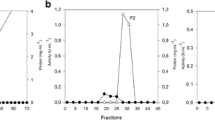
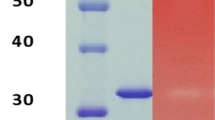
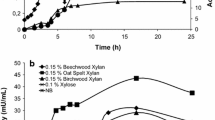
Abstract
Xylanolytic enzymes are involved in xylan hydrolysis, the main ones being endo-β-1,4-xylanases (xylanases). This can be applied in the bioconversion of lignocellulosic materials into value-added products such as xylooligosaccharides (XOS). This study aimed to establish a protocol for the purification of xylanases, as well as to characterize and apply the purified enzyme extract in the production of XOS. The enzyme purification techniques studied were ammonium sulfate ((NH4)2SO4) and ethanol precipitation. Purification of xylanase by fractional precipitation (20–60%) with (NH4)2SO4 was more efficient than with ethanol because the salt precipitation reached a purification factor of 10.27-fold and an enzymatic recovery of 48.6% The purified xylanase exhibited optimum temperature and pH of 50 °C and 4.5, respectively. The Michaelis–Menten constant using beechwood xylan for the enzyme was 74.9 mg/mL. The addition of salts such as CaCl2, ZnCl2, and FeCl3 in the reaction medium increased the xylanase activity. Xylanase showed greater thermal stability (half-life = 169 h) at 45 °C and pH 4.5. Under these conditions and in the presence of Ca2+ (10 mmol/L) the enzyme was even more stable (half-life = 231 h). Total XOS contents (6.7 mg/mL) and the conversion of xylan to XOS (22.3%) between 2 and 24 h were statistically equal. The hydrolysates showed the majority composition of xylobiose, xylotriose, and xylose. The addition of Ca2+ ions did not contribute to an increase in the total XOS content or to a greater conversion of xylan into XOS, but the hydrolysate was richer in xylobiose and had a lower xylose content.
Graphical abstract



Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Amorim C, Silvério SC, Prather KLJ, Rodrigues LR (2019) From lignocellulosic residues to market: production and commercial potential of xylooligosaccharides. Biotechnol Adv 37:107397. https://doi.org/10.1016/j.biotechadv.2019.05.003
An C, Ma SJ, Chang F, Xue WJ (2017) Efficient production of pullulan by Aureobasidium pullulans grown on mixtures of potato starch hydrolysate and sucrose. Braz J Microbiol 48:180–185. https://doi.org/10.1016/j.bjm.2016.11.001
Bailey MJ, Biely P, Poutanen K (1992) Interlaboratory testing of methods for assay of xylanase activity. J Biotechnol 23:257–270. https://doi.org/10.1016/0168-1656(92)90074-J
Bakir U, Yavascaoglu S, Guvenc F, Ersayin A (2001) An endo-β-1,4-xylanase from Rhizopus oryzae: production, partial purification and biochemical characterization. Enzyme Microb Technol 29:328–334. https://doi.org/10.1016/S0141-0229(01)00379-9
Bankeeree W, Lotrakul P, Prasongsuk S, Chaiareekij S, Eveleigh DE, Kim SW, Punnapayak H (2014) Effect of polyols on thermostability of xylanase from a tropical isolate of Aureobasidium pullulans and its application in prebleaching of rice straw pulp. Springerplus 3:1–11. https://doi.org/10.1186/2193-1801-3-37
Bhardwaj N, Kumar B, Agarwal K, Chaturvedi V, Verma P (2019) Purification and characterization of a thermo-acid/alkali stable xylanases from Aspergillus oryzae LC1 and its application in Xylo-oligosaccharides production from lignocellulosic agricultural wastes. Int J Biol Macromol 122:1191–1202. https://doi.org/10.1016/j.ijbiomac.2018.09.070
