Abstract
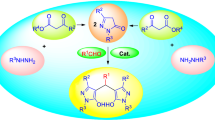
4-Sulfophthalic acid (4-H3SPA) solution 50 wt% in H2O has been effectively catalyzed the synthesis of a series of biologically relevant bis(indolyl)methanes by the electrophilic substitution of indole derivatives on aldehyde compounds and 4,4′-(arylmethylene)-bis(3-methyl-1-phenyl-1H-pyrazol-5-ol)s by condensing 5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one with various aldehydes under aqua conditions at room temperature. 3,3′-(Arylmethylene)-bis-4-hydroxycoumarins have also been synthesized in the presence of 0.1 mL (0.262 mmol) of 4-H3SPA solution 50 wt% in H2O at 80 °C. The procedure is simple and the expected bis-heterocyclic compounds were isolated in good to excellent yields. The present protocol provides the benefits of convenience, mild reaction conditions, eco-friendliness, and no use of hazardous organic solvents.


Similar content being viewed by others
References
M. Shiri, M.A. Zolfigol, H.G. Kruger, Z. Tanbakouchian, Chem. Rev. 110, 2250 (2010)
P.P. Kaishap, C. Dohutia, Int. J. Pharm. Sci. Res. 4, 1312 (2013)
S. Sarva, J.S. Harinath, S.P. Sthanikam, S. Ethiraj, M. Vaithiyalingam, S.R. Cirandur, Chin. Chem. Lett. 27, 16 (2016)
G. Sivaprasad, P.T. Perumal, V.R. Prabavathy, N. Mathivanan, Bioorg. Med. Chem. Lett. 16, 6302 (2006)
K. Sujatha, P.T. Perumal, D. Muralidharan, M. Rajendran, Indian J. Chem. 48B, 267 (2009)
T. Osawa, M. Namiki, Tetrahedron Lett. 24, 4719 (1983)
K.V. Sashidhara, A. Kumar, M. Kumar, A. Srivastava, A. Puri, Bioorg. Med. Chem. Lett. 20, 6504 (2010)
M. Mari, A. Tassoni, S. Lucarini, M. Fanelli, G. Piersanti, G. Spadoni, Eur. J. Org. Chem. 2014, 3822 (2014)
Y. Gong, G.L. Firestone, L.F. Bjeldanes, Mol. Pharmacol. 69, 1320 (2006)
J. Povszasz, G.P. Katalin, S. Foleat, B. Malkovics, Acta Phys. Acad. Sci. Hung. 29, 299 (1996)
S.B. Bharate, J.B. Bharate, S.I. Khan, B.L. Tekwani, M.R. Jacob, R. Mudududdla, R.R. Yadav, B. Singh, P.R. Sharma, S. Maity, B. Singh, I.A. Khan, R.A. Vishwakarma, Eur. J. Med. Chem. 63, 435 (2013)
M. Kobayashi, S. Aoki, K. Gato, K. Matsunami, M. Kurosu, I. Kitagawa, Chem. Pharm. Bull. 42, 2449 (1994)
C. Bonnesen, I.M. Eggleston, J.D. Hayes, Cancer Res. 61, 6120 (2001)
R. Martinez, A. Espinosa, A. Tarraga, P. Molina, Tetrahedron 64, 2184 (2008)
R. Pegu, R. Mandal, A.K. Guha, S. Pratihar, New J. Chem. 39, 5984 (2015)
