Abstract
Background and aims
Iron (Fe) is an essential micronutrient for all higher organisms. Fe is sparingly available in calcareous soils and Fe deficiency is a major agricultural problem worldwide. Nicotianamine (NA) is a metal chelator involved in metal translocation in plants. Sweet potato is an attractive crop that can grow in poor soil and thus is useful for planting in uncultivated soil. In addition, the sweet potato has recently been suggested as a source of bioethanol. Our aim is to increase NA concentration in sweet potato to ameliorate Fe deficiency.
Method
Sweet potato plants expressing the barley NA synthase 1 (HvNAS1) gene under the control of CaMV 35S promoter were produced by Agrobacterium-mediated transformation.
Results
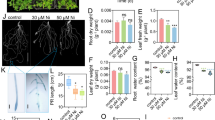
The transgenic sweet potato exhibited tolerance to low Fe availability when grown in calcareous soil. The level of tolerance to low Fe availability was positively correlated with the HvNAS1 expression level. The NA concentration of the transgenic sweet potato leaves was up to 7.9-fold greater than that of the non-transgenic (NT) plant leaves. Furthermore, the Fe and zinc concentrations were 3- and 2.9-fold greater, respectively, in transgenic sweet potato than in NT plant leaves.
Conclusions
Our results suggest that increasing the NA concentration of sweet potato by overexpression of HvNAS1 could significantly improve agricultural productivity and energy source.








Similar content being viewed by others
References
Akiyama K, Nakamura S, Suzuki T, Wisniewska SN, Kawasaki S (1997) Development of a system of rice transformation with long genome inserts for their functional analysis for positional cloning. Plant Cell Physiol 38:s94
Benes I, Schreiber K, Ripperger H, Kircheiss A (1983) Metal complex formation by nicotianamine, a possible phytosiderophore. Experientia 39:261–262
Conway G (2012) One billion hungry: can We feed the world? Cornell University Press 7:125–142
Eide D, Broderius M, Fett J, Guerinot ML (1996) A novel iron-regulated metal transporter from plants identified by functional expression in yeast. Proc Natl Acad Sci USA 93:5624–5628
Foresight The Future of Food and Farming: Final Project Report (2011) The Government office for science; p. 49–74. http://www.bis.gov.uk/assets/foresight/docs/food-and-farming/11-546-future-of-food-and-farming-report.pdf
Hell R, Stephan UW (2003) Iron uptake, trafficking and homeostasis in plants. Planta 216:541–551
Higuchi K, Nishizawa NK, Yamaguchi H, Römheld H, Marschner H, Mori S (1995) Response of nicotianamine synthase activity to Fe-deficiency in tobacco plants as compared with barley. J Exp Bot 46:1061–1063
Higuchi K, Suzuki K, Nakanishi H, Yamaguchi H, Nishizawa NK, Mori S (1999) Cloning of nicotianamine synthase genes, novel genes involved in the biosynthesis of phytosiderophores. Plant Physiol 119:471–480
Higuchi K, Watanabe S, Takahashi M, Kawasaki S, Nakanishi H, Nishizawa NK, Mori S (2001) Nicotianamine synthase gene expression differs in barley and rice under Fe-deficient conditions. Plant J 25:159–167
Ishimaru Y, Kim S, Tsukamoto T, Oki H, Kobayashi T, Watanabe S, Matsuhashi S, Takahashi M, Nakanishi H, Mori S, Nishizawa NK (2007) Mutational reconstructed ferric chelate reductase confers enhanced tolerance in rice to iron. Proc Natl Acad Sci USA 104:7373–7278
Ishimaru Y, Masuda H, Bashir K, Inoue H, Tsukamoto T, Takahashi M, Nakanishi H, Aoki N, Hirose T, Ohsugi R, Nishizawa NK (2010) Rice metal-nicotianamine transporter, OsYSL2, is required for the long-distance transport of iron and manganese. Plant J 62:379–390
