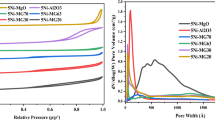
Abstract
Catalytic decomposition of methane (CDM) is an environmentally friendly technique to produce hydrogen and nano-carbon materials without greenhouse gas emissions. To compare the catalytic activity and hydrogen productivity of various active metals in the CDM reaction, experiments were performed by synthesizing Fe–Al, Co–Al, and Ni–Al catalysts through the co-precipitation method. According to the hydrogen production calculation for each catalyst, the Ni-based catalyst exhibited the best hydrogen production at a reaction temperature of 600 °C. It could be attributed to the influence of the strong metal–support interaction on the catalytic activity. Based on the Ni–Al catalyst, which showed better hydrogen production than the other active metals,
the activity and hydrogen production of the CDM process were analyzed according to different process factors. 90 L/gcat of hydrogen was produced through the optimization of process factors such as reduction temperature, reduction time, spatial velocity, methane flow, and gas addition.














Similar content being viewed by others
References
I. Dincer, Renew. Sust Energ. 4, 157 (2000)
D.W. Kim, L.K. Kwac, H.G. Kim, S.K. Ryu, Carbon Lett. 32, 295 (2022)
A.-S. Al-Fatesh, S. Barama, A.-A. Ibrahim, A. Barama, W.-U. Khan, A. Fakeeha, Chem. Eng. Commun. 204, 739 (2017)
N. Naeem, A.H. Khoja, F.A. Butt, M. Arfan, R. Liaquat, A.U. Hasnat, Res. Chem. Intermed. 48, 2007 (2022)
H.F. Abbas, W.W. Daud, Int. J. Hydrogen Energy 35, 1160 (2010)
H.F. Abbas, W.W. Daud, Appl Catal A: Gen 388, 232 (2010)
M. Balat, M. Balat, Int. J. Hydrogen Energy 34, 3589 (2009)
A.H. Khoja, M. Anwar, S. Shakir, M.T. Mehran, A. Mazhar, A. Javed, N.A.S. Amin, Res. Chem. Intermed. 46, 3817 (2020)
R. Musamali, Y.M. Isa, Energy Technol. 7, 1800593 (2019)
R. Minhas, A.H. Khoja, N. Naeem, S. Shakir, R. Liaquat, I.U. Din, Res. Chem. Intermed. 49, 3181 (2023)
Y.A. Al-Baqmaa, A.S. Al-Fatesh, A.A. Ibrahim, A.A. Bagabas, F.S. Almubadde, A.I. Alromaeh, J.K. Abu-Dahrieh, A.E. Abasaeed, A.H. Fakkeh, Res. Chem. Intermed. 49, 5015 (2023)
Y. Li, J. Li, W. Yang, X. Wang, Nanoscale Horiz. 5, 1174 (2020)
N. Bayat, M. Rezaei, F. Meshkani, Int. J. Hydrogen Energy 41, 1574 (2016)
K.R. Parmar, K. Pant, S. Roy, Energy Convers. Manag. 232, 113893 (2021)
N. Muradov, F. Smith, C. Huang, T. Ali, Catal. Today 116, 281 (2006)
V. Palma, E. Meloni, S. Renda, M. Martino, C 6, 52 (2020)
D. Torres, S. De Llobet, J. Pinilla, M. Lázaro, I. Suelves, R. Moliner, J Nat Gas Sci Eng 21, 367 (2012)
T. Koerts, M.J. Deelen, R.A. Van Santen, J. Catal. 138, 101 (1992)
R.A. Couttenye, M.H. De Vila, S.I. Suib, J. Catal. 233, 317 (2005)
R.R. Silva, H.A. Oliveira, A.C. Guarino, B.B. Toledo, M.B. Moura, B.T. Oliveira, F.B. Passos, Int. J. Hydrogen Energy 41, 6763 (2016)
A.A. Ibrahim, A.H. Fakeeha, A.S. Al-Fatesh, A.E. Abasaeed, W.U. Khan, Int. J. Hydrogen Energy 40, 7593 (2015)
Y. Echegoyen, I. Suelves, M. Lazaro, R. Moliner, J. Palacios, J. Power. Sour 169, 150 (2007)
B. Han, F. Wang, L. Zhang, Y. Wang, W. Fan, L. Xu, H. Yu, Z. Li, Res. Chem. Intermed. 46, 1735 (2020)
A.C. Dupuis, Prog. Mater. Sci. 50, 929 (2005)
H. Gergeroglu, M.F. Ebeoglugil, Carbon Lett. 32, 1729 (2022)
S.-P. Chai, S.H.S. Zein, A.R. Mohamed, Chem. Phys. Lett. 426, 345 (2006)
E. Välimäki, L. Yli-Varo, H. Romar, U. Lassi, C. 7, 50 (2021)
S.J. Park, K.D. Kim, Y.S. Park, K.S. Go, W. Kim, M. Kim, N.S. Nho, D.H. Lee, J. Ind. Eng. Chem. 109, 384 (2022)
S.-P. Chai, S.H.S. Zein, A.R. Mohamed, Appl Catal A: Gen 326, 173 (2007)
de Jong KP, (2009) Synthesis of solid catalysts, 1st edn. Wiley, UK, pp 135–151.