X. He, S. Hu, K. Liu, Y. Guo, J. Xu, S. Shao, Org. Lett. 8, 333 (2006)
D. Sain, C. Kumari, A. Kumar, S. Dey, Supramol. Chem. 28, 239 (2016)
A. Khorshidi, N. Mardazad, Z. Shaabanzadeh, Tetrahedron Lett. 55, 3873 (2014)
R. Surasani, D. Kalita, K.B. Chandrasekhar, Green Chem. Lett. Rev. 6, 113 (2013)
A. Ganesan, J. Kothandapani, J.B. Nanubolu, S.S. Ganesan, RSC Adv. 5, 28597 (2015)
A.K. Mallik, R. Pal, C. Guha, H. Mallik, Green Chem. Lett. Rev. 5, 321 (2012)
M. El-Sayed, K. Mahmoud, A. Hilgeroth, Curr. Chem. Lett. 3, 7 (2014)
H. Veisi, R. Gholbedaghi, J. Malakootikhah, A. Sedrpoushan, B. Maleki, D. Kordestani, J. Heterocycl. Chem. 47, 1398 (2010)
K. Karthikeyan, G. Sivaprasad, Org. Prep. Proced. Int. 47, 449 (2015)
D. Sun, G. Jiang, Z. Xie, Z. Le, Chin. J. Chem. 33, 409 (2015)
Z.B. Xie, D.Z. Sun, G.F. Jiang, Z.G. Le, Molecules 19, 19665 (2014)
K. Ravi, B. Krishnakumar, M. Swaminathan, Res. Chem. Intermed. 41, 5353 (2015)
A.V. Reddy, K. Ravinder, V.L.N. Reddy, T.V. Goud, V. Ravikanth, Y. Venkateswarlu, Synth. Commun. 33, 3687 (2003)
J.S. Yadav, M.K. Gupta, R. Jain, N.N. Yadav, B.V.S. Reddy, Monatsh. Chem. 141, 1001 (2010)
B.Y. Giri, B.L.A.P. Devi, K. Vijayalakshmi, R.B.N. Prasad, N. Lingaiah, P.S.S. Prasad, Indian J. Chem. 51B, 1731 (2012)
M.M. Meshram, N.N. Rao, P.B. Thakur, B.C. Reddy, P. Ramesh, Indian J. Chem. 52B, 814 (2013)
R. Vaid, M. Gupta, O.S. Chambyal, R. Gupta, J. Chem. Sci. 127, 987 (2015)
L.Z. Fekri, M. Nikpassand, M. Kohansal, Rus. J. Gen. Chem. 85, 2861 (2015)
D. Talukdar, A.J. Thakur, Green Chem. Lett. Rev. 6, 55 (2013)
D. Liang, W. Huang, L. Yuan, Y. Ma, J. Ma, D. Ning, Catal. Commun. 55, 11 (2014)
N.C. Ganguly, P. Mondal, S.K. Barik, Green Chem. Lett. Rev. 5, 73 (2012)
S.J. Ji, M.F. Zhou, D.G. Gu, Z.Q. Jiang, T.P. Loh, Eur. J. Org. Chem. 1584, 37 (2004)
C.C. Silveira, S.R. Mendes, M.A. Villetti, D.F. Back, T.S. Kaufman, Green Chem. 14, 2912 (2012)
A.C. Shaikh, C. Chen, J. Chin. Chem. Soc. 58, 899 (2011)
A. Swetha, B.M. Babu, H.M. Meshram, Tetrahedron Lett. 56, 1775 (2015)
M. Auria, Tetrahedron 47, 9225 (1991)
M. Kalantari, Arab. J. Chem. 5, 319 (2012)
S.A.R. Mulla, A. Sudalai, M.Y. Pathan, S.A. Siddique, S.M. Inamdar, S.S. Chavan, R.S. Reddy, RSC Adv. 2, 3525 (2012)
E.L. Armstrong, H.K. Grover, M.A. Kerr, J. Org. Chem. 78, 10534 (2013)
S. Handy, N.M. Westbrook, Tetrahedron Lett. 55, 4969 (2014)
