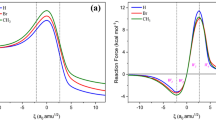
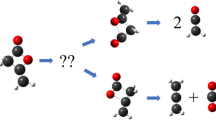
Abstract
Theoretical studies of the retro-Diels-Alder reaction of 1,4-cyclohexadiene, 4H-pyran 4H-thiopyran, 1,4-dioxine, and 1,4-dithiine in the gas phase were carried out using DFT methods at the B3LYP/6–311 + G(d,p) levels of theory. The barrier height (ΔE‡) and thermodynamic parameters (ΔG‡ and ΔH‡) were estimated. The progress of the reactions was followed by means of the Wiberg bond indices. The synchronicity values of the reactions were calculated. The kinetic parameters were calculated for both reactions in 300–1200-K temperature range. Also, fitted equations to the gas phase Arrhenius equation were determined. Effect of the character and number of heteroatoms were illustrated on the thermodynamic and kinetic parameters.





Similar content being viewed by others
References
Fringuelli F, Taticchi A (2002) The Diels-Alder reaction. Selected Practical Methods. John Wiley & Sons, New York
Fleming I (1999) Pericyclic reactions. Oxford University Press Inc., New York
Nicolau KC, Snyder SA, Montagnon T, Vassilikogiannakis G (2002). Angew Chem Int Ed 41:1668
Diels O, Alder K (1928). Justus Liebigs Ann Chem 98:460
Carruthers W (1978) Some Modern Methods of Organic SynthesisSecond edn. Cambridge University Press, Cambridge
Carruthers W (1990) Ycloaddition Reactions in Organic Synthesis. Pergamon, Oxford
Geerlings P, Proft FD, Langenaeker W (2003). Chem Rev 103:1793
Ess DH, Jones GO, Houk KN (2006). Adv Synth Catal 348:2337
Ormachea CM, Mancini PME, Kneeteman MN, Domingo LR (2015). Comput Theor Chem 1072:37
Sarotti AM (2014). Org Biomol Chem 12:187
Mancinia PME, Kneeteman MN, Cainelli M, Ormachea CM, Domingo LR (2017). J Mol Struct 1147:155
Rickborn B (1998) The retro Diels-Alder reaction. Part I. C-C dienophiles in Organic Reactions. In: Al LP e (ed), vol Vol. 52. John Wiley and Sons, Inc, New York
Ramirez B, Cordova T, Ruette F, Chuchani G (2015). Comput Theor Chem 1067:103
Tsang W (1965). J Chem Phys 42:1805
Sakai T, Nakatani T, Toshiaki N, Kunugi T (1972). Ind Eng Chem Fundam 11:529
Lewis DK, Brandt B, Crockford L, Glenar DA, Rauscher G, Rodriguez J, Baldwin JE (1993). J Am Chem Soc 115:11728
Lewis D, Glenar DA, Hughes S, Kalra BL, Schlier J, Shukla R, Baldwin JE (2001). J Am Chem Soc 123:996
Abu-Laban M, Kumal RR, Casey J, Becca J, Hayes DJ (2018). J Colloid Interface Sci 526:312
Lyu B, Cha W, Mao T, Wu Y, Qian H, Zhou Y, Chen X, Zhang S, Liu L, Yang G, Lu Z, Zhu Q, Ma H (2015). ACS Appl Mater Interfaces 7:6254
Shrivastav G, Khan TS, Agarwal M, Haider MA (2018). J Phys Chem C 122:11599
Yang K, Dang Q, Cai P-J, Gao Y, Yu Z-X, Bai X (2017). J Org Chem 82:2336
Pinelo L, Gudmundsdottir AD, Ault BS (2013). J Phys Chem A 117:4174
Darensbourg DJ, Chung W-C, Yeung AD, Luna M (2015). Macromolecules 48:1679
Krier JM, Komvopoulos K, Somorjai GA (2016). J Phys Chem C 120:8246
Gomez N, Hénon E, Bohr F, Devolder P (2001). J Phys Chem A 105:11204
Gao Y, DeYonker NJ, Garrett EC, Wilson AK, Cundari TR, Marshall P (2009). J Phys Chem A 113:6955
Benson SW, Shaw R (1967). Trans Faraday Soc 63:985
Rice FO, Stallbaumer AL (1942). J Am Chem Soc 64:1527
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalman G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision A.02. Gaussian, Inc., Wallingford CT
Krishnan R, Binkley JS, Seeger R, Pople JA (1980). J Chem Phys 72:650
McLean AD, Chandler GS (1980). J Chem Phys 72:5639
Curtiss LA, McGrath MP, Blandeau J-P, Davis NE, Binning RC, Radom JL (1995). J Chem Phys 103:6104
Becke AD (1993). J Chem Phys 98:5648
Barone V (2005). J Chem Phys 122:014108
Barone V, Manichino C (1995). J Mol Struct 339:365
Reed AE, Curtiss LA, Weinhold F (1988). Chem Rev 88:899
Glendening ED, Reed AE, Carpenter JE, Weinhold F (1988). NBO (Version 3.1)
Fukui K (1981). Acc Chem Res 14:363
Fukui K (1970). J Phys Chem 74:4161
Gonzalez C, Schlegel HB (1990). J Phys Chem 94:5523
Gonzalez C, Schlegel HB (1989). J Chem Phys 90:2154
Miyoshi A (2010) Gaussian Post Processor(GPOP). University of Tokyo, Tokyo
Garrett BC, Truhlar DG (1979). J Phys Chem 83:2921
Shavitt I (1959). J Chem Phys 31:1359
Miessler GL, Fischer PJ, Tarr DA (2014) Inorganic ChemistryFifth edn. Pearson Education, Inc
Gavnholt J, Olsen T, Engelund M, Schiøtz J (2008). Phys Rev B 78:075441
Parr RG, Pearson RG (1983). J Am Chem Soc 105:7512
Yang W, Parr RG (1985). Proc Natl Acad Sci U S A 82:6723
Chen X-F, Bu J-H, Yu T, Lai WP, Ge Z-X (2013). Commun Comput Chem 1:118
Wiberg KB (1968). Tetrahedron 24:1083
Moyano A, Perica’s MA, EV (1995). J Organomet Chem 54:573
Manoharan M, Venuvanalingam P (1997). J Mol Struct (THEOCHEM) 394:41
Manoharan M, Venuvanalingam P (1997). J Chem Soc Perkin Trans 1799
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ansari, E.S., Ghiasi, R. & Forghaniha, A. Thermodynamic and kinetic studies of the retro-Diels-Alder reaction of 1,4-cyclohexadiene, 4H-pyran 4H-thiopyran, 1,4-dioxine, and 1,4-dithiine: a theoretical investigation. Struct Chem 30, 877–885 (2019). https://doi.org/10.1007/s11224-018-1241-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-018-1241-y