiol Metab. 2010;299:E506–15.
Kannt A, Pfenninger A, Teichert L, Toenjes A, Dietrich A, Schon MR, et al. Association of nicotinamide-N-methyltransferase mRNA expression in human adipose tissue and the plasma concentration of its product, 1-methylnicotinamide, with insulin resistance. Diabetologia. 2015;58:799–808.
Bradford MM. Rapid and sensitive method for quantitation of microgram quantities of protein utilizing principle of protein-dye binding. Anal Biochem. 1976;72:248–54.
Kuzyk MA, Smith D, Yang J, Cross TJ, Jackson AM, Hardie DB, et al. Multiple reaction monitoring-based, multiplexed, absolute quantitation of 45 proteins in human plasma. Mol Cell Proteomics. 2009;8:1860–77.
Farrah T, Deutsch EW, Omenn GS, Campbell DS, Sun Z, Bletz JA, Mallick P, Katz JE, Malmstrom J, Ossola R, Watts JD, Lin BAY, Zhang H, Moritz RL, Aebersold R. A high-confidence human plasma proteome reference set with estimated concentrations in peptide atlas. Mol. Cell Proteomics. 2011;10:M110.006353.
Wolters DA, Washburn MP, Yates JR. An automated multidimensional protein identification technology for shotgun proteomics. Anal Chem. 2001;73:5683–90.
Oberg AL, Vitek O. Statistical design of quantitative mass spectrometry-based proteomic experiments. J Proteome Res. 2009;8:2144–56.
Thornalley PJ, Battah S, Ahmed N, Karachalias N, Agalou S, Babaei-Jadidi R, et al. Quantitative screening of advanced glycation endproducts in cellular and extracellular proteins by tandem mass spectrometry. Biochem J. 2003;375:581.
Malmstroem J, Lee H, Aebersold R. Advances in proteomic workflows for systems biology. Curr Opin Biotechnol. 2007;18:378–84.
Odani H, Iijima K, Nakata M, Miyata S, Kusunoki H, Yasuda Y, et al. Identification of N-omega-carboxymethylarginine, a new advanced glycation endproduct in serum proteins of diabetic patients: possibility of a new marker of aging and diabetes. Biochem Biophys Res Commun. 2001;285:1232–6.
Odani H, Shinzato T, Matsumoto Y, Takai I, Nakai S, Miwa M, et al. First evidence for accumulation of protein-bound and protein-free pyrraline in human uremic plasma by mass spectrometry. Biochem Biophys Res Commun. 1996;224:237–41.
Usui T, Shimohira K, Watanabe H, Hayase F. Detection and determination of glyceraldehyde-derived pyridinium-type advanced glycation end product in streptozotocin-induced diabetic rats. Biosci Biotechnol Biochem. 2007;71:442.
Schmidt R, Boehme D, Singer D, Frolov A. Specific tandem mass spectrometric detection of AGE-modified arginine residues in peptides. J Mass Spectrom. 2015;50:613–24.
Henning C, Smuda M, Girndt M, Ulrich C, Glomb MA. Molecular basis of maillard amide-advanced glycation end product (AGE) formation in vivo. J Biol Chem. 2011;286:44350.
Liyasova MS, Schopfer LM, Lockridge O. Reaction of human albumin with aspirin in vitro: mass spectrometric identification of acetylated lysines 199, 402, 519, and 545. Biochem Pharmacol. 2010;79:784–91.
Finamore F, Priego-Capote F, Nolli S, Zufferey A, Fontana P, Sanchez JC. Characterisation of the influences of aspirin-acetylation and glycation on human plasma proteins. J Proteome. 2015;114:125–35.
Greifenhagen U, Nguyen VD, Moschner J, Giannis A, Frolov A, Hoffmann R. Sensitive and site-specific identification of carboxymethylated and carboxyethylated peptides in tryptic digests of proteins and human plasma. J Proteome Res. 2016.
Korwar AM, Vannuruswamy G, Jagadeeshaprasad MG, Jayaramaiah RH, Bhat S, Regin BS, et al. Development of diagnostic fragment ion library for glycated peptides of human serum albumin: targeted quantification in prediabetic, diabetic, and microalbuminuria plasma by parallel reaction monitoring, SWATH, and MSE. Mol Cell Proteomics. 2015;14:2150–9.
Kimzey MJ, Kinsky OR, Yassine HN, Tsaprailis G, Stump CS, Monks TJ, et al. Site specific modification of the human plasma proteome by methylglyoxal. Toxicol Appl Pharmacol. 2015;289:155–62.
Zhang Q, Tang N, Schepmoes AA, Phillips LS, Smith RD, Metz TO. Proteomic profiling of nonenzymatically glycated proteins in human plasma and erythrocyte membranes. J Proteome Res. 2008;7:2025–32.
Kilhovd BK, Giardino I, Torjesen PA, Birkeland KI, Berg TJ, Thornalley PJ, et al. Increased serum levels of the specific AGE-compound methylglyoxal-derived hydroimidazolone in patients with type 2 diabetes. Metab Exp. 2003;52:163–7.
Teerlink T, Barto R, Ten Brink HJ, Schalkwijk CG. Measurement of N-epsilon-(carboxymethyl)lysine and N-epsilon-(carboxyethyl)lysine in human plasma protein by stable-isotope-dilution tandem mass spectrometry. Clin Chem. 2004;50:1222.
Acknowledgments
The authors thank Sandro Spiller and Dr. Ravi Chand Bollineni for assisting parallel sample processing. Funding from the Deutsche Forschungsgemeinschaft (DFG, grant number HO-2222/7-1 to RH) and a PhD scholarship from the Ernst Schering Foundation to UG are gratefully acknowledged.
Author contributions
Uta Greifenhagen conducted the experiments, data analysis, and interpretation and wrote the manuscript together with Ralf Hoffmann. Andrej Frolov contributed to sample preparation and the experimental design. Matthias Blüher designed the cohorts and provided the plasma samples. Ralf Hoffmann initiated the project, contributed to data interpretation, and wrote the manuscript together with Uta Greifenhagen. All the authors reviewed and agreed upon the final version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the Ethics Committee of Universität Leipzig (approval no: 159-12-21052012) and performed in accordance with the Declaration of Helsinki. All the subjects gave written informed consent before taking part in this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 281 kb)
Rights and permissions
About this article
Cite this article
Greifenhagen, U., Frolov, A., Blüher, M. et al. Site-specific analysis of advanced glycation end products in plasma proteins of type 2 diabetes mellitus patients. Anal Bioanal Chem 408, 5557–5566 (2016). https://doi.org/10.1007/s00216-016-9651-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9651-4