P. Rammohan, Indian J. Chem. 53B, 763 (2014)
P. Rammohan, Int. J. Org. Chem. 3, 136 (2013)
K. Ghodrati, S.H. Hosseini, R. Mosaedi, C. Karami, F. Maleki, A. Farrokhi, Z. Hamidi, Int. Nano Lett. 3, 13 (2013)
R. Pegu, K.J. Majumdar, D.J. Talukdar, S. Pratihar, RSC Adv. 4, 33446 (2014)
A. Shaabani, R. Afshari, S.E. Hooshmand, A.T. Tabatabaei, F. Hajishaabanha, RSC Adv. 6, 18113 (2016)
D.A. Reddy, J. Choi, S. Lee, R. Ma, T.K. Kim, RSC Adv. 5, 67394 (2015)
S. Sobhani, R. Jahanshahi, New J. Chem. 37, 1009 (2013)
H. Mahmoudi, A.A. Jafari, S. Saeedi, H. Firouzabadi, RSC Adv. 5, 3023 (2015)
M. Kour, S. Paul, New J. Chem. 39, 6338 (2015)
P.K. Chhattise, S.S. Arbuj, K.C. Mohite, S.V. Bhavsar, A.S. Horne, K.N. Handore, V.V. Chabukswar, RSC Adv. 4, 28623 (2014)
B. Sadeghi, F. Amiri-Tavasoli, A. Hassanabadi, Synth. React. Inorg. Met.Org. Nano Met. Chem. 45, 1396 (2015)
D.W. Zhang, Y.M. Zhang, Y.L. Zhang, T.Q. Zhao, H.W. Liu, Y.M. Gan, Q. Gu, Chem. Pap. 69, 470 (2015)
M. Zahran, Y. Abdin, H. Salama, Arkivoc xi, 256 (2008)
S.R. Mendes, S. Thurow, F. Penteado, M. da Silva, R.A. Gariani, G. Perinb, E.J. Lenardão, Green Chem. 17, 4334 (2015)
M. Imam Uddin, J.R. Buck, M.L. Schulte, D. Tang, S.A. Saleh, Y.Y. Cheung, J. Harp, H.C. Manning, Tetrahedron Lett. 55, 169 (2014)
S.S. Sonar, S.A. Sadaphal, A.H. Kategaonkar, R.U. Pokalwar, B.B. Shingate, M.S. Shingare, Bull. Korean Chem. Soc. 30, 825 (2009)
A.P.G. Nikalje, S.I. Shaikh, World J. Pharm. Pharmaceut. Sci. 3, 1282 (2014)
J.T. Li, M.X. Sun, G.Y. He, X.Y. Xu, Ultrasound Sonochem. 18, 412 (2011)
F. Shirini, N.G. Khaligh, Chin. J. Catal. 34, 1890 (2013)
K. Niknam, D. Saberi, M. Baghernejad, Phosphor. Sulfur Silicon Relat. Elem. 185, 875 (2010)
S.M. Baghbanian, Y. Babajani, H. Tashakorian, S. Khaksar, M. Farhang, C. R. Chimie 16, 129 (2013)
F. Shirini, N.G. Khaligh, O.G. Jolodar, Dyes Pigments 98, 290 (2013)
H. Alinezhad, A.H. Haghighi, F. Salehian, Chin. Chem. Lett. 21, 183 (2010)
A. Zare, F. Bahrami, M. Merajoddin, M. Bandari, A.R. Moosavi-Zare, M.A. Zolfigol, A. Hasaninejad, M. Shekouhy, M.H. Beyzavi, V. Khakyzadeh, M. Mokhlesi, Z. Asgari, Org. Prep. Proced. Int. 45, 211 (2013)
S.A. Sadaphal, S.S. Sonar, M.N. Ware, M.S. Shingare, Green Chem. Lett. Rev. 1, 191 (2008)
M. Seddighi, F. Shirini, M. Mamaghani, RSC Adv. 3, 24046 (2013)
V.J. Rani, K.V. Vani, C.V. Rao, Synth. Commun. 42, 2048 (2012)
