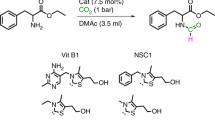
Abstract
Developing and/or modifying fundamental chemical reactions using chemical industry-favorite heterogeneous recoverable catalytic systems in the water solvent is very important. In this paper, we developed convenient, green, and efficient approaches for the chemoselective reduction of nitroarenes, N-acetylation of arylamines, and one-pot reductive acetylation of nitroarenes in the presence of the recoverable heterogeneous carbon-supported palladium (Pd/C) catalytic system in water. The utilize of the simple, effective, and recoverable catalyst and also using of water as an entirely green solvent along with relatively short reaction times and good-to-excellent yields of the desired products are some of the noticeable features of the presented synthetic protocols.
Graphic abstract








Similar content being viewed by others
References
S. Noble, K.L. Goa, Drugs 60, 1383 (2000)
B.A. Lipsky, M.B. Dorr, D.J. Magner, G.H. Talbot, Clin. Ther. 21, 148 (1999)
M.J. Hochmair, S. Shwab, H. Porsch, Anticancer Drugs 28, 928 (2017)
D.B. Huang, M. Dryden, Future Microbiol. 13, 957 (2018)
C.J.M. Wright, P.L. McCormack, Drugs 73, 1245 (2013)
A. Aminoshariae, A. Khan, J. Endodontics 41, 588 (2015)
P.C. Cruz, F.A. Rocha, A.M. Ferreira, Org. Process Res. Dev. 23, 2592 (2019)
F. Gao, R. Bai, F. Ferlin, L. Vaccaro, M. Li, Y. Gu, Green Chem. 22, 6240 (2020)
P.T. Anastas, J.B. Zimmerman, Green Chem. 21, 6545 (2019)
C.J. Clarke, W.-C. Tu, O. Levers, A. Bröhl, J.P. Hallet, Chem. Rev. 118, 747 (2018)
M.H. Muhammad, X.-L. Chen, Y. Liu, T. Shi, Y. Peng, L. Qu, B. Yu, ACS Sustain. Chem. Eng. 8, 2682 (2020)
M. Rimaz, H. Mousavi, P. Keshavarz, B. Khalili, Curr. Chem. Lett. 4, 159 (2015)
M. Rimaz, H. Mousavi, M. Behnam, B. Khalili, Curr. Chem. Lett. 5, 145 (2016)
M. Rimaz, Z. Jalalian, H. Mousavi, R.H. Prager, Tetrahedron Lett. 57, 105 (2016)
M.H. Sayahi, S. Bahadorikhalili, S.J. Saghanezhad, M.A. Miller, M. Mahdavi, Res. Chem. Intermed. 46, 491 (2020)
S.F. Hojati, A. Amiri, M. Mohamed, Res. Chem. Intermed. 46, 1091 (2020)
M. Zahedifar, B. Pouramiri, R. Razavi, Res. Chem. Intermed. 46, 2749 (2020)
B. Zeynizadeh, M. Gilanizadeh, Res. Chem. Intermed. 46, 2969 (2020)
N. Hosseini Mohtashami, M. Gholizadeh, Res. Chem. Intermed. 46, 3037 (2020)
N. Kerru, L. Gummidi, S.V.H.S. Bhaskaruni, S.N. Maddila, S.B. Jonnalagadda, Res. Chem. Intermed. 46, 3067 (2020)
R.C. Patil, U.P. Patil, A.A. Jagdale, S.K. Shinde, S.S. Patil, Res. Chem. Intermed. 46, 3527 (2020)
Z. Lasemi, M. Tajbakhsh, H. Alinezhad, F. Mehrparvar, Res. Chem. Intermed. 46, 3667 (2020)
