Abstract
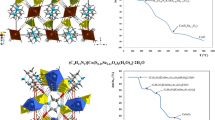
Purple crystals of ethylenediammonium cobalt bis(hydrogen phosphite) dichloride were produced from an aqueous solution containing ethylenediamine, cobalt chloride, and phosphorous acid. The new compound is monoclinic, P21/c, a = 8.6665(3) Å, b = 7.2866(2) Å, c = 9.7300(3) Å, β = 112.726(3); V = 566.74(3) Å3, Z = 2. The 2D structure comprised ethylenediammonium cations sandwiched between the [Co(HPHO3)2Cl2]2− layers. The latter are built of trans-CoO4Cl2 octahedra linked by hydrogen-bonded dimers of hydrophosphite ions, (HPHO3)22−. The new compound is a complete structural analog of the recently reported (C2N2H10)[Co(HSeO3)2Cl2] and isostructural analogs with other transition metal dications; it is, in fact, the first representative of a new “layered hydrophosphite” family. Its structure provides yet another illustration of essential similarities in the crystal chemistry of selenites and phosphites, including protonated species, wherein the lone pair of SeIV and the nearly nonpolar P–H enhance formation of loose and/or open-framework structural architectures.




Similar content being viewed by others
Availability of data and materials
Not applicable.
References
Yan M, Xue H-G, Guo S-P (2021) Recent achievements in lone-pair cation-based infrared second-order nonlinear optical materials. Cryst Growth Des 21:698–720. https://doi.org/10.1021/acs.cgd.0c01407
Choudhury A, Kumar U, Rao CNR (2002) Three-dimensional organically templated open-framework transition metal selenites. Angew Chemie Int Ed 41:158–161. https://doi.org/10.1002/1521-3773(20020104)41:1%3c158::AID-ANIE158%3e3.0.CO;2-%23
Boldt K, Engelen B, Panthöfer M, Unterderweide K (2000) Stereochemical equivalence of PIII-bonded hydrogen atoms and SeIV lone electron pairs in Sr(H2PO3)2 and Sr(HSeO3)2. Eur J Inorg Chem 2000:2071–2075. https://doi.org/10.1002/1099-0682(200009)2000:9%3c2071::AID-EJIC2071%3e3.0.CO;2-O
Kovrugin VM, Gordon EE, Kasapbasi EE et al (2016) Bonding scheme, hydride character, and magnetic paths of (HPO3)2− versus (SeO3)2− building units in solids. J Phys Chem C 120:1650–1656. https://doi.org/10.1021/acs.jpcc.5b10889
Němec I, Matulková I, Krumbe W et al (2021) Linear and nonlinear optical properties, pyroelectricity and vibrational spectroscopy of polar guanidinium hydrogen phosphite, GuH2PO3, and hydrogen selenite, GuHSeO3. Opt Mater 111:110722. https://doi.org/10.1016/j.optmat.2020.110722
Charkin DO, Grishaev VY, Borisov AS et al (2023) A nonpolar bond to hydrogen vs. lone pair: incorporation of HPO32− and SeEO32− into a lead perrhenate framework. J Solid State Chem 318:123706. https://doi.org/10.1016/j.jssc.2022.123706
Grishaev VY, Siidra OI, Markovski MR et al (2023) Synthesis and crystal structure of two novel polymorphs of (NaCl)[Cu(HSeO3)2]: a further contribution to the family of layered copper hydrogen selenites. Zeitschrift für Krist - Cryst Mater 238:177–185. https://doi.org/10.1515/zkri-2023-0004
Wagener M (2005) Synthese, Charakterisierung und struktur-chemische Aspekte von Kupfer- und Silberchalkogenohalogeniden sowie von Halogeno- und Oxochalkogenaten(IV). Uinversität Siegen 300
Pasha I, Choudhury A, Rao CNR (2003) An organically templated open-framework cadmium selenite. Solid State Sci 5:257–262. https://doi.org/10.1016/S1293-2558(02)00100-0
Feng M-L, Prosvirin AV, Mao J-G, Dunbar KR (2006) Syntheses, structural studies, and magnetic properties of divalent cu and co selenites with organic constituents. Chem - A Eur J 12:8312–8323. https://doi.org/10.1002/chem.200600031
Smith PA, Aksenov SM, Jablonski S, Burns PC (2018) Structural unit charge density and molecular cation templating effects on orientational geometric isomerism and interlayer spacing in 2-D uranyl sulfates. J Solid State Chem 266:286–296. https://doi.org/10.1016/j.jssc.2018.07.028
Lafront AM, Trombe JC (1995) ‘Layered hydrogenselenite’ I. Synthesis, structure redetermination of [Cu(HSeO3)2(H2O)2] and determination of [Cu(HSeO3)2(NO3)2]2−·2NH4+, NH4NO3. Structural relationships of these complexes with [Cu(HSeO3)2]. Inorganica Chim Acta 234:19–25. https://doi.org/10.1016/0020-1693(95)04500-9
Oxford Diffraction (2009) CrysAlisPro. Oxford Diffraction Ltd, Abingdon, Oxfordshire, UK
Palatinus L, Chapuis G (2007) Superflip — a computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J Appl Crystallogr 40:786–790. https://doi.org/10.1107/S0021889807029238
