Abstract
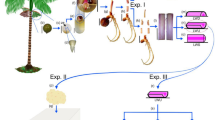
Euterpe edulis Martius is an endangered palm tree native to the Atlantic Forest. As it propagates only with seeds and does not tiller, the tree dies after its highly appreciated palm heart is harvested. In this study, we analyzed the embryogenic response of E. edulis with respect to maturity of the explant and concentration of picloram, an auxin mimetic. Immature fruits were harvested, and their seeds were extracted and germinated in vitro. After 2, 4, 6, and 8 months, the aerial parts of normal seedlings were excised and stem segments were used to induce somatic embryogenesis in the presence of 100, 125, 150, 175, and 200 µM picloram. The number of proembryos, induction rate, explant mass, oxidation, global DNA methylation, 1-aminocyclopropane-1-carboxylic acid, proline, polyamines, and ultrastructural analysis of cells were assessed. Six-month-old seedling explants achieved the highest number of proembryos and embryogenic induction rate at most picloram concentrations, as well as the highest 1-aminocyclopropane-1-carboxylic acid content, but lowest spermine and putrescine levels. Explants with somatic embryos exhibited lower DNA methylation levels than non-embryogenic calli. Proline content was highest in stem segments of younger seedlings (2-month-old). Despite being asynchronous, maturation with 5 µM abscisic acid was achieved. Therefore, 6-month-old E. edulis seedlings supplemented with 200 µM picloram could be used for ex situ conservation of this endangered species.
Key message
Six-month-old seedlings of the palm Euterpe edulis are the best source of explants for in vitro germination of this endangered species and, therefore, may contribute to its conservation.









Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Amaral-Silva PM, Clarindo WR, Guilhen JHS, Passos ABRJ, Sanglard NA, Ferreira A (2021) Global 5-methylcytosine and physiological changes are triggers of indirect somatic embryogenesis in Coffea canephora. Protoplasma 258:45–57. https://doi.org/10.1007/s00709-020-01551-8
Borges GSC, Gonzaga LV, Jardini FA, Mancini Filho J, Hellerm MG, Costa ACO, Fett R (2013) Protective effect of Euterpe edulis M. on Vero cell culture and antioxidant evaluation based on phenolic composition using HPLC− ESI-MS/MS. Food Rese Int 51(1):363–369. https://doi.org/10.1016/j.foodres.2012.12.035
Chakrabarty D, Yuk KW, Paek KY (2003) Detection of DNA methylation changes during somatic embryogenesis of Siberian ginseng (Eleuterococcus senticosus). Plant Sci 165(1):61–68. https://doi.org/10.1016/S0168-9452(03)00127-4
Corredoira E, Cano V, Bárány I, Solís MT, Rodríguez H, Vieitez AM, Risueño MC, Testillano PS (2017) Initiation of leaf somatic embryogenesis involves high pectin esterification, auxin accumulation and DNA demethylation in Quercus alba. J Plant Physiol 213:42–54. https://doi.org/10.1016/j.jplph.2017.02.012
Corredoira E, Merkle SA, Martínez MT, Toribio M, Canhoto JM, Correia SI, Ballester A, Vieitez AM (2019) Non-zygotic embryogenesis in hardwood species. Crit Rev Plant Sci 38(1):29–97. https://doi.org/10.1080/07352689.2018.1551122
De-La-Peña C, Nic-Can GI, Galaz-Ávalos RM, Avilezmontalvo R, Loyola- Argas VM (2015) The role of chromatin modifications in somatic embryogenesis in plants. Front Plant Sci 6:635. https://doi.org/10.3389/fpls.2015.00635
Dodeman VL, Ducreux G, Kreis M (1997) Zygotic embryogenesis versus somatic embryogenesis. J Exp Bot 48:1493–1509