Bhushan B, Pal A, Kumar S, Jain V (2015) Biochemical characterization and kinetic comparison of encapsulated haze removing acidophilic xylanase with partially purified free xylanase isolated from Aspergillus flavus MTCC 9390. J Food Sci Technol 52:191–200. https://doi.org/10.1007/s13197-013-1013-z
Bibi Z, Ul Qader SA, Aman A, Ur Rehman H, Nawaz MA, Karim A, Us Salam I, Waqas M, Kamran A (2019) Xylan deterioration approach: Purification and catalytic behavior optimization of a novel β-1,4-d-xylanohydrolase from Geobacillus stearothermophilus KIBGE-IB29. Biotechnol Rep 21:e00299. https://doi.org/10.1016/j.btre.2018.e00299
Boonrung S, Katekaew S, Mongkolthanaruk W, Aimi T, Boonlue S (2016) Purification and characterization of low molecular weight extreme alkaline xylanase from the thermophilic fungus Myceliophthora thermophila BF1-7. Mycoscience 57:408–416. https://doi.org/10.1016/j.myc.2016.07.003
Chen Z, Zaky AA, Liu Y, Chen Y, Liu L, Li S, Jia Y (2019) Purification and characterization of a new xylanase with excellent stability from Aspergillus flavus and its application in hydrolyzing pretreated corncobs. Protein Expr Purif 154:91–97. https://doi.org/10.1016/j.pep.2018.10.006
Christov LP, Myburgh J, Van Tonder A, Prior BA (1997) Hydrolysis of extracted and fibre-bound xylan with Aureobasidium pullulans enzymes. J Biotechnol 55:21–29. https://doi.org/10.1016/S0168-1656(97)00048-5
Cortez EV, Pessoa A (1999) Xylanase and β-xylosidase separation by fractional precipitation. Process Biochem 35:277–283. https://doi.org/10.1016/S0032-9592(99)00064-3
Costa MAL, Farinas CS, Miranda EA (2018) Ethanol precipitation as a downstream processing step for concentration of xylanases produced by submerged and solid-state fermentation. Braz J Chem Eng 35:477–488. https://doi.org/10.1590/0104-6632.20180352s20160502
Ding C, Li M, Hu Y (2018) High-activity production of xylanase by Pichia stipitis: purification, characterization, kinetic evaluation and xylooligosaccharides production. Int J Biol Macromol 117:72–77. https://doi.org/10.1016/j.ijbiomac.2018.05.128
Fu LH, Jiang N, Li CX, Luo XM, Zhao S, Feng JX (2019) Purification and characterization of an endo-xylanase from Trichoderma sp., with xylobiose as the main product from xylan hydrolysis. World J Microbiol Biotechnol 35:1–13. https://doi.org/10.1007/s11274-019-2747-1
Gautam A, Kumar A, Dutt D (2017) Production and characterization of cellulase-free xylanase by Aspergillus flavus ARC-12 and its application in pre-bleaching of ethanol-soda pulp of eulaliopsis binata. Res J Biotechnol 12:63–71
Gautério GV, Silva LGG, Hübner T, Rosa Ribeiro T, Kalil SJ (2020) Maximization of xylanase production by Aureobasidium pullulans using a by-product of rice grain milling as xylan source. Biocatal Agric Biotechnol 23:101511. https://doi.org/10.1016/j.bcab.2020.101511
Gautério GV, da Silva LGG, Hübner T, da Rosa Ribeiro T, Kalil SJ (2021) Xylooligosaccharides production by crude and partially purified xylanase from Aureobasidium pullulans: biochemical and thermodynamic properties of the enzymes and their application in xylan hydrolysis. Process Biochem 104:161–170. https://doi.org/10.1016/j.procbio.2021.03.009
Gautério GV, Hübner T, da Rosa Ribeiro T, Ziotti APM, Kalil SJ (2021) Xylooligosaccharide production with low xylose release using crude xylanase from Aureobasidium pullulans: effect of the enzymatic hydrolysis parameters. Appl Biochem Biotechnol. https://doi.org/10.1007/s12010-021-03658-x
Ghose TK (1987) Measurement of cellulase activites. Pure Appl Chem 59:1703–1720