Petříček V, Palatinus L, Plášil J, Dušek M (2023) Jana 2020 — a new version of the crystallographic computing system Jana. Zeitschrift für Krist - Cryst Mater. https://doi.org/10.1515/zkri-2023-0005
Prince E (2006) International tables for crystallography. International Union of Crystallography, Chester, England
Brandenburg K, Putz H (2005) DIAMOND, Version 3. Crystal Impact GbR. Bonn, Germany
le Bail A, Duroy H, Fourquet JL (1988) Ab-initio structure determination of LiSbWO6 by X-ray powder diffraction. Mat Res Bull 23:447–452. https://doi.org/10.1016/0025-5408(88)90019-0
Hamdi N, Chaouch S, da Silva I et al (2022) Synthesis, structural characterization, and biological activities of organically templated cobalt phosphite (H2DAB)[Co(H2PO3)4]·2H2O. Sci 4:5. https://doi.org/10.3390/sci4010005
Mackenzie CF, Spackman PR, Jayatilaka D, Spackman MA (2017) CrystalExplorer model energies and energy frameworks: extension to metal coordination compounds, organic salts, solvates and open-shell systems. IUCrJ 4:575–587. https://doi.org/10.1107/S205225251700848X
Blatov VA, Shevchenko AP, Proserpio DM (2014) Applied topological analysis of crystal structures with the program package ToposPro. Cryst Growth Des 14:3576–3586. https://doi.org/10.1021/cg500498k
Shevchenko AP, Shabalin AA, Karpukhin IY, Blatov VA (2022) Topological representations of crystal structures: generation, analysis and implementation in the TopCryst system. Sci Technol Adv Mater Methods 2:250–265. https://doi.org/10.1080/27660400.2022.2088041
O’Keeffe M, Peskov MA, Ramsden SJ, Yaghi OM (2008) The reticular chemistry structure resource (RCSR) database of, and symbols for, crystal nets. Acc Chem Res 41:1782–1789. https://doi.org/10.1021/ar800124u
Macrae CF, Sovago I, Cottrell SJ et al (2020) Mercury 4.0: from visualization to analysis, design and prediction. J Appl Crystallogr 53:226–235. https://doi.org/10.1107/S1600576719014092
Johnston MG, Harrison WTA (2003) Cobalt hydrogen selenite chloride dihydrate, Co(HSeO3)Cl·2H2O. Acta Crystallogr E 59:i62–i64. https://doi.org/10.1107/S1600536803006378
Fridrichová M, Němec I, Císařová I, Němec P (2010) Guanylurea(1+)hydrogen phosphite: a novel promising phase-matchable material for second harmonic generation. CrystEngComm 12:2054. https://doi.org/10.1039/b924973g
Johansson GB, Lindqvist O (1976) A neutron diffraction refinement of the crystal structure of lithium hydrogenphosphite, LiH2PO3. Acta Crystallogr B 32:412–414. https://doi.org/10.1107/S0567740876003130
Infantes L, Motherwell S (2002) Water clusters in organic molecular crystals. CrystEngComm 4:454. https://doi.org/10.1039/b204934a
Banaru A, Slovokhotov YL (2010) On the topology of layered motifs (H2O)∞. CrystEngComm 12:1054–1056. https://doi.org/10.1039/B918793F
Banaru AM, Banaru GA (2011) Cairo tiling and the topology of layered hydrates. Moscow Univ Chem Bull 66:159–160. https://doi.org/10.3103/S0027131411030023
Lord EA, Banaru AM (2012) Number of generating elements in space group of a crystal. Moscow Univ Chem Bull 67:50–58. https://doi.org/10.3103/S0027131412020034
Banaru AM, Banaru DA, Aksenov SM (2022) On the subset of intermolecular contacts generating a molecular crystal: topological features of organic minerals. Crystallogr Reports 67:1133–1145. https://doi.org/10.1134/S1063774522070410
Funding
The X-ray study was carried out in accordance with the state task of the Russian Federation, state registration number 122011300125–2. The topological analysis was supported by the Russian Science Foundation, grant no. 20–77-10065-П.
Author information
Authors and Affiliations
Contributions
Dmitri O. Charkin – conceptualization; synthesis; writing—original draft preparation. Alexander M. Banaru – formal analysis; writing—original draft preparation. Dmitry N. Dmitriev – formal analysis; synthesis. Alevtina N. Gosteva – formal analysis. Vadim E. Kireev – formal analysis. Dina V. Deyneko – formal analysis. Sergey M. Aksenov – conceptualization; formal analysis; writing—original draft preparation; supervision.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The published paper has missing pages.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Charkin, D.O., Banaru, A.M., Dmitriev, D.N. et al. (C2N2H10)[Co(HPHO3)2Cl2]: the first phosphite analog of layered hydrogen selenites. Struct Chem 35, 39–46 (2024). https://doi.org/10.1007/s11224-023-02254-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-023-02254-5