Abstract
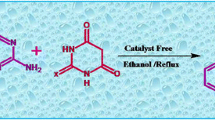
We describe a novel and simple regioselective synthesis of 5-aryl-3-methyl-1-phenyl-1,2-dihydro-7aH-pyrazolo[3,4-c]pyridazin-7a-ol derivatives via one-pot three-component reaction of arylglyoxalmonohydrates, 5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one, and hydrazine hydrate in presence of 1,4-diazabicyclo[2.2.2]octane (DABCO) as base-organocatalyst at room temperature in acetonitrile. This one-pot method has the advantages of simple methodology, high atom economy, cost-effectiveness, high regioselectivity, and easy workup.
Graphical Abstract






Similar content being viewed by others
References
N.G. Shabalala, S. Maddila, S.B. Jonnalagadda, Res. Chem. Intermed. 42, 8097 (2016)
S. Jiang, J. Gao, L. Han, Res. Chem. Intermed. 42, 1017 (2016)
S.S. Sajadikhah, M.T. Maghsoodlou, N. Hazeri, S. Mohamadian-Souri, Res. Chem. Intermed. 42, 2805 (2016)
A. Farhadi, J. Noei, R.H. Aliyari, M. Albakhtiyari, M.A. Takassi, Res. Chem. Intermed. 42, 1401 (2016)
M. Rimaz, H. Mousavi, P. Keshavarz, B. Khalili, Curr. Chem. Lett. 4, 159 (2015)
M. Abbasi, Res. Chem. Intermed. 42, 3303 (2016)
B. Eftekhari-Sis, M. Zirak, A. Akbari, Chem. Rev. 113, 2958 (2013)
B. Khalili, P. Jajarmi, B. Eftekhari-Sis, M.M. Hashemi, J. Org. Chem. 73, 2090 (2008)
M. Rimaz, H. Mousavi, M. Behnam, L. Sarvari, B. Khalili, Curr. Chem. Lett. (2017). doi:10.5267/j.ccl.2016.12.001
P.H. Yang, Res. Chem. Intermed. 42, 5617 (2016)
S.S. Wang, Q.W. Zhu, S. Liu, Y. Yang, Z.T. Wang, B. Jiang, S.J. Tu, Res. Chem. Intermed. 41, 2879 (2015)
S.A. Higgins, Chem. Soc. Rev. 26, 247 (1997)
K. Ogawa, R.C. Rasmussen, J. Org. Chem. 68, 2921 (2003)
A.T. Khan, M. Lal, S. Ali, M.M. Khan, Tetrahedron Lett. 52, 5327 (2011)
Z.-L. Shen, S.-J. Ji, T.-P. Loh, Tetrahedron Lett. 46, 507 (2005)
F. Xiao, Y. Liu, J. Wang, Tetrahedron Lett. 48, 1147 (2007)
W.A. Hermann, Angew. Chem. Int. Ed. 41, 1290 (2002)
H. Kefayati, S. Jirsaray Bazargard, P. Vejdansefat, S. Shariati, A. Mehtar Kohankar, Dyes Pigments 125, 309 (2016)
G. Ge, P. Dai, Z. Lu, M. Liang, H. Dong, Z. Sun, S. Xue, Dyes Pigments 128, 8 (2016)
G. Hussain, M. Ather, M.U.A. Khan, A. Saeed, R. Saleem, G. Shabir, P.A. Channar, Dyes Pigments 130, 90 (2016)
K. Abouzid, M.A. Hakeem, O. Khalil, Y. Maklad, Bioorg. Med. Chem. 16, 382 (2008)
D.W. Combs, M.S. Rampulla, S.C. Bell, D.H. Klaubert, A.J. Tobia, R. Falotico, B. Haertlein, C.L. Weiss, J.B. Moore, J. Med. Chem. 22, 380 (1990)
E. Sotelo, A. Coelho, E. Ravina, Tetrahedron Lett. 44, 4459 (2003)
M. Rimaz, H. Mousavi, M. Behnam, B. Khalili, Curr. Chem. Lett. 5, 145 (2016)
S. Fustero, M. Sánchez-roselló, P. Barrio, A. Simón-Fuentes, Chem. Rev. 111, 6948 (2011)
R.S. Keri, K. Chand, T. Ramakrishnappa, B.M. Nagaraja, Arch. Pharm. Chem. Life Sci. 384, 299 (2015)
M.-M. Zhang, W. Wang, T.-J. Li, C.-S. Yao, X.-S. Wang, Res. Chem. Intermed. 39, 1781 (2013)
K. Shekarrao, P.P. Kaishap, V. Saddanapu, A. Addlagatta, S. Gogoi, R.C. Boruah, RSC Adv. 4, 24001 (2014)
J.-Y. Kato, H. Aoyama, T. Yokomatsu, Org. Biomol. Chem. 11, 1171 (2013)
S. Schenone, M. Radi, F. Musumeci, C. Brullo, M. Botta, Chem. Rev. 114, 7189 (2014)
M. Kangani, N. Hazeri, M.T. Mghsoodlou, S.M. Habibi-Khorasani, S. Salahi, Res. Chem. Intermed. 41, 2513 (2015)
M. Alam, M.S. Zaman, M.M. Alam, K. Arora, A. Ahmad, A. Husain, Int. J. Curr. Pharm. Sci. 1, 159 (2015)
