Abstract
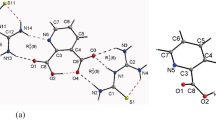
Single crystals of 8-hydroxy-5-nitroquinolinium p-toluene sulfonate (HNT) were grown by the slow evaporation solution growth technique. The structure was elucidated by single-crystal X-ray diffraction analysis, and the crystal belongs to the monoclinic system with the space group C2/c. The crystallinity of HNT was studied by powder X-ray diffraction analysis. The presence of functional groups was determined by FT-IR spectral analysis. The band gap energy is estimated by the application of the Kubelka–Munk algorithm. The charge transfer characteristic of the compound was studied by frontier molecular orbital (FMO) analysis. The first-order hyperpolarizability of the HNT molecule was found to be 285.45 × 10−30 esu, which is ~ 750 times higher compared to the reference urea molecule. Investigation of the intermolecular interactions and crystal studies packing via Hirshfeld surface analysis, based on single-crystal XRD, reveals that the close contacts are associated with molecular interactions. Fingerprint plots of the Hirshfeld surfaces were used to locate and analyze the percentage of hydrogen-bonding interactions. The Mulliken charge of the present molecule was theoretically analyzed. The Kurtz-Perry powder technique has been used to estimate the second harmonic generation. Observed small SHG and large hyperpolarizability are rationalized.













Similar content being viewed by others
Availability of data and materials
Not applicable.
Abbreviations
- HNT:
-
8-Hydroxy-5-nitroquinolinium p-toluene sulfonate
- FT-IR:
-
Fourier transform infrared spectroscopy
- TG-DTA:
-
Thermo-gravimetric and differential thermal analysis
- NLO:
-
Nonlinear optical
- SHG:
-
Second harmonic generation
- MEP:
-
Molecular electrostatic potential
- DFT:
-
Density functional theory
- FMO:
-
Frontier molecular orbitals
- HOMO:
-
Highest occupied molecular orbital
- LUMO:
-
Lowest unoccupied molecular orbital
- UV–Vis DRS:
-
Ultraviolet visible near-infrared diffuse reflectance spectroscopy
- XRD:
-
X-ray diffraction
- CCDC:
-
Cambridge Crystallographic Data Centre
References
Slodek M, Filapek G, Szafraniec I, Grudzka WA, Pisarski JG, Malecki L, Zur M, Grela WD, Krompiec S (2014) Synthesis, electrochemistry, crystal structures, and optical properties of quinoline derivatives with a 2,2′‐bithiophene. Eur J Org Chem 5256–5264
Fox JJ (1910) XCIII-the salts of 8-hydroxyquinoline. J Chem Soc Trans 97:1119–1125
Mason SF, Philp J, Smith BE (1968) Prototropic equilibria of electronically excited molecules. Part II. 3-, 6 and 7-Hydroxyquinoline. J Chem Soc A 3051–3056
Ho CL, Li H, Wong WY (2014) Red to near-infrared organometallic phosphorescent dyes for OLED applications. J Organomet Chem 751:261–285
Xie LH, Yin CR, Lai WY, Fan QL, Huang W (2012) Polyfluorene-based semiconductors combined with various periodic table elements for organic electronics. Prog Polym Sci 37:1192–1264
Guo X, Baumgarten M, Muellen K (2013) Designing π-conjugated polymers for organic electronics. Prog Polym Sci 38:1832–1908
Kalyani NT, Dhoble SJ (2012) Organic light emitting diodes: energy saving lighting technology-a review. Renewable Sustainable Energy Rev 16:2696–2723
Baghernejad B (2011) Application of p-toluenesulfonic acid (PTSA) in organic synthesis. Curr Org Chem 15:3091–3097
Mahindaratne MPD, Wimalasena K (1998) Detailed characterization of p-toluenesulfonic acid monohydrate as a convenient, recoverable, safe, and selective catalyst for alkylation of the aromatic nucleus. J Org Che 632858–2866