W.J. Li, X.F. Lin, J. Wang, G.L. Li, Y.G. Wang, Synth. Commun. 35, 2765 (2005)
M. Kidwai, R. Chauhan, D. Bhatnagar, Arab. J. Chem. (2014). doi:10.1016/j.arabjc.2014.05.009
S.S. Ekbote, K.M. Deshmukh, Z.S. Qureshi, B.M. Bhanage, Green Chem. Lett. Rev. 4, 177 (2011)
S.R. Sheng, Q.Y. Wang, Y. Ding, X.L. Liu, M.Z. Cai, Catal. Lett. 128, 418 (2009)
K.L. Dhumaskar, S.G. Tilve, Green Chem. Lett. Rev. 5, 353 (2012)
V.D. Patil, G.B. Dere, P.A. Rege, J.J. Patil, Synth. Commun. 41, 736 (2011)
A. Kundu, A. Ganguly, K. Dhara, P. Patra, N. Guchhait, RSC Adv. 5, 53220 (2015)
D.K. Sharma, A. Hussain, M.R. Lambu, S.K. Yousuf, S. Maiety, B. Singh, D. Mukherjee, RSC Adv. 3, 2211 (2013)
N. Baig, G.M. Shelke, A. Kumar, A.K. Sah, Catal. Lett. 146, 333 (2016)
N. Seyedi, M. Kalantari, J. Sci. I. R. Iran 24, 205 (2013)
H. Hikawa, Y. Yokoyama, RSC Adv. 3, 1061 (2013)
N. Gupta, D. Goyal, Chem. Heterocyl. Compd. 51, 4 (2015)
S. Sugiura, S. Ohno, O. Ohtani, K. Izumi, T. Kitamikado, H. Asai, K. Kato, J. Med. Chem. 20, 80 (1977)
L.C. Behr, R. Fusco, C.H. Jarboe, in The Chemistry of Heterocyclic Compounds, Pyrazoles, Pyrazolines, Pyrazolidines, Indazoles and Condensed Rings, ed. by A. Weissberger (Interscience Publishers, New York, 1967)
C.E. Rosiere, M.I. Grossman, Science 113, 651 (1951)
D.M. Bailey, P.E. Hansen, A.G. Hlavac, E.R. Baizman, J. Pearl, A.F. Defelice, M.E. Feigenson, J. Med. Chem. 28, 256 (1985)
R.N. Mahajan, F.H. Havaldar, P.S. Fernandes, J. Indian Chem. Soc. 68, 245 (1991)
P.M.S. Chauhan, S. Singh, R.K. Chatterjee, Indian J. Chem. Sect B: Org. Chem. Incl. Med. Chem. 32, 858 (1993)
K. Sujatha, G. Shanthi, N.P. Selvam, S. Manoharan, P.T. Perumal, M. Rajendran, Bioorg. Med. Chem. Lett. 19, 4501 (2009)
X. Yang, P. Zhang, Y. Zhou, J. Wang, H. Liu, Chin. J. Chem. 30, 670 (2012)
N. Das, A. Verma, P.K. Shrivastava, S.K. Shrivastava, Indian J. Chem. 47B, 1555 (2008)
D. Singh, D. Singh, J. Indian Chem. Soc. 68, 165 (1991)
M. Londershausen, Pestic. Sci. 48, 269 (1996)
H.A. Lubs (ed.), The Chemistry of Synthetic Dyes and Pigments (American Chemical Society, Washington, DC, 1970)
A.D. Garnovskii, A.I. Uraev, V.I. Minkin, Arkivoc iii, 29 (2004)
M. Abbasi-Tarighat, E. Shahbazi, K. Niknam, Food Chem. 138, 991 (2013)
S. Tayebi, M. Baghrnejad, D. Saberi, K. Niknam, Chin. J. Catal. 32, 1477 (2011)
Z. Karimi-Jaberi, B. Pooladian, M. Moradi, E. Ghasemi, Chin. J. Catal. 33, 1945 (2012)