M. Rajabi, J. Albadi, A. Momeni, Res. Chem. Intermed. 46, 3879 (2020)
N. Nikooei, M.G. Dekamin, E. Valiey, Res. Chem. Intermed. 46, 3891 (2020)
S.M. Arde, A.D. Patil, A.H. Mane, P.R. Salokhe, R.S. Salunkhe, Res. Chem. Intermed. 46, 5069 (2020)
M. Rimaz, J. Khalafy, H. Mousavi, Res. Chem. Intermed. 42, 8185 (2016)
M. Rimaz, J. Khalafy, H. Mousavi, S. Bohlooli, B. Khalili, J. Heterocyclic Chem. 54, 3174 (2017)
J. Sun, Y. Gui, Y. Huang, J. Li, Z. Zha, Y. Yang, Z. Wang, ACS Omega 21, 11962 (2020)
Y. Wei, J. He, Y. Liu, L. Xu, L. Vaccaro, P. Liu, Y. Gu, ACS Omega 29, 18515 (2020)
A.Y. Li, A. Moores, ACS Sustain. Chem. Eng. 7, 10182 (2019)
R.A. Sheldon, Chem. Commun. 5, 3352 (2008)
P.T. Anastas, M.M. Krichhoff, T.C. Williamson, Appl. Catal. A Gen. 221, 3 (2001)
G. Centi, S. Perathoner, Top. Catal. 52, 948 (2009)
M. Rimaz, H. Mousavi, M. Behnam, L. Sarvari, B. Khalili, Curr. Chem. Lett. 6, 55 (2017)
E.D. Goodman, C. Zhao, M. Cargnello, ACS Cent. Sci. 6, 1916 (2020)
R. Schlögl, Angew. Chem. Int. Ed. 54, 3465 (2015)
R. Schlögl, Angew. Chem. Int. Ed. 32, 381 (1993)
R. Lung, X. Du, Y. Huang, X. Jiang, Q. Zhang, Y. Guo, K. Liu, B. Qiao, A. Wang, T. Zhang, Chem. Rev. 120, 11986 (2020)
R. Lippi, C.J. Coghlan, S.C. Howard, C.D. Easton, Q. Gu, J. Patel, C.J. Sumby, D.F. Kennedy, C.J. Dooman, Aust. J. Chem. 73, 1271 (2020)
D. Sarkar, S. Behera, Tetrahedron Lett. 61, 151341 (2020)
H. Peng, K. Jiang, G. Zhen, F. Wang, B. Yin, RSC Adv. 10, 11750 (2020)
B.S. Takale, R.R. Thakore, E.S. Gao, F. Gallou, B.H. Lipshutz, Green Chem. 22, 6055 (2020)
Y. Hu, C. Liu, P. Wang, G. Li, A. Wang, Y. Cong, X. Liang, W. Li, X. Zhang, N. Li, Ind. Eng. Chem. Res. 59, 7367 (2020)
X.-Y. Zhou, X. Chen, Tetrahedron Lett. 61, 151447 (2020)
M. Vilches-Herrera, S. Werkmeister, K. Junge, A. Börner, M. Beller, Catal. Sci. Technol. 4, 629 (2014)
K. Lévay, K.D. Tóth, T. Kárpáti, L. Hegedűs, ACS Omega 5, 5487 (2020)
R.R. Thakore, B.S. Takale, G. Casotti, E.S. Gao, H.S. Jin, B.H. Lipshutz, Org. Lett. 22, 6324 (2020)
Y. Wu, Y. Zhao, H. Wang, B. Yu, X. Yu, H. Zhang, Z. Liu, Ind. Eng. Chem. Res. 58, 6333 (2019)
V. Goyal, J. Gahtori, A. Narani, P. Gupta, A. Bordoloi, K. Natte, J. Org. Chem. 84, 15389 (2019)
W. Liang, F. Xie, Z. Yang, Z. Zhang, C. Xia, Y. Li, Z. Zhu, X. Chen, Org. Lett. 22, 8291 (2020)
Y. Yua, B. Chen, Y. Zhang, X.-F. Wu, J. Org. Chem. 85, 5733 (2020)
A. Punzi, N. Zappimbulso, G.M. Farinola, Eur. J. Org. Chem. 2020, 3229 (2020)
R. Vankateshwarlu, S.N. Singh, V. Siddaiah, H. Ramamohan, R. Dandela, M. Pal, Tetrahedron Lett. 60, 151326 (2019)
S. Mao, X. Shi, J.-F. Soulé, H. Doucet, Eur. J. Org. Chem. 2020, 91 (2020)