Gírio FM, Fonseca C, Carvalheiro F, Duarte LC, Marques S, Bogel-Łukasik R (2010) Hemicelluloses for fuel ethanol: a review. Biores Technol 101:4775–4800. https://doi.org/10.1016/j.biortech.2010.01.088
Kamble RD, Jadhav AR (2012) Production, purification and characterisation of alkali stable xylanase from Cellulosimicrobium sp. MTCC 10645. Asian Pac J Trop Biomed 2:S1790–S1797. https://doi.org/10.1016/S2221-1691(12)60496-1
Knob A, Carmona EC (2010) Purification and characterization of two extracellular xylanases from Penicillium sclerotiorum: a novel acidophilic xylanase. Appl Biochem Biotechnol 162:429–443. https://doi.org/10.1007/s12010-009-8731-8
Kumar S, Haq I, Prakash J, Singh SK, Mishra S, Raj A (2017) Purification, characterization and thermostability improvement of xylanase from Bacillus amyloliquefaciens and its application in pre-bleaching of kraft pulp. 3 Biotech 7(1):20. https://doi.org/10.1007/s13205-017-0615-y
Leathers TD (2003) Biotechnological production and applications of pullulan. Appl Microbiol Biotechnol 62:468–473. https://doi.org/10.1007/s00253-003-1386-4
Lemes AC, Silvério SC, Rodrigues S, Rodrigues LR (2019) Integrated strategy for purification of esterase from Aureobasidium pullulans. Sep Purif Technol 209:409–418. https://doi.org/10.1016/j.seppur.2018.07.062
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56:658–666. https://doi.org/10.1021/ja01318a036
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/0922-338X(96)89160-4
Luo L, Cai J, Wang C, Lin J, Du X, Zhou A, Xiang M (2016) Purification and characterization of an alkaliphilic endo-xylanase from Streptomyces althioticus LMZM and utilization in the pulp paper industry. J Chem Technol Biotechnol 91:1093–1098. https://doi.org/10.1002/jctb.4690
Ma ZC, Fu WJ, Liu GL, Wang ZP, Chi ZM (2014) High-level pullulan production by Aureobasidium pullulans var. melanogenium P16 isolated from mangrove system. Appl Microbiol Biotechnol 98:4865–4873. https://doi.org/10.1007/s00253-014-5554-5
Mathews SL, Pawlak J, Grunden AM (2015) Bacterial biodegradation and bioconversion of industrial lignocellulosic streams. Appl Microbiol Biotechnol 99:2939–2954. https://doi.org/10.1007/s00253-015-6471-y
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Mizrahi L, Achituv Y (1989) Effect of heavy metals ions on enzyme activity in the mediterranean mussel, Donax trunculus. Bull Environ Contam Toxicol 42:854–859. https://doi.org/10.1007/BF01701626
Ninawe S, Kapoor M, Kuhad RC (2008) Purification and characterization of extracellular xylanase from Streptomyces cyaneus SN32. Biores Technol 99:1252–1258. https://doi.org/10.1016/j.biortech.2007.02.016
Pal A, Khanum F (2011) Purification of xylanase from Aspergillus niger DFR-5: individual and interactive effect of temperature and pH on its stability. Process Biochem 46:879–887. https://doi.org/10.1016/j.procbio.2010.12.009
Prajapati VD, Jani GK, Khanda SM (2013) Pullulan: An exopolysaccharide and its various applications. Carbohydr Polym 95:540–549. https://doi.org/10.1016/j.carbpol.2013.02.082
Raj A, Kumar S, Singh SK, Prakash J (2018) Production and purification of xylanase from alkaliphilic Bacillus licheniformis and its pretreatment of eucalyptus kraft pulp. Biocatal Agric Biotechnol 15:199–209. https://doi.org/10.1016/j.bcab.2018.06.018
Reddy SS, Krishnan C (2016) Production of high-pure xylooligosaccharides from sugarcane bagasse using crude β-xylosidase-free xylanase of Bacillus subtilis KCX006 and their bifidogenic function. LWT Food Sci Technol 65:237–245. https://doi.org/10.1016/j.lwt.2015.08.013