E. Mosaddegh, M.R. Islami, Z. Shojaie, Arab. J. Chem. (2013). doi:10.1016/j.arabjc.2013.02.016
Z. Zhou, Y. Zhang, Green Chem. Lett. Rev. 7, 18 (2014)
J. Safaei-Ghomi, B. Khojastehbakht-Koopaei, S. Zahedi, Chem. Heterocycl. Compd. 51, 34 (2015)
K. Eskandari, B. Karami, S. Khodabakhshi, M. Farahi, Lett. Org. Chem. 12, 38 (2015)
K.M. Khan, M.T. Muhammad, I. Khan, S. Perveen, W. Voelter, Monatsh. Chem. 146, 1587 (2015)
H. Zang, Q. Su, Y. Mo, B. Cheng, Ultrason. Sonochem. 18, 68 (2011)
K. Niknam, S. Mirzaee, Synth. Commun. 41, 2403 (2011)
J. Safaei-Ghomi, B. Khojastehbakht-Koopaei, H. Shahbazi-Alavi, RSC Adv. 4, 46106 (2014)
C. Yang, L. Pang, A. Wang, Asian J. Chem. 23, 749 (2011)
A. Vafaee, A. Davoodnia, M. Pordel, Res. Chem. Intermed. 41, 8242 (2015)
K.R. Phatangare, V.S. Padalkar, V.D. Gupta, V.S. Patil, P.G. Umape, N. Sekar, Synth. Commun. 42, 1349 (2012)
S. Sobhani, Z. Pakdin-Parizi, R. Nasseri, J. Chem. Sci. 125, 975 (2013)
N.G. Khaligh, S.B.A. Hamid, S.J.J. Titinchi, Chin. Chem. Lett. 27, 104 (2016)
M.N. Elinson, O.O. Sokolova, R.F. Nasybullin, Heterocycl. Commun. 21, 97 (2015)
A.D. Gupta, R. Pal, A.K. Mallik, Green Chem. Lett. Rev. 7, 404 (2014)
M.A. Gouda, J. Heterocycl. Chem. (2015). doi:10.1002/jhet.2313
D. Qu, J. Li, X.H. Yang, Z.D. Zhang, X.X. Luo, M.K. Li, X. Li, Molecules 19, 19868 (2014)
H. Zhao, N. Neamati, Y. Pommier, T.R. Burke Jr., Heterocycles 45, 2277 (1997)
I. Manolov, C. Maichle-Moessmer, I. Nicolova, N. Danchev, Arch. Pharm. Chem. Life Sci. 339, 319 (2006)
K.M. Khan, S. Iqbal, M.A. Lodhi, G.M. Maharvi, M.I. Choudhary, Z. Ullah, A.U. Rahman, S. Perveen, Bioorg. Med. Chem. 12, 1963 (2004)
O. Talhi, M. Schnekenburger, J. Panning, D.G.C. Pinto, J.A. Fernandes, F.A. Almeida Paz, C. Jacob, M. Diederich, A.M. Silva, Bioorg. Med. Chem. 22, 3008 (2014)
K.M. Khan, F. Rahim, A. Wadood, N. Kosar, M. Taha, S. Lalani, A. Khan, M.I. Fakhri, M. Junaid, W. Rehman, M. Khan, S. Perveen, M. Sajid, M.I. Choudhary, Eur. J. Med. Chem. 81, 245 (2014)
A.K. Mahapatra, K. Maiti, P. Sahoo, P.K. Nandi, J. Lumin. 143, 349 (2013)
J. Li, Z. Hou, F. Li, Z.D. Zhang, Y. Zhou, X.X. Luo, M.K. Li, J. Mol. Struct. 1075, 509 (2014)
A.D. Gupta, S. Samanta, R. Mondal, A.K. Mallik, Bull. Korean Chem. Soc. 33, 4239 (2012)
H. Ammar, S. Abid, S. Fery-Forgues, Dyes Pigments 78, 1 (2008)
H. Mehrabi, H. Abusaidi, J. Iran. Chem. Soc. 7, 890 (2010)