Anandha Babu G, Ramasamy P (2011) Growth and characterization of 2-amino-4-picolinium toluene sulfonate single crystal. Spectrochimica Acta Part A 82:521–526
Ramesh Kumar R, Sathya P, Gopalakrishnan R (2016) Structural, vibrational, thermal and optical studies of organic single crystal: benzotriazolium p-toluene sulfonate (BTPTS). AIP Conf Proc 1728:020508
Anandha Babu G, Perumal Ramasamy R, Ramasamy P, Krishna Kumar V (2009) Synthesis, crystal growth, and characterization of an organic nonlinear optical donor-π-acceptor single crystal:2-amino-5-nitropyridinium-toluenesulfonate. Cryst Growth Des 9:3333–3337
Thailam pillai balasubramanian, Packianathan thamos muthaiah, (1996) 8-hydroxy-7-nitroquinoline-5-sulfonic acid monohydrate. Acta Cryst C 52:1017–1019
Muralidharan S, Nagapandiselvi P, Srinivasan T, Gopalakrishnan R, Velmurugan D (2013) L-histidinium p-toluenesulfonate Acta Cryst E 69:0804
Coe Benjamin J, Hall Jonathan J, Harris James A, Brun schwig Bruce S, Coles Simon J, Hursthouse Michael B (2005) trans-4-[(4-dimethylaminophenyl)ethenyl]-N-methylquinolinium p-toluenesulfonate monohydrate. Acta Cryst E 61:0464–0467
Rui-jun Xu (2010) 4-methylanilinium p-toluenesulfonate. Acta Cryst E 66:01794
Aravinth K, Anandha Babu G, Ramasamy P (2012) Growth of <201> 8-hydroxyquinoline organic crystal by Czochralski method and its characterizations. J Therm Anal Calorim 110:1333–1339
Thirumurugan R, Babu B, Anitha K, Chandrasekaran J (2015) Investigation on growth, structure and characterization of succinate salt of 8-hydroxyquinoline: an organic NLO crystal. Spectrochim Acta A Mol Biomol Spectrosc 140:44–53
Sudharsana N, Krishnakumar V, Nagalakshmi R (2014) Synthesis, experimental and theoretical studies of 8-hydroxyquinolinium 3,5-dinitrobenzoate single crystal. J Cryst Growth 398:45–57
Peramaiyan G, Pandi P, Vijayan N, Bhagavannarayana G, Mohan Kumar R (2013) Crystal growth, structural, thermal, optical and laser damage threshold studies of 8-hydroxyquinolinium hydrogen maleate single crystals. J Cryst Growth 375:6–9
Goel S, Yadav H, Sinha N, Singh B, Bdikin I, Kumar B (2018) X-ray, dielectric, piezoelectric and optical analyses of a new nonlinear optical 8-hydroxyquinolinium hydrogen squarate crystal. Acta Cryst B 74:1–12
Burtman V, Teplitsky A, Zelichenok A (2000) Quinolinium derived acentric single crystals. Tetrahedron Lett 41:5397–5402
Zelichenok A, Burtman V, Zenou N, Yitzchaik S (1999) Quinolinium-derived acentric crystals for second-order NLO applications with transparency in the blue. J Phys Chem B 103:8702–8705
Zhao G (2017) Crystal structure of (8-hydroxy-5-nitroquinolinium) perchlorate – 8-hydroxy-5-nitroquinoline (1/1) C18H13ClN4O10. Z Kristallogr 1–3
Selvakumar E, Anandha babu G, Ramasamy P, Rajnikant, Murugesan V, Chandramohan A (2014) Synthesis, growth and spectroscopic investigation of an organic molecular charge transfer crystal: 8-hydroxy quinolinium 4-nitrobenzoate 4-nitrobenzoic acid. Spectrochim Acta A 117:259–263
Mohana J, Divya M, Bharathi G, Ahila G, Chakkaravarthi AG (2015) Crystal structure of quinolinium 2-carboxy-6-nitrobenzoate monohydrate. Acta Cryst E 71:0270–0271
Kobayashi H, Marumo F, Saito Y (1971) The crystal structure of quinolinium bis(7,7,8,8-tetracyanoquinodimethanide) Q+(TCNQ)2-. Acta Cryst B 27:373–378
Jebamony JR, Thomas MP (1998) 8-hydroxyquinolinium-salicylate-salicylic acid (1/1/1) complex. C9H8NO+.C7H5O3-.-C7H6O3. Acta Cryst C 54539–540
