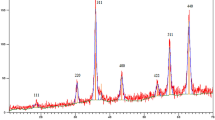
Abstract
Isolation of structural motifs from minerals can be used to improve the understanding of fundamental processes such as catalytic mechanisms and spectroscopic vibrations. In this study, we designed an oxidative dissolution reaction using 2,2′-bipyridine in combination with minerals from the vivianite family. Vivianites are structurally similar to amorphous cobalt oxide water oxidation catalysts (CoCat). Specifically, we used pakhomovskyite (Pak: Co3(PO4)2•8H2O) and erythrite (Ery: Co3(AsO4)2•8H2O)). Through the designed synthetic route, we crystallized two mononuclear isostructural complexes containing phosphate or arsenate and a cobalt-arsenate trinuclear cluster. We observed three main binding modes from the anions: (i) both arsenates or phosphates bind to cobalt forming an O-μ2-bridge, (ii) arsenate forms a bridge between two cobalt atoms, and (iii) arsenate binds between three cobalt atoms expanding its coordination sphere to five. This structural information allows us to propose an updated structural model for amorphous cobalt oxide catalysts containing arsenate.





Similar content being viewed by others
References
Janet G, Hering WS (1990) Oxidative and reductive dissolution of minerals. Mineral-water interface geochemistry23rd edn. Mineralogical Society of America, Washington DC, pp 427–469
Zhu Z, Zhang H, Lü Z, Chen D (2007) Oxidative dissolution of cobalt oxalate and manganese oxalate in acetic acid solutions. Chem Eng Process Process Intensif 46:159–164. https://doi.org/10.1016/j.cep.2006.05.009
Li X, Van den Bossche A, Vander Hoogerstraete T, Binnemans K (2018) Ionic liquids with trichloride anions for oxidative dissolution of metals and alloys. Chem Commun 54:475–478. https://doi.org/10.1039/C7CC08645H
Corkhill CL, Wincott PL, Lloyd JR, Vaughan DJ (2008) The oxidative dissolution of arsenopyrite (FeAsS) and enargite (Cu3AsS4) by Leptospirillum ferrooxidans. Geochim Cosmochim Acta 72:5616–5633. https://doi.org/10.1016/j.gca.2008.09.008
Adamczyk Z, Oćwieja M, Mrowiec H et al (2016) Oxidative dissolution of silver nanoparticles: a new theoretical approach. J Colloid Interface Sci 469:355–364. https://doi.org/10.1016/j.jcis.2015.12.051
O’Brien AB, Segal MG, Williams WJ (1987) Kinetics of metal oxide dissolution. Oxidative dissolution of chromium from mixed nickel–iron–chromium oxides by permanganate. J Chem Soc Faraday Trans 1 Phys Chem Condens Phases 83:371–382. https://doi.org/10.1039/F19878300371
O’Brien MN, Jones MR, Brown KA, Mirkin CA (2014) Universal noble metal nanoparticle seeds realized through iterative reductive growth and oxidative dissolution reactions. J Am Chem Soc 136:7603–7606. https://doi.org/10.1021/ja503509k
Chiriţă P, Bădică CE, Constantin CA et al (2014) Influence of 2,2′-bipyridine on oxidative dissolution of iron monosulfide. Surf Interface Anal 46:842–846. https://doi.org/10.1002/sia.5459
Kumagai Y, Barreiro Fidalgo A, Jonsson M (2019) Impact of stoichiometry on the mechanism and kinetics of oxidative dissolution of UO2 induced by H2O2 and γ-irradiation. J Phys Chem C 123:9919–9925. https://doi.org/10.1021/acs.jpcc.9b00862
Lavrent’ev IP, Khidekel’ ML (1983) Oxidative dissolution of transition metals in a liquid phase. role of oxygen and of the surface oxide layer. Russ Chem Rev 52:337–349. https://doi.org/10.1070/rc1983v052n04abeh002821
Xue Y, Wang Y, Zheng S et al (2017) Efficient oxidative dissolution of V2O3 by the in situ electro-generated reactive oxygen species on N-doped carbon felt electrodes. Electrochim Acta 226:140–147. https://doi.org/10.1016/j.electacta.2017.01.001