H.-J. Ai, R. Franke, X.-F. Wu, Mol. Catal. 493, 111043 (2020)
F.M. Harvey, C.G. Bochet, J. Org. Chem. 11, 7611 (2020)
S. Takayama, T. Yatabe, Y. Koizumi, X. Jin, K. Nozaki, M. Mizuno, K. Yamaguchi, Chem. Sci. 11, 4074 (2020)
I.P. Andrews, R.J. Atkins, G.F. Breen, J.S. Carey, M.A. Forth, D.O. Morgan, A. Shamji, A.C. Share, S.A.C. Smith, T.C. Walsgrove, A.S. Wells, Org. Process Res. Dev. 7, 655 (2003)
K.S. Reddy, N. Srinivasan, C.R. Reddy, N. Kolla, Y. Anjaneyulu, S. Venkatraman, A. Bhattacharya, V.T. Mathad, Org. Process Res. Dev. 11, 81 (2007)
F.M. Adam, G. Bish, F. Calo, C.L. Carr, N. Castro, D. Hay, P.B. Hodgson, P. Jones, C.J. Knight, M. Paradowski, G.C. Parsons, K.J.W. Proctor, D.C. Pryde, F. Rota, M.C. Smith, T.-D. Tran, J. Hitchin, R. Dixon, Org. Process Res. Dev. 15, 788 (2011)
C. Luo, Q. Xu, C. Huang, L. Luo, J. Zhu, R. Zhang, G. Huang, D. Yin, Org. Process Res. Dev. 24, 1024 (2020)
X. Liu, D. Astruc, Adv. Synth. Catal. 360, 3426 (2018)
H. Mousavi, B. Zeynizadeh, R. Younesi, M. Esmati, Aust. J. Chem. 71, 595 (2018)
B. Zeynizadeh, R. Younesi, H. Mousavi, Res. Chem. Intermed. 44, 7331 (2018)
B. Zenizadeh, F. Mohammad Aminzadeh, H. Mousavi, Res. Chem. Intermed. 45, 3329 (2019)
B. Zeynizadeh, H. Mousavi, S. Zarrin, J. Chin. Chem. Soc. 66, 928 (2019)
B. Zeynizadeh, F. Mohammad Aminzadeh, H. Mousavi, Green Process. Synth. 8, 742 (2019)
B. Zeynizadeh, F. Sepehraddin, H. Mousavi, Ind. Eng. Chem. Res. 58, 16379 (2019)
R. Bakhshi, B. Zeynizadeh, H. Mousavi, J. Chin. Chem. Soc. 67, 623 (2020)
B. Zeynizadeh, H. Mousavi, F. Sepehraddin, Res. Chem. Intermed. 46, 3361 (2020)
I.D. Entwistle, A.E. Jackson, R.A.W. Johnstone, R.P. Telford, J. Chem. Soc. Perkin Trans. 1433 (1977)
S. Ram, R.E. Ehrenkaufer, Tetrahedron Lett. 25, 3415 (1984)
M. Li, M.E. Johnson, Tetrahedron Lett. 35, 6255 (1994)
J.W. Bae, Y.J. Cho, S.H. Lee, C.M. Yoon, Tetrahedron Lett. 41, 175 (2000)
D.C. Gowda, S. Gowda, Indian J. Chem. 39B, 709 (2000)
H. Berthold, T. Schotten, H. Hönig, Synlett. 1607 (2002)
K. Abiraj, G.R. Srinivasa, D.C. Gowda, Synth. Commun. 35, 223 (2005)
G.S. Vanier, Synlett. 131 (2007)
J.F. Quinn, C.E. Bryant, K.C. Golden, B.T. Gregg, Tetrahedron Lett. 51, 786 (2010)
M. Baron, E. Métay, M. Lemaire, F. Popwycz, Green Chem. 15, 2013 (2013)
F. Li, B. Frett, H.-Y. Li, Synlett. 1403 (2014)
Y. Zhou, H. Zhou, S. Liu, D. Pi, G. Shen, Tetrahedron 73, 3898 (2017)
Y. Taskesenligil, F. Lafzi, H. Kilic, N. Saracoglu, J. Heterocyclic Chem. 56, 3289 (2019)
A. Cheddie, S.A. Shintre, A. Bantho, C. Mocktar, N.A. Koorbanally, J. Heterocyclic Chem. 57, 299 (2020)
W.B. Smith, J. Heterocyclic Chem. 24, 745 (1987)
