Abstract
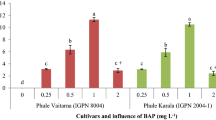
An improved organogenesis protocol for multiplication of sweet orange cv. Mosambi has been standardized by the use of different PGRs, basal media and ethylene adsorbents. Incorporation of cytokinins, BAP (8.8 µM) and kinetin (6.97 µM) resulted in best shoot organogenesis with the highest response (81.40%), no. of micro-shoots/explant (2.06), mean micro-shoot length (1.30 cm) and no. of leaves/micro-shoot (3.55) owing to complementary effect of these factors. However, the regenerated micro-shoots failed to establish due to 100% leaf abscission of micro-shoot. To retard the effect of ethylene accumulation on the regenerated micro-shoots, two types of ethylene adsorbents carrying silver ions, namely, AgNO3 and Ag2S2O3 and two gelling agents (agar–agar and Phytagel™) were tested at different concentrations. Addition of AgNO3 (5.88 µM) to the medium containing Phytagel™ along with cytokinin (BAP 8.8 µM and kinetin 6.97 µM) led to significant reduction in shoot abscission rate (4.20), while 17.66 µM AgNO3 supplementation improved no. of micro-shoots/explants (2.19) and micro-shoot length (3.36 cm) whereas Ag2S2O3 at 20 µM enhanced the total chlorophyll content (3.47 mg g−1 FW) three times as compared to control. Similarly, among the tested basal media, MS basal medium induced best response on shoot organogenesis. Rooting of micro-shoots was highest (81.12%) with the supplementation of NAA (5.37 µM), which also affected no. of roots/explant (4.52) and mean root length (5.26 cm). The supplementation of ethylene adsorbents during in vitro micro-shoot multiplication significantly improved their quality, which provides ideal rooting for development of complete plantlets in sweet orange cv. Mosambi.
Key message
Ethylene accumulation in glass containers inhibited micro-shoot establishment of Citrus sinensis L. Use of silver ions enhanced culture establishment and quality of the micro-shoot.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- BAP:
-
6-Benzylaminopurine
- NAA:
-
1-Napthalene acetic acid
- IBA:
-
Indole-3-butyric acid
- SFI:
-
Shoot forming index
- STS:
-
Silver thiosulfate
- DMSO:
-
Dimethyl sulfoxide
- AC:
-
Activated charcoal
- CRD:
-
Completely randomized design
- LSD:
-
Least significant difference
- MS:
-
Murashige and Skoog medium
- MT:
-
Murashige and Tucker medium
- DKW:
-
Driver and Kuniyuki medium
- WPM:
-
Lloyd and Mc Cown medium
- B5:
-
Gamborg B5 medium
References
Al-Bahrany AM (2002) Effect of phytohormones on in vitro shoot multiplication and rooting of lime Citrus aurantifolia (Christm.) Swing. Sci Hortic 95(4):285–295. https://doi.org/10.1016/S0304-4238
Cervera M, Navarro A, Navarro L, Peña L (2008) Production of transgenic adult plants from clementine mandarin by enhancing cell competence for transformation and regeneration. Tree Physiol 28:55–66. https://doi.org/10.1093/treephys/28.1.55
Driver JA, Kuniyuki AH (1984) In vitro propagation of Paradox walnut rootstock. Hort Sci 19:507–509
Duran Villa N, Ortega V, Navarro L (1989) Morphogenesis and tissue culture of three different citrus species. Plant Cell Tissue Organ Cult 16(2):123–133
Epstein E, Ludwig-Muller J (1993) Indole 3-butyric acid in plants: occurance, synthesis, metabolism and transport. Physiol Plant 88:382–289. https://doi.org/10.1111/j.1399-3054.1993.tb05513
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228. https://doi.org/10.1023/A:1024033216561