Hong J, Liu L, Tan W, Qiu G (2020) Arsenic release from arsenopyrite oxidative dissolution in the presence of citrate under UV irradiation. Sci Total Environ 726:138429. https://doi.org/10.1016/j.scitotenv.2020.138429
Fallon EK, Petersen S, Brooker RA, Scott TB (2017) Oxidative dissolution of hydrothermal mixed-sulphide ore: an assessment of current knowledge in relation to seafloor massive sulphide mining. Ore Geol Rev 86:309–337. https://doi.org/10.1016/j.oregeorev.2017.02.028
Marfunin AS (2012) Advanced mineralogy: volume 1 composition, structure, and properties of mineral matter: concepts, results, and problems. Springer, Berlin Heidelberg
Kanan MW, Nocera DG (2008) In situ formation of an water containing phosphate and Co 2 +. Science 321:1072–1075. https://doi.org/10.1126/science.1162018
Risch M, Khare V, Zaharleva I et al (2009) Cobalt-oxo core of a water-oxidizing catalyst film. J Am Chem Soc 131:6936–6937. https://doi.org/10.1021/ja902121f
Kanan MW, Yano J, Surendranath Y et al (2010) Structure and valency of a cobalt-phosphate water oxidation catalyst determined by in situ X-ray spectroscopy. J Am Chem Soc 132:13692–13701. https://doi.org/10.1021/ja1023767
Risch M, Klingan K, Ringleb F et al (2012) Water oxidation by electrodeposited cobalt oxides-role of anions and redox-inert cations in structure and function of the amorphous catalyst. ChemSusChem 5:542–549. https://doi.org/10.1002/cssc.201100574
Rigsby ML, Mandal S, Nam W et al (2012) Cobalt analogs of Ru-based water oxidation catalysts: overcoming thermodynamic instability and kinetic lability to achieve electrocatalytic O 2 evolution. Chem Sci 3:3058–3062. https://doi.org/10.1039/c2sc20755a
Dimitrou K, Folting K, Streib WE, Christou G (1993) “Dimerization” of the [CoIII2(OH)2] core to the first example of a [CoIII4O4] cubane: potential insights into photosynthetic water oxidation. J Am Chem Soc 115:6432–6433. https://doi.org/10.1021/ja00067a077
Brodsky CN, Hadt RG, Hayes D et al (2017) In situ characterization of cofacial Co(IV) centers in Co 4 O 4 cubane: modeling the high-valent active site in oxygen-evolving catalysts. Proc Natl Acad Sci 114:3855–3860. https://doi.org/10.1073/pnas.1701816114
Hong D, Jung J, Park J et al (2012) Water-soluble mononuclear cobalt complexes with organic ligands acting as precatalysts for efficient photocatalytic water oxidation. Energy Environ Sci 5:7606–7616. https://doi.org/10.1039/c2ee21185h
Wang D, Groves JT (2013) Efficient water oxidation catalyzed by homogeneous cationic cobalt porphyrins with critical roles for the buffer base. Proc Natl Acad Sci 110:15579–15584. https://doi.org/10.1073/pnas.1315383110
Nakazono T, Parent AR, Sakai K (2013) Cobalt porphyrins as homogeneous catalysts for water oxidation. Chem Commun 49:6325–6327. https://doi.org/10.1039/c3cc43031f
Fu S, Liu Y, Ding Y et al (2014) A mononuclear cobalt complex with an organic ligand acting as a precatalyst for efficient visible light-driven water oxidation. Chem Commun 50:2167–2169. https://doi.org/10.1039/c3cc48059c
Leung CF, Ng SM, Ko CC et al (2012) A cobalt(ii) quaterpyridine complex as a visible light-driven catalyst for both water oxidation and reduction. Energy Environ Sci 5:7903–7907. https://doi.org/10.1039/c2ee21840b
Ullman AM, Brodsky CN, Li N et al (2016) Probing edge site reactivity of oxidic cobalt water oxidation catalysts. J Am Chem Soc 138:4229–4236. https://doi.org/10.1021/jacs.6b00762
Wang JW, Sahoo P, Lu TB (2016) Reinvestigation of water oxidation catalyzed by a dinuclear cobalt polypyridine complex: identification of CoOx as a real heterogeneous catalyst. ACS Catal 6:5062–5068. https://doi.org/10.1021/acscatal.6b00798