M. Petrini, R. Ballini, G. Rosini, Synthesis. 713 (1987)
T. Yoshida, S. Kawamura, K. Nakata, Tetrahedron Lett. 58, 1181 (2017)
F. Piazzolla, A. Temperini, Tetrahedron Lett. 59, 2615 (2018)
J.S. Yadav, A.V. Narsaiah, A.K. Basak, P.R. Goud, D. Sreenu, K. Nagaiah, J. Mol. Catal. A Chem. 255, 78 (2006)
S. Farhadi, M. Zaidi, J. Mol. Catal. A Chem. 299, 18 (2009)
N. Ghaffari Khaligh, J. Mol. Catal. A Chem. 363–364, 90 (2012)
S. Farhadi, K. Jahanara, Chin. J. Catal. 35, 368 (2014)
U.P. Saikia, F.L. Hussain, M. Suri, P. Pahari, Tetrahedron Lett. 57, 1158 (2016)
H. Veisi, S. Vafajoo, K. Bahrami, B. Mozafari, Catal. Lett. 148, 2734 (2018)
S.K. Warkhade, V. Cahurasiya, M. Rawat, G.S. Gaikwad, S.P. Zodape, U.R. Pratap, A.V. Wankhude, ChemistrySelect 3, 2515 (2018)
S. Mahdian, M.R. Naimi-Jamal, L. Panahi, ChemistrySelect 3, 11223 (2018)
H. Veisi, A. Nikseresht, A. Rostami, S. Hemmati, Res. Chem. Intermed. 45, 597 (2019)
E. Yazdani, M. Kazemi Miraki, A. Salamatmanesh, J. Azarnia, K. Azizi, L. Ghandi, A. Heydari, Res. Chem. Intermed. 45, 1775 (2019)
M. Rimaz, H. Mousavi, Turk. J. Chem. 37, 252 (2013)
S. Kamalifar, H. Kiyani, Res. Chem. Intermed. 45, 5975 (2019)
M. Rimaz, B. Khalili, G. Khatyal, H. Mousavi, F. Aali, Aust. J. Chem. 70, 1274 (2017)
M. Rimaz, H. Mousavi, L. Nikpey, B. Khalili, Res. Chem. Intermed. 43, 3925 (2017)
M. Rimaz, H. Mousavi, B. Khalili, F. Aali, J. Chin. Chem. Soc. 65, 1389 (2018)
M. Rimaz, H. Mousavi, B. Khalili, L. Sarvari, J. Iran. Chem. Soc. 16, 1687 (2019)
M. Rimaz, H. Mousavi, L. Ozzar, B. Khalili, Res. Chem. Intermed. 45, 2673 (2019)
A. Ahmad, S.N.A. Shah, M. Arshad, F. Bélanger-Gariepy, E.R.T. Tiekink, Z.u. Rehman, Appl. Organomet. Chem. 35, e6065 (2021)
S. Majhi, K. Sharma, R. Singh, M. Ali, C.S.P. Tripathi, D. Guin, ChemistrySelect 5, 12365 (2020)
A.H.T.N. Sorenson, Y. Wu, E.K. Orcutt, R.V. Kent, H.C. Anderson, A.J. Matzger, K.J. Stowers, J. Mater. Chem. A 8, 15066 (2020)
B.J. Borah, P. Bharali, Appl. Organomet. Chem. 34, e5753 (2020)
S.V. Kite, D.J. Sathe, A.N. Kadam, S.S. Chavan, K.M. Garadkar, Res. Chem. Intermed. 46, 1255 (2020)
Acknowledgements
We are thankful to the Research Council of Urmia University for the partial support of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zeynizadeh, B., Mohammad Aminzadeh, F. & Mousavi, H. Chemoselective reduction of nitroarenes, N-acetylation of arylamines, and one-pot reductive acetylation of nitroarenes using carbon-supported palladium catalytic system in water. Res Chem Intermed 47, 3289–3312 (2021). https://doi.org/10.1007/s11164-021-04469-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04469-9