Fehér A (2005) Why somatic plant cells start to form embryos? In: Mujib A, Šamaj J (eds) Somatic embryogenesis. Springer, Berlin, pp 85–101. https://doi.org/10.1007/7089_019
Ferreira JCB, Silva-Cardoso IMA, Meira RO, Scherwinski-Pereira JE (2022) Somatic embryogenesis and plant regeneration from zygotic embryos of the palm tree Euterpe precatoria Mart. Plant Cell, Tissue Organ Cult 148:667–686. https://doi.org/10.1007/s11240-022-02227-2
Forcat S, Bennett MH, Mansfield JW, Grant MR (2008) A rapid and robust method for simultaneously measuring changes in the phytohormones ABA, JA and SA in plants following biotic and abiotic stress. Plant Methods 4(1):16. https://doi.org/10.1186/1746-4811-4-16
Freitas EO, Monteiro TR, Nogueira GF, Scherwinski-Pereira JE (2016) Somatic embryogenesis from immature and mature zygotic embryos of the açai palm (Euterpe oleracea): Induction of embryogenic cultures, morphoanatomy and its morphological characteristics. Sci Hort 212(14):126–135. https://doi.org/10.1016/j.scienta.2016.09.044
Garcia JAA, Corrêa RCG, Barros L, Pereira C, Abreu RMV, Alves MJ, Calhelha RC, Bracht A, Peralta RM, Ferreira ICFR (2019) Chemical composition and biological activities of Juçara (Euterpe edulis Martius) fruit by-products, a promising underexploited source of high-added value compounds. J Func Foods 55:325–332. https://doi.org/10.1016/j.jff.2019.02.037
Ghosh A, Igamberdiev AU, Debnath SC (2021) Tissue culture-induced DNA methylation in crop plants: a review. Mol Biol Rep 48:823–841. https://doi.org/10.1007/s11033-020-06062-6
Guerra MP, Handro W (1988) Somatic embryogenesis and plant regeneration in embryo cultures of Euterpe edulis Mart. (Palmae). Plant Cell Rep 7(7):550–552. https://doi.org/10.1007/BF00272754
Guerra MP, Handro W (1998) Somatic embryogenesis and plant regeneration in different organs of Euterpe edulis Mart. (Palmae): Control and structural features. J Plant Res 111(1):65–71. https://doi.org/10.1007/BF02507151
Gutmann M, Von Aderkas P, Label P, Lelu M-A (1996) Effects of abscisic acid on somatic embryo maturation of hybrid larch. J Exp Bot 47:1905–1917. https://doi.org/10.1093/jxb/47.12.1905
Hildebrandt TM, Nesi AN, Araújo WL, Braun HP (2015) Amino acid catabolism in plants. Mol Plant 8(11):1563–1579. https://doi.org/10.1016/j.molp.2015.09.005
Huang XL, Li XJ, Li Y, Huang LZ (2008) The effect of AOA on ethylene and polyamine metabolism during early phases of somatic embryogenesis in Medicago sativa. Physiol Plant 113(3):424–429. https://doi.org/10.1034/j.1399-3054.2001.1130317.x
Ikeuchi M, Ogawa Y, Iwase A, Sugimoto K (2016) Plant regeneration: cellular origins and molecular mechanisms. Development 143:1442–1451. https://doi.org/10.1242/dev.134668
Isah T (2016) Induction of somatic embryogenesis in woody plants. Acta Physiol Plant 38(118):1–22. https://doi.org/10.1007/s11738-016-2134-6
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron-microscopy. J Cell Biol 27:137–138A
Kępczyńska E, Ruduś I, Kępczyński J (2009) Endogenous ethylene in indirect somatic embryogenesis of Medicago sativa L. Plant Growth Regul 59(1):63–73. https://doi.org/10.1007/s10725-009-9388-6
Ledo AS, Lameira AO, Benbadis AK, Menezes IC, Oliveira MSP, Medeiros Filho S (2002) Somatic embryogenesis from zygotic embryos of Euterpe oleracea Mart. Rev Bras Frut 24(3):601–603. https://doi.org/10.1590/S0100-29452002000300004
Mello T, Rosa TLM, Simoes IM, Lima PAM, Anjos BB, Araujo CP, Hegedus CEN, Santos HO, Otoni WC, Alexandre RS, Lopes JC (2022) Reserve mobilization and in vitro germination of Euterpe edulis (Martius) seeds at different maturation stages. Trees 36:415–426. https://doi.org/10.1007/s00468-021-02216-6