González-Flores D, Klingan K, Chernev P et al (2018) Nickel-iron catalysts for electrochemical water oxidation-redox synergism investigated by: in situ X-ray spectroscopy with millisecond time resolution. Sustain Energy Fuels 2:1986–1994. https://doi.org/10.1039/c8se00114f
Drevon D, Görlin M, Chernev P et al (2019) Uncovering the role of oxygen in Ni-Fe(OxHy) electrocatalysts using in situ soft X-ray absorption spectroscopy during the oxygen evolution reaction. Sci Rep 9:1–11. https://doi.org/10.1038/s41598-018-37307-x
Smith RDL, Pasquini C, Loos S et al (2017) Spectroscopic identification of active sites for the oxygen evolution reaction on iron-cobalt oxides. Nat Commun 8:2022. https://doi.org/10.1038/s41467-017-01949-8
Zaharieva I, González-Flores D, Asfari B et al (2016) Water oxidation catalysis-role of redox and structural dynamics in biological photosynthesis and inorganic manganese oxides. Energy Environ Sci 9:2433–2443. https://doi.org/10.1039/c6ee01222a
Costentin C, Nocera DG (2017) Self-healing catalysis in water. Proc Natl Acad Sci 114:13380–13384. https://doi.org/10.1073/PNAS.1711836114
Eom C, Suntivich J (2019) In situ stimulated Raman spectroscopy reveals phosphate network in amorphous cobalt oxide catalyst and its role in the catalyst formation. J Phys Chem C 123:acs.Jpcc.9b10308. https://doi.org/10.1021/acs.jpcc.9b10308
Villalobos J, González-Flores D, Klingan K et al (2019) Structural and functional role of anions in electrochemical water oxidation probed by arsenate incorporation into cobalt-oxide materials. Phys Chem Chem Phys:12485–12493. https://doi.org/10.1039/c9cp01754b
González-Flores D, Sánchez I, Zaharieva I et al (2015) Heterogeneous water oxidation: surface activity versus amorphization activation in cobalt phosphate catalysts. Angew Chem Int Ed 54:2472–2476. https://doi.org/10.1002/anie.201409333
Coe BJ, Glenwright SJ (2000) Trans-effects in octahedral transition metal complexes. Coord Chem Rev 203:5–80. https://doi.org/10.1016/S0010-8545(99)00184-8
Robinson K, Gibbs GV, Ribbe PH (1971) Quadratic enlongation: a quantitative measure of distortion in Coordinatino Polyhedra. Science (80-) 172:567–570
Yaghoobi Nia N, Farahani P, Sabzyan H et al (2014) A combined computational and experimental study of the [Co(bpy)3]2+/3+ complexes as one-electron outer-sphere redox couples in dye-sensitized solar cell electrolyte media. Phys Chem Chem Phys 16:11481–11491. https://doi.org/10.1039/C3CP55034F
O’Keeffe M, Brese NE (1991) Atom sizes and bond lengths in molecules and crystals. J Am Chem Soc 113:3226–3229. https://doi.org/10.1021/ja00009a002
Wood RM, Palenik GJ (2002) Bond valence sums in coordination chemistry. A simple method for calculating the oxidation state of cobalt in complexes containing only Co−O bonds. Inorg Chem 37:4149–4151. https://doi.org/10.1021/ic980176q
Urusov VS (1995) Semi-empirical groundwork of the bond-valence model. Acta Crystallogr Sect B 51:641–649. https://doi.org/10.1107/S0108768195003417
Dimitrou K, Folting K, Streib WE et al (1993) “Dimerization” of the [co III2 (OH) 2 ] core to the first example of a [co III4 O 4 ] cubane: potential insights into photosynthetic water oxidation. J Am Chem Soc 115:6432–6433. https://doi.org/10.1021/ja00067a077
Yakovenchuk VN, Ivanyuk GY, Mikhailova YA, Selivanova EA (2006) Pakhomovskyite, Co3(PO4)2·8H2O, a new mineral species from Kovdor, Kola Peninsula, Russia. Can Mineral 44:117–123
Cotton FA, Cotton FA (1999) Advanced inorganic chemistry
Stubbert BD, Peters JC, Gray HB (2011) Rapid water reduction to H2 catalyzed by a cobalt bis(iminopyridine) complex. J Am Chem Soc 133:18070–18073. https://doi.org/10.1021/ja2078015