Scopes RK (1993) Protein purification: principles and practice. Springer, New York
Seemakram W, Boonrung S, Aimi T, Ekprasert J, Lumyong S, Boonlue S (2020) Purification, characterization and partial amino acid sequences of thermo-alkali-stable and mercury ion-tolerant xylanase from Thermomyces dupontii KKU–CLD–E2–3. Sci Rep 10:1–10. https://doi.org/10.1038/s41598-020-78670-y
Shahrestani H, Taheri-Kafrani A, Soozanipour A, Tavakoli O (2016) Enzymatic clarification of fruit juices using xylanase immobilized on 1,3,5-triazine-functionalized silica-encapsulated magnetic nanoparticles. Biochem Eng J 109:51–58. https://doi.org/10.1016/j.bej.2015.12.013
Spurway TD, Morland C, Cooper A, Sumner I, Hazlewood GP, O’Donnell AG, Pickersgill RW, Gilbert HJ (1997) Calcium protects a mesophilic xylanase from proteinase inactivation and thermal unfolding. J Biol Chem 272:17523–17530. https://doi.org/10.1074/jbc.272.28.17523
Sugumaran KR, Gowthami E, Swathi B, Elakkiya S, Srivastava SN, Ravikumar R, Gowdhaman D, Ponnusami V (2013) Production of pullulan by Aureobasidium pullulans from Asian palm kernel: a novel substrate. Carbohydr Polym 92:697–703. https://doi.org/10.1016/j.carbpol.2012.09.062
Tan LUL, Mayers P, Saddler JN (1987) Purification and characterization of a thermostable xylanase from a thermophilic fungus Thermoascus aurantiacus. Can J Microbiol 33:689–692. https://doi.org/10.1139/m87-120
Walia A, Guleria S, Mehta P, Chauhan A, Parkash J (2017) Microbial xylanases and their industrial application in pulp and paper biobleaching: a review. 3 Biotech 7:1–12. https://doi.org/10.1007/s13205-016-0584-6
Wang D, Yu X, Gongyuan W (2013) Pullulan production and physiological characteristics of Aureobasidium pullulans under acid stress. Appl Microbiol Biotechnol 97:8069–8077. https://doi.org/10.1007/s00253-013-5094-4
Yan QJ, Wang L, Jiang ZQ, Yang SQ, Zhu HF, Li LT (2008) A xylose-tolerant β-xylosidase from Paecilomyces thermophila: characterization and its co-action with the endogenous xylanase. Biores Technol 99:5402–5410. https://doi.org/10.1016/j.biortech.2007.11.033
Yegin S (2017) Single-step purification and characterization of an extreme halophilic, ethanol tolerant and acidophilic xylanase from Aureobasidium pullulans NRRL Y-2311-1 with application potential in the food industry. Food Chem 221:67–75. https://doi.org/10.1016/j.foodchem.2016.10.003
Zhang F, Hu SN, Chen JJ, Lin LB, Wei YL, Tang SK, Xu LH, Li WJ (2012) Purification and partial characterisation of a thermostable xylanase from salt-tolerant Thermobifida halotolerans YIM 90462 T. Process Biochem 47:225–228. https://doi.org/10.1016/j.procbio.2011.10.032
Acknowledgements
The authors are grateful to “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)” and “Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq)” for the financial support.
Funding
This study was financed in part by the “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)”—Brazil—Finance Code 001, and by the “Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq)”—Brazil—Finance Codes 423285/2018-1 and 304857/2018-1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Corrêa Junior, L.C.S., Gautério, G.V., de Medeiros Burkert, J.F. et al. Purification of xylanases from Aureobasidium pullulans CCT 1261 and its application in the production of xylooligosaccharides. World J Microbiol Biotechnol 38, 52 (2022). https://doi.org/10.1007/s11274-022-03240-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-022-03240-5