Mello T, Correia LNF, Hegedus CEN, Schmildt ER, Ferreira A, Lopes JC, Otoni WC, Alexandre RS (2023) Cell reprogramming via direct somatic embryogenesis in an Atlantic Forest species vulnerable to extinction: Euterpe edulis stem segments induced with picloram. Plant Cell Tissue Organ Cult 154:131–140. https://doi.org/10.1007/s11240-023-02521-7
Mello T, Silva TD, Zanardo TÉC, Almeida FAN, Oliveira LB, Hegedus CEN, Anjos BB, Schmildt ER, Ferreira A, Ferreira MFS, Lopes JC, Aquije GMFV, Otoni WC, Alexandre RS (2024a) Somatic embryogenesis in Euterpe edulis Martius is improved by wounding, explant orientation, and suspension culture. Plant Cell Tissue Organ Cult 156:31. https://doi.org/10.1007/s11240-023-02649-6
Mello T, Taliuli YS, Silva TD, Zanardo TÉC, Hegedus CEN, Anjos BB, Schmildt ER, Ferreira A, Lourenço MP, Pinheiro PF, Aquije GMFV, Lopes JC, Otoni WC, Alexandre RS (2024b) Optimization of somatic embryogenesis in Euterpe edulis Martius using auxin analogs and atomic force microscopy. Sci Hort 331:113164. https://doi.org/10.1016/j.scienta.2024.113164
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nandy S, Das T, Tudu CK, Mishra T, Ghorai M, Gadekar VS, Anand U, Kumar M, Behl T, Shaikh NK, Jha NK, Shekhawat MS, Pandey DK, Dwivedi PR, Dey A (2022) Unravelling the multi-faceted regulatory role of polyamines in plant biotechnology, transgenics and secondary metabolomics. Appl Microbiol Biotechnol 106:905–929. https://doi.org/10.1007/s00253-021-11748-3
O’Brien TP, Mccully ME (1981) The study of plant structure principles and select methods. Termarcarphi Pty, Melbourne, p 45
Oliveira LB, Mello T, Araujo CP, Oliveira JPB, Ferreira A, Zanardo EC, Vieira LM, Otoni WC, Alexandre RS, Carvalho VS (2022) Morphoanotomical aspects of auxinic herbicides-induced somatic embryogenesis in Euterpe edulis Martius - a symbol and threatened species of the Atlantic Forest. Sci Hort 299:111051. https://doi.org/10.1016/j.scienta.2022.111051
R Core Team (2024) R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing. Available in: <https://www.rproject.org/>. Accessed on: 13 Jan. 2024.
Rakesh B, Sudheer WN, Nagella P (2021) Role of polyamines in plant tissue culture: An overview. Plant Cell Tiss Organ Cult 145:487–506. https://doi.org/10.1007/s11240-021-02029-y
Rocha DI, Pinto DLP, Vieira LM, Tanaka FAO, Dornelas MC, Otoni WC (2016) Cellular and molecular changes associated with competence acquisition during passion fruit somatic embryogenesis: ultrastructural characterization and analysis of SERK gene expression. Protoplasma 253(2):595–609. https://doi.org/10.1007/s00709-015-0837-y
Rodriguez-Sanz H, Manzanera JA, Solis MT, Gomez-Garay A, Pintos B, Risueno MC, Testillano PS (2014) Early markers are present in both embryogenesis pathways from microspores and immature zygotic embryos in cork oak. Quercus Suber l BMC Plant Biol 14(224):1–18. https://doi.org/10.1186/s12870-014-0224-4
Scherwinski-Pereira JE, Guedes RS, Silva RA, Fermino JR, Luis ZG, Freitas EO (2012) Somatic embryogenesis and plant regeneration in açaí palm (Euterpe oleracea). Plant Cell, Tissue Organ Cult 109(3):501–508. https://doi.org/10.1007/s11240-012-0115-z
Schulz M, Borges GDSC, Gonzaga LV, Costa ACO, Fett R (2016) Juçara fruit (Euterpe edulis Mart.): Sustainable exploitation of a source of bioactive compounds. Food Res Int 89:14–26. https://doi.org/10.1016/j.foodres.2016.07.027