M. Popov, P. Kurmashov, A. Bannov, J. Phys. Conf. Ser. 1942, 012038 (2021)
K. Harun, S. Adhikari, H. Jahromi, RSC Adv. 10, 40882 (2020)
R. Yang, X. Li, J. Wu, X. Zhang, Z. Zhang, Y. Cheng, J. Guo, Appl Catal A: Gen 368, 105 (2009)
N.W. Hurst, S.J. Gentry, A. Jones, B.D. McNicol, Chem. Rev. 24, 233 (1982)
A. Venugopal, S.N. Kumar, J. Ashok, D.H. Prasad, V.D. Kumari, K. Prasad, M. Subrahmanyam, Int. J. Hydrogen Energy 32, 1782 (2007)
G. Wang, F. Luo, K. Cao, Y. Zhang, J. Li, F. Zhao, R. Chen, J. Hong, Energy Technol. 7, 1800359 (2019)
B. Saini, M. Yadav, S.K. Jha, R. Krishnapriya, P. Kang, V. Kant, R. Singhal, R.K. Sharma, Sustain Energy Fuels 7, 2568 (2023)
A. Gharibi, A. Shariati, M.A. Takassi, Chin. J. Chem. Eng. 21, 1007 (2013)
W. Ma, G. Jacobs, W.D. Shafer, Y. Ji, J.L.S. Klettlinger, S. Khalid, S.D. Hopps, B.H. Davis, Catalysts 9, 862 (2019)
T.S.T. Saharuddin, F. Salleh, A. Samsuri, R. Othaman, M.A. Yarmo, Int. J. Chem. Eng. Appl. 6, 405 (2015)
J. Zieliński, I. Zglinicka, L. Znak, Z. Kaszkur, Appl. Catal. A 381, 191 (2010)
H. Ago, K. Nakamura, N. Uehara, M. Tsuji, J. Phys. Chem. B 108, 18908 (2004)
B. Gao, I.-W. Wang, L. Ren, T. Haines, J. Hu, Ind. Eng. Chem. Res. 58, 798 (2018)
J.B. Choi, J.S. Im, S.C. Kang, Y.S. Lee, C.W. Lee, Carbon Lett. 33, 477 (2023)
L. Wang, L. Wang, X. Meng, F.S. Xiao, Adv. Mater. 31, 1901905 (2019)
R.K. Singha, A. Shukla, A. Yadav, L.S. Konathala, R. Bal, Appl. Catal. B 202, 473 (2017)
S. Takenaka, M. Serizawa, K. Otsuka, J. Catal. 222, 520 (2004)
I. Suelves, J. Pinilla, M. Lázaro, R. Moliner, J. Palacios, J. Power. Sour. 192, 35 (2009)
N. Rodriguez, M. Kim, R. Baker, J. Catal. 144, 93 (1993)
Y. Zhang, K.J. Smith, Catal. Today 77, 257 (2002)
Q. Chen, A.C. Lua, Chem. Eng. J. 389, 124366 (2020)
Acknowledgements
This work was supported the Technology Innovation Program (20010853, Development of natural gas-based carbon material on graphitic structure for high crystalline conductivity) funded by the Ministry of Trade, Industry & Energy (MOTIE, Korea). This work was also supported by the Technology Innovation Program (RS-2023-00265608, Development of production technology of high value-added chemical using by-product gas in Naphtha Cracker) funded by the Ministry of Trade Industry & Energy (MOTIE, Korea).
Author information
Authors and Affiliations
Contributions
In ho Seong wrote the main manuscript text and prepared figures and tables. Seok Chang Kang, Jong Dae Lee, Ji Sun Im reiviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
There are no potential conflicts of interest relevant to this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Seong, I.H., Kang, S.C., Lee, J.D. et al. Effect of active metals and process factors on the catalytic decomposition of methane for hydrogen production. Res Chem Intermed (2024). https://doi.org/10.1007/s11164-024-05284-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11164-024-05284-8