Gamborg OL, Miller R, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exper Cell Res 50:151–158. https://doi.org/10.1016/0014-4827(68)90403-5
George EF, Hall MA, Klerk GJD (2008) Plant propagation by tissue culture, 3rd edn. Springer, Dordrecht
Heyl A, Schmulling T (2003) Cytokinin signal perception and transduction. Curr Opin Plant Biol 6:480–488
Hiscox JD, Israelstom GF (1979) A method for the extraction of chlorophyll from leaf tissues without maceration. Can J Bot 57:1332–1334. https://doi.org/10.1139/b79-163.https://apps.fas.usda.gov/psdonline/circulars/citrus.pdf
Hu WW, Gong H, Pua EC (2006) Modulation of SAMDC expression in Arabidopsis thaliana alters in vitro shoot organogenesis. Physiol Plant 128(4):740–750. https://doi.org/10.1111/j.1399-3054.2006.00799.x
Jan A, Bhat KM, Bhat SJA, Mir MA, Bhat MA, Imtiyaz A, Rather JA (2013) Surface sterilization method for reducing microbial contamination of field grown strawberry explants intended for in vitro culture. Afr J Biotechnol 12(39):1
Kotsias D, Roussos PA (2001) An investigation on the effect of different plant growth regulating compounds in in vitro shoot tip and node culture of lemon seedlings. Sci Hortic 89:115–128. https://doi.org/10.1016/S0304-4238(00)00227-2
Kumar PP, Lakshmanan P, Thorpe TA (1998) Regulation of morphogenesis in plant tissue culture by ethylene. In Vitro Cell Dev Biol Plant 34(2):94–103. https://doi.org/10.1007/BF02822771
Lei C, Wang H, Liu B, Ye H (2014) Effects of silver nitrate on shoot regeneration of Artemisia annua L. Plant Biotechnol. https://doi.org/10.5511/plantbiotechnology.13.1109a
Lemos EEP, Blake J (1996) Control of leaf abscission in nodal cultures of Annona squamosa L. J Hortic Sci 71(5):721–728. https://doi.org/10.1080/14620316.1996.11515452
Lloyd G, Cown Mc (1980) Commercially feasible micropropagation of mountain laurel Kalmia latifolia, by use of shoot tip culture. Proc Int Comb Plant Prop Soc 30:421–427
Mahmoud LM, Grosser JW, Dutt M (2020). Silver compounds regulate leaf drop and improve in vitro regeneration from mature tissues of Australian finger lime (Citrus australasica). Plant Cell Tissue Organ Cult 141(3):455–464. https://doi.org/10.1007/s11240-020-01803-8
Marutani-Hert M, Evens TJ, Mc Collum GT, Niedz RP (2011) Bud emergence and shoot growth from mature citrus nodal stem segments. Plant Cell Tissue Organ Cult 106(1):81–91. https://doi.org/10.1007/s11240-010-9896-0
Murashige T, Tucker DPH (1969) Growth factor requirements of citrus tissue culture. Proc 1st Int Citrus Symp 3:1155
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Naik SK, Chand NK (2003) Silver nitrate and amino-ethoxy-vinylglycine promote in vitro adventitious shoot regeneration of pomegranate (Punica granatum L.). J Plant Physiol 160:423–430. https://doi.org/10.1078/0176-1617-00949
Navarro-García N, Morte A, Pérez-Tornero O (2016b) In vitro adventitious organogenesis and histological characterization from mature nodal explants of Citrus limon. In Vitro Cell Dev Biol Plant 52(2):161–173. https://doi.org/10.1007/s11627-015-9743-4
Navarro-García N, Martínez-Romero D, Pérez-Tornero O (2016a) Assessment of the impact of ethylene and ethylene modulators in Citrus limon organogenesis. Plant Cell Tissue Organ Cult 127:405–415. https://doi.org/10.1007/s11240-016-1062-x
Nwe YY, Myint KT, Mochizuki Y, Vazirzanjani M, Okayasu K, Suzuki S, Ogiwara I (2014) In vitro regeneration through direct shoot organogenesis in Honey Orange (Citrus tangerina). Plant Biotechnol J 31(4):341–344. https://doi.org/10.5511/plantbiotechnology.14.0929a