Silva-Cardoso IMA, Meira FS, Gomes ACMM, Scherwinski-Pereira JE (2020) Histology, histochemistry and ultrastructure of pre-embryogenic cells determined for direct somatic embryogenesis in the palm tree Syagrus oleracea. Physiol Plant 168(4):845–875. https://doi.org/10.1111/ppl.13026
Silva-Cardoso IMA, Gomes ACMM, Scherwinski-Pereira JE (2022) Cellular responses of oil palm genotypes during somatic embryogenesis involve participation of procambial cells, DNA demethylation, and auxin accumulation. Plant Cell Rep 41:1875–1893. https://doi.org/10.1007/s00299-022-02898-3
Stein VC, Paiva R, Vargas DP, Soares FP, Alves E, Nogueira GF (2010) Ultrastructural calli analysis of Inga vera Willd subsp. affinis (DC) T.D. Penn Rev Árv 34(5):789–796. https://doi.org/10.1590/S0100-67622010000500004
Toumi I, Moschou PN, Paschalidis KA, Bouamama B, Salem-Fnayou AB, Ghorbel AW, Mliki A, Roubelakis-Angelakis KA (2010) Abscisic acid signals reorientation of polyamine metabolism to orchestrate stress responses via the polyamine exodus pathway in grapevine. J Plant Physiol 167:519–525. https://doi.org/10.1016/j.jplph.2009.10.022
Valledor L, Hasbún R, Meijón M, Rodríguez JL, Santamaría E, Viejo M, Berdasco M, Feito I, Fraga MF, Cañal MJ, Rodríguez R (2007) Involvement of DNA methylation in tree development and micropropagation. Plant Cell, Tissue Organ Cult 91:75–86. https://doi.org/10.1007/s11240-007-9262-z
Acknowledgements
The authors would like to thank Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa e Inovação do Espírito Santo (FAPES) for research funding and Editage (www.editage.com) for English language editing. In addition, the authors are grateful to the Carlos Alberto Redins Cellular Ultrastructure Multiuser Laboratory (LUCCAR) on behalf of Prof. Dr. Breno Valentim Nogueira.
Funding
This study was funded by the Fundação de Amparo à Pesquisa e Inovação do Espírito Santo (FAPES) (EDITAL FAPES/CNPq N° 05/2017—PRONEM—PROGRAMA DE APOIO A NÚCLEOS EMERGENTES). The agreement was registered with SICONV under n° 794009/2013, Process FAPES n° 72660945, and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Finance code 308365/2019–4).
Author information
Authors and Affiliations
Contributions
Tamyris de Mello: Investigation, Methodology, Writing – original draft, Writing – review and editing. Tadeu Ériton Caliman Zanardo, Tatiane Dulcineia Silva, Joana Silva Costa, Débora Pellanda Fagundes, Caroline Palacio de Araujo, and Clovis Eduardo Nunes Hegedus: Investigation. Breno Benvindo do Anjos, Edilson Romais Schmildt, and Adésio Ferreira: Formal analysis. Marcia Flores da Silva Ferreira, José Carlos Lopes, and Wagner Campos Otoni: Supervision, Writing – review and editing. Rodrigo Sobreira Alexandre: Conceptualization, Supervision, Project administration, Funding acquisition, Writing – review and editing.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interests to disclose. Co-author Wagner Campos Otoni is Associate Editor of the journal, but this did not influence the arbitration or decision-making process when evaluating the submission.
Additional information
Communicated by Vijay Kumar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Mello, T., Zanardo, T.É.C., Silva, T.D. et al. Chronological age, changes in DNA methylation, and endogenous hormone levels of explants promote somatic embryogenesis of Euterpe edulis Martius. Plant Cell Tiss Organ Cult 157, 38 (2024). https://doi.org/10.1007/s11240-024-02757-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11240-024-02757-x