Eckenhoff WT, McNamara WR, Du P, Eisenberg R (2013) Cobalt complexes as artificial hydrogenases for the reductive side of water splitting. Biochim Biophys Acta Bioenerg 1827:958–973. https://doi.org/10.1016/j.bbabio.2013.05.003
Klingan K, Ringleb F, Zaharieva I et al (2014) Water oxidation by amorphous cobalt-based oxides: volume activity and proton transfer to electrolyte bases. ChemSusChem 7:1301–1310. https://doi.org/10.1002/cssc.201301019
Berthet P, Berthon J, d’Yvoire F (1995) XAFS study of sixfold coordinated arsenic(V) in LiAsO3 and CaAs2O6. Phys B Condens Matter 208–209:597–598. https://doi.org/10.1016/0921-4526(94)00877-X
Padhi SK, Sahu R, Manivannan V (2011) Syntheses and structures of cobalt(III) alcoholate complexes formed by addition of a water molecule across 2-pyridyl substituted imine function. Inorg Chim Acta 367:57–63. https://doi.org/10.1016/j.ica.2010.11.035
Housecroft CE, Sharpe AG (2007) Inorganic chemistry, 6th ed
Xu R, Zeng HC (2004) Self-generation of tiered surfactant superstructures for one-pot synthesis of Co3O4 nanocubes and their close- and non-close-packed organizations. Langmuir 20:9780–9790. https://doi.org/10.1021/la049164+
Farhadi S, Safabakhsh J, Zaringhadam P (2013) Synthesis, characterization, and investigation of optical and magnetic properties of cobalt oxide (Co3O4) nanoparticles. J Nanostructure Chem 3:69. https://doi.org/10.1186/2193-8865-3-69
He T, Chen D, Jiao X et al (2005) Solubility-controlled synthesis of high-quality Co3O4 nanocrystals. Chem Mater 17:4023–4030. https://doi.org/10.1021/cm050727s
Herrero M, Benito P, Labajos FM, Rives V (2007) Nanosize cobalt oxide-containing catalysts obtained through microwave-assisted methods. Catal Today 128:129–137. https://doi.org/10.1016/j.cattod.2007.06.070
Barreca D, Massignan C, Daolio S et al (2001) Composition and microstructure of cobalt oxide thin films obtained from a novel cobalt(II) precursor by chemical vapor deposition. Chem Mater 13:588–593. https://doi.org/10.1021/cm001041x
Risch M, Ringleb F, Kohlhoff M et al (2015) Water oxidation by amorphous cobalt-based oxides: in situ tracking of redox transitions and mode of catalysis. Energy Environ Sci 8:661–674. https://doi.org/10.1039/C4EE03004D
Safdari R, Mohammadi MR, Hołyńska M et al (2018) A mononuclear cobalt complex for water oxidation: new controversies and puzzles. Dalton Trans 47:16668–16673. https://doi.org/10.1039/C8DT03147A
Riou A, Cudennec Y, Gerault Y (1989) Cobalt(II) orthophosphate octahydrate. Acta Crystallogr Sect C 45:1412–1413. https://doi.org/10.1107/S0108270189003616
Zhu YN, Zhang XH, Chen YD et al (2013) Characterisation, dissolution and solubility of synthetic erythrite [Co 3 (AsO 4 ) 2 .8H 2 O] and annabergite [Ni 3 (AsO 4 ) 2 .8H 2 O] at 25°C. Can Metall Q 52:7–17. https://doi.org/10.1179/1879139512Y.0000000030
Acknowledgments
We thank Dr. Leslie W. Pineda for data collection and Dr. Mauricio Gutiérrez and Ravyn Malatesta for productive discussion.
Funding
This work was funded by Vicerrectoría de Investigación (UCR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
We did not violate any ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 5435 kb).
Rights and permissions
About this article
Cite this article
Rojas-Gatjens, E., Viales-Montero, C., Urcuyo, R. et al. Oxidative dissolution of synthetic vivianites as a method for the crystallization of molecular structural motifs. Struct Chem 32, 445–455 (2021). https://doi.org/10.1007/s11224-020-01644-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-020-01644-3