Oliveira MLP, Costa MGC, Silva CV, Otoni WC (2010) Growth regulators, culture media and antibiotics in the in vitro shoot regeneration from mature tissue of citrus cultivars. Pesqui Agropecu Bras 45:654–660. https://doi.org/10.1590/S0100-204X2010000700004
Rashad M, Khan MM, Ranzan R, Adnan S, Khan FA (2005) In vitro regeneration and somatic embryogenesis in Citrus aurantifolia and Citrus sinensis. Int J Agric Biol 7:518–520
Rattanpal HS, Kaur G, Gupta M (2011) In vitro plant regeneration in rough lemon (Citrus jambhiri Lush) by direct organogenesis. Afr J Biotechnol 10(63):13724–13728. https://doi.org/10.5897/AJB09.1264
Sirisom Y, Te-Chato S (2012) The effect of peptone and silver nitrate on in vitro shoot formation in Hevea brasiliensis Muell Arg. J Agric Technol 8:1509–1516. http://www.ijat-aatsea.com/pdf/v8_n4_12_July/31_IJAT_2012_8_4__YupapornSirisom-PlantScience-accepted.pdf
Sridevi V, Giridhar P, Simmi PS, Ravishankar GA (2010) Direct shoot organogenesis on hypocotyl explants with collar region from in vitro seedlings of Coffeacanephora Pierre ex. Frohner cv. CxR and Agrobacterium tumefaciens-mediated transformation. Plant Cell Tissue Organ Cult 101:339–347. https://doi.org/10.1007/s11240-010-9694-8
Sujata U, Syamal MM (2010) Micropropagation of sweet orange (Citrus sinensis L. Osbeck) cv Mosambi through nodal and internodal segments. Ecol Environ Conserv 28(1B):672–677
Tallón CI, Porras I, Pérez-Tornero O (2013) High efficiency in vitro organogenesis from mature tissue explants of Citrus macrophylla and Citrus aurantium. In Vitro Cell Dev Biol Plant 49:145–155. https://doi.org/10.1007/s11627-012-9476-6
Titov S, Bhowmik SK, Mandal A, Alam MS, Uddin SN (2006) Control of phenolic compound secretion and effect of growth regulators for organ formation from Musa spp. cv. Kanthali floral bud explants. Am J Biochem Biotechnol 2(3):97–104
Veen H, Van de Geijn SC (1978) Mobility and ionic form of silver as related to longevity of cut carnations. Planta 140(1):93–96. https://doi.org/10.1007/BF00389386
Zhang P, Phansiri S, Puonti-Kaerlas J (2001) Improvement of cassava shoot organogenesis by the use of silver nitrate in vitro. Plant Cell Tissue Organ Cult 67:47–54. https://doi.org/10.1023/A:1011654128198
Acknowledgements
The main author sincerely acknowledges the ICAR-Indian Agricultural Research Institute, New Delhi for providing the facility for carrying out the research and Department of Science and Technology, Ministry of Science and Technology (DST) Government of India for the grand of DST-INSPIRE fellowship.
Author information
Authors and Affiliations
Contributions
Research work was conceptualized by Dr. OPA. Data collection, statistical analysis and the original draft was prepared by the main author. The draft was corrected by Dr. SKS and Dr. KK.
Corresponding author
Ethics declarations
Conflict of interest
Authors have no conflict of interest. The research was funded by DST, Govt. of India.
Additional information
Communicated by Maria Antonietta Germanà.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Prusty, R., Awasthi, O.P., Singh, S.K. et al. In vitro shoot organogenesis in sweet orange (Citrus sinensis L.) cv. Mosambi and the effect of ethylene adsorbents on micro-shoot quality. Plant Cell Tiss Organ Cult 153, 625–637 (2023). https://doi.org/10.1007/s11240-023-02499-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02499-2