Izawa H, Yoshida N, Shiragai N, Aoyagi Y (2008) Nicotianamine content in various beans and its inhibition activity of angiotensun-I converting enzyme. Nippon Shokuhin Kagaku Kogaku Kaishi 55:253–357
Kakei Y, Yamaguchi I, Kobayashi T, Takahashi M, Nakanishi H, Yamakawa T, Nishizawa NK (2009) A highly sensitive, quick and simple quantification method for nicotianamine and 2'-deoxymugineic acid from minimum samples using LC/ESI-TOF-MS achieves functional analysis of these components in plants. Plant Cell Physiol 50:1988–1993
Kawai S, Kamei S, Matsuda Y, Ando R, Kondo S, Ishizawa A, Alam S (2001) Concentrations of iron and phytosiderophores in xylem sap of iron-deficient barley plants. Soil Sci Plant Nutr 47:265–272
Kim S, Takahashi M, Higuchi K, Tsunoda K, Nakanishi H, Yoshimura E, Mori S, Nishizawa NK (2005) Increased nicotianamine biosynthesis confers enhanced tolerance of high levels of metals, in particular nickel, to plants. Plant Cell Physiol 46:1809–1818
Li S-Z, Chan-Halbrendt C (2009) Ethanol production in (the) People’s republic of China: potential and technologies. Applied Energy Journal 86:S162e9
Marschner H, Römheld V, Kissel M (1986) Different strategies in higher plants in mobilization and uptake of iron. J Plant Nutr 9:695–713
Masuda H, Suzuki M, Morikawa K, Kobayashi T, Nakanishi H, Takahashi M, Saigusa M, Mori S, Nishizawa NK (2008) Increase in iron and zinc concentrations in rice grains via the introduction of barley genes involved in phytosiderophore synthesis. Rice 1:100–108
Masuda H, Usuda K, Kobayashi T, Ishimaru Y, Kakei T, Takahashi M, Higuchi K, Nakanishi H, Mori S, Nishizawa NK (2009) Overexpression of the barley nicotianamine synthase gene HvNAS1 increases iron and zinc concentration in rice grains. Rice 2:155–166
Mizuno D, Higuchi K, Sakamoto T, Nakanishi H, Mori S, Nishizawa NK (2003) Three nicotianamine synthase genes isolated from maize are differentially regulated by iron nutritional status. Plant Physiol 132:1989–1997
Mori S, Nishizawa NK, Hayashi H, Chino M, Yoshimura E, Ishihara J (1991) Why are young rice plants highly susceptible to Fe-deficiency? Plant Soil 130:143–156
Mori S, Nishizawa N (1987) Methionine as a dominant precursor of phytosiderophores in graminaceae plants. Plant and Cell Physiol 28:1081–1092
Murakami T, Ise K, Hayakawa K, Kamei S, Takagi S (1989) Stabilities of metal complexes of mugineic acids and their specific affinities for iron(III). Chem Lett 18:2137–2140
Nakanishi H, Yamaguchi H, Sasakuma T, Nishizawa NK, Mori S (2000) Two dioxygenase genes, Ids3 and Ids2, from Hordeum vulgare are involved in the biosynthesis of mugineic acid family phytosiderophores. Plant Mol Biol 44:199–207
Nishiyama R, Kato M, Nagata S, Yanagisawa S, Yoneyama T (2012) Identification of Zn-nicotianamine and Fe-2'-deoxymugineic acid in the phloem sap from rice plants. Plant Cell Physiol 53:381–390
Noma M, Noguchi M, Tamaki E (1971) A new amino acid, nicotianamine, from tobacco leaves. Tetrahedron Lett 22:2017–2020
Nozoye T, Kim S, Kakei Y, Takahashi M, Nakanishi H, Nishizawa NK (2014a) Enhanced levels of nicotianamine promote iron accumulation and tolerance to calcareous soil in soybean. Biosci Biotech Bioch 78:1677–1684
Nozoye T, Nagasaka S, Bashir K, Takahashi M, Kobayashi T, Nakanishi H, Nishizawa NK (2014b) Nicotianamine synthase 2 localizes to the vesicles of iron-deficient rice roots, and its mutation in the YXXφ or LL motif causes the disruption of vesicle formation or movement in rice. Plant J 77:246–260
Nozoye T, Tsunoda K, Nagasaka S, Bashir K, Takahashi M, Kobayashi