P.G. Baraldi, S. Manfredini, R. Romagnoli, L. Stevanato, A.N. Zaid, R. Manservigi, Nucleos. Nucleot. 17, 2165 (1998)
O.I. El-Sabbagh, S. Mostafa, H.A. Abdel-Aziz, H.S. Ibrahim, M.M. Elaasser, Arch. Pharm. Chem. Life Sci. 3346, 1 (2013)
A.K. Tewari, A. Mishra, Bioorg. Med. Chem. 9, 715 (2001)
A.K. Tewari, R. Dubey, A. Mishra, Med. Chem. Res. 20, 125 (2011)
R.K. Tonk, S. Bawa, G. Chawla, G.S. Deora, S. Kumar, V. Rathore, N. Mulakayala, A. Rajaram, A.M. Kalle, O. Afzal, Eur. J. Med. Chem. 57, 176 (2012)
M.F. Mady, T.S. Saleh, A.A. El-Ketab, N.M.A. El-Rahman, S.I.A. El-Moez, Res. Chem. Intermed. 42, 753 (2016)
M. Hosny, F. El-Mariah, Phosphorus Sulfur Silicon Relat. Elem. 182, 1475 (2007)
M.F. Braña, M. Cacho, M.L. García, E.P. Moyral, B. López, B. de Pascual-Teresa, A. Ramos, N. Acero, F. Llinares, D. Muñoz-Mingarro, O. Lozach, L. Meijer, J. Med. Chem. 48, 6843 (2005)
M.P. Giovannoni, C. Vergelli, C. Biancalani, N. Cesari, A. Greziano, P. Biagini, J. Gracia, A. Gavalà, V. Dal Piaz, J. Med. Chem. 49, 5363 (2006)
J. Feixas, M.P. Giovannoni, C. Vergelli, A. Gavaldà, N. Cesari, A. Graziano, V. Dal Piaz, Bioorg. Med. Chem. Lett. 15, 2381 (2005)
J. Witherington, V. Bordas, D. Haigh, D.M.B. Hickey, R.J. Ife, A.D. Rawlings, B.P. Slingsby, D.G. Smith, R.W. Ward, Bioorg. Med. Chem. Lett. 13, 1581 (2003)
A. Deeb, E. Mourad, D. Elenany, Phosphorus Sulfur Silicon Relat. Elem. 185, 222 (2010)
M.B. Teimouri, F. Mansouri, R. Bazhrang, Tetrahedron 66, 259 (2010)
E.A. El-Rady, M.A. Barsy, J. Heterocyclic Chem. 45, 1233 (2008)
M. Rimaz, H. Rabiei, B. Khalili, R.H. Prager, Aust. J. Chem. 67, 283 (2014)
M. Rimaz, A. Mirshokraie, B. Khalili, P. Motiee, Arkivoc v, 88 (2015)
M. Rimaz, Z. Jalalian, H. Mousavi, R.H. Prager, Tetrahedron Lett. 57, 105 (2016)
M. Rimaz, Z. Jalalian, H. Mousavi, Res. Chem. Intermed. 42, 8185 (2016)
M. Rimaz, H. Mousavi, Turk. J. Chem. 37, 252 (2013)
M. Rimaz, P. Pourhossein, B. Khalili, Turk. J. Chem. 39, 244 (2015)
M. Rimaz, Aust. J. Chem. 68, 1529 (2015)
M. Rimaz, F. Aali, Chin. J. Catal. 37, 517 (2016)
M. Rimaz, F. Aali, B. Khalili, R.H. Prager, Aust. J. Chem. (2016). doi:10.1071/CH16364
H.A. Riley, A.R. Gray, Organic Syntheses, Collect, vol. II (Wiley, New York, 1943), p. 509
O.A. Attanasi, G. Favi, P. Filippone, F.R. Perrulli, S. Santeusanio, Org. Lett. 11, 309 (2009)
A.J. Turbiaak, J.W. Kampf, H.D.H. Showalter, Tetrahedron Lett. 51, 1326 (2010)
H. Mei, S.A. Ingale, F. Seela, Tetrahedron 71, 6170 (2015)
S. Roscales, A. Sancho, A.G. Csákÿ, Synthesis 47, 2233 (2015)
H. Behbahani, H.M. Ibrahim, RSC Adv. 5, 89226 (2015)
Y. Zhong, S. Ma, B. Li, X. Jiang, R. Wang, J. Org. Chem. 80, 6870 (2015)
M.S. Abaee, S. Cheraghi, Turk. J. Chem. 38, 650 (2014)
G. Cai, S. Liu, J. Zhang, Y. Ren, H. Wang, Z. Miao, Synth. Commun. 46, 793 (2016)
Y.-Y. Liu, S.-W. Duan, R. Zhang, Y.-H. Liu, J.-R. Chen, W.-J. Xiao, Org. Biomol. Chem. 14, 5224 (2016)
Acknowledgements
Financial support from the Research Council of Payame Noor University is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rimaz, M., Mousavi, H., Nikpey, L. et al. Novel and convenient one-pot strategy for regioselective synthesis of new 5-aryl-3-methyl-1-phenyl-1,2-dihydro-7aH-pyrazolo[3,4-c]pyridazin-7a-ol derivatives. Res Chem Intermed 43, 3925–3937 (2017). https://doi.org/10.1007/s11164-016-2848-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2848-5