F. Shirini, S. Esmaeeli-Ranjbar, M. Seddighi, Chin. J. Catal. 35, 1017 (2014)
S. Zahiri, M. Mokhtary, J. Taibah Univ. Sci. 9, 89 (2015)
R. Rezaei, F. Moezzi, M.M. Doroodmand, Chin. Chem. Lett. 25, 183 (2014)
Z. Karimi-Jaberi, M.R. Nazarifar, B. Pooladian, Chin. Chem. Lett. 23, 781 (2012)
B. Karmakar, A. Nayak, J. Banerji, Tetrahedron Lett. 53, 4343 (2012)
V. Padalkar, K. Phatangare, S. Takale, R. Pisal, A. Chaskar, J. Saudi Chem. Soc. 19, 42 (2015)
K. ParvanakBoroujeni, P. Ghasemi, Z. Rafienia, Monatsh. Chem. 145, 1023 (2014)
W. Li, Y. Wang, Z. Wang, L. Dai, Y. Wang, Catal. Lett. 141, 1651 (2011)
A. Tzani, A. Douka, A. Papadopoulos, E.A. Pavlatou, E. Voutsas, A. Detsi, A.C.S. Sustain, Chem. Eng. 1, 1180 (2013)
B. Sadeghi, T. Ziya, J. Chem. 2013, 5, Article ID 179013 (2013). doi:10.1155/2013/179013
A. Zhu, S. Bai, L. Li, M. Wang, J. Wang, Catal. Lett. 145, 1089 (2015)
Z.N. Siddiqui, F. Farooq, Catal. Sci. Technol. 1, 810 (2011)
J. Albadi, A. Mansournezhad, S. Salehnasab, Res. Chem. Intermed. 41, 5713 (2015)
R. Karimian, F. Piri, A. Safari, S.J. Davarpanah, J. Nanostruct. Chem. 3, 52 (2013)
J. Safaei-Ghomi, F. Eshteghal, M.A. Ghasemzadeh, Acta Chim. Slov. 61, 703 (2014)
Z. Karimi-Jaberi, M.R. Nazarifar, Eur. Chem. Bull. 3, 512 (2014)
A. Davoodnia, Bull. Korean Chem. Soc. 32, 4286 (2011)
F. Shirini, M. Abedini, S.A. Kiaroudi, Phosphorus Sulfur Silicon Relat. Elem. 189, 1279 (2014)
E. Sheikhhosseini, Trend. Mod. Chem. 3, 34 (2012)
S. Qadir, A.A. Dar, K.Z. Khan, Synth. Commun. 38, 3490 (2008)
A.D. Gupta, S. Samanta, R. Mondal, A.K. Malik, Chem. Sci. Trans. 2, 524 (2013)
G. Cravotto, G.M. Nano, G. Palmisano, S. Tagliapietra, Synthesis 2003, 1286 (2003)
A. Hasaninejed, M. Rasekhi Kazerooni, A. Zare, ACS Sustain Chem. Eng. 1, 679 (2013)
H. Hagiwara, N. Fujimoto, T. Suzuki, M. Ando, Heterocycles 53, 549 (2000)
W. Zhang, B. Cue (eds.), Green Techniques for Organic Synthesis and Medicinal Chemistry, 1st edn. (Wiley-VCH, New York, 2012)
Acknowledgments
Damghan University is acknowledged for provision of facilities and materials.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Banari, H., Kiyani, H. & Pourali, A. Efficient synthesis of bis(indolyl)methanes, bispyrazoles and biscoumarins using 4-sulfophthalic acid. Res Chem Intermed 43, 1635–1649 (2017). https://doi.org/10.1007/s11164-016-2720-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2720-7