Barczynski P, Komasa A, Ratajczak-Sitarz M, Katrusiak A, Brzezinski B (2006) Molecular structure of 8-hydroxy-1-methylquinolinium iodide hydrate in crystal and solution. J Mol Struct 791:106–110
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision C.01, Gaussian, Inc., Wallingford CT
Schlegel B (1982) Optimization of equilibrium geometries and transition structures. J Comput Chem 3214–218
Dennington R, Keith T, Millam J (2009) Gauss View, Version 5. Semichem Inc., Shawnee Mission KS
Wolff SK, Grimwood DJ, McKinnon JJ, Turner MJ, Jayatilaka D, Spackman MA (2012) CrystalExplorer (Version 3.1), University of Western Australia
Kaminsky W (2005) WinXMorph: a computer program to draw crystal morphology, growth sectors and cross sections with export files in VRML V2.0 utf8-virtual reality format. J Appl Cryst 38:566–567
Macrae CF, Bruno IJ, Chisholm JA, Edgington PR, McCabe P, Pidcock E, Rodriguez Monge L, Taylor R, van de Streek J, Wood PA (2008) Mercury CSD 2.0—new features for the visualization and investigation of crystal structures. J Appl Crystallogr 41:466–470
Kubelka P, Munk F (1931) Ein beitrag zur optik der farbanstriche. Zeitschrift fur technische Physik 12:593–601
Zyss J, Oudar JL (1982) Relations between microscopic and macroscopic lowest-order optical nonlinearities of molecular crystals with one- or two-dimensional units. Phy Rew A 26:2028–2048
Garito A, Shi RF, Marvin Wu (1994) Nonlinear optics of organic and polymer materials. Phys Today 47:51–57
Kurtz SK, Perry TT (1968) A powder technique for the evaluation of nonlinear optical materials. J Appl Phys 39:3798–3813
Yatsenko AV, Paseshnichenko KA, Chernysheva VV, Schenk H (2002) Powder diffraction study of the hydrogen bonds in nitroxoline and its hydrochloride. Acta Cryst C 58:019–021
Arora SK, Sundaralingam M (1971) The crystal and molecular structure of 4-methyl sulfonic acid (p-toluenesulfonic acid) monohydrate, C7H8SO3−.H3O+, an oxonium salt. Acta Cryst B 27:1293–1298
Hirshfeld FL (1977) Bonded-atom fragments for describing molecular charge densities. Theor Chim Acta 44:129–138
Spackman MA, McKinnon JJ (2002) Finger printing intermolecular interactions in molecular crystals. CrystEngComm 4:378–392
Jauhar ROMU, Murugakoothan P (2017) Optimization, frontier molecular orbitals and dielectric studies on 2, 6 Diaminopyridinium Tosylate single crystals. AIP Conf Proc 1832:100005
Acknowledgements
Author SS is grateful to the CSIR Emeritus Scientist Scheme for the award of the SRF.
Funding
The Council of Scientific and Industrial Research, New Delhi, provided financial support through research grant No. 21 (1024)/16/EMR-II.
Author information
Authors and Affiliations
Contributions
S S: data curation, investigation, visualization, validation, writing, original draft. K P: data curation, investigation; SP M: review, editing, and supervision. The final version of the manuscript submitted was approved by all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11224_2023_2123_MOESM1_ESM.docx
Supplementary file1 CCDC 1871548 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/datarequest/cif. (DOCX 2997 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sivaraman, S., Markkandan, R., Pandiarajan, K. et al. Hydrogen-bonded structure and optical nonlinearities in the proton-transfer complex of 8-hydroxy-5-nitroquinoline with ρ-toluenesulfonic acid. Struct Chem 34, 1801–1815 (2023). https://doi.org/10.1007/s11224-023-02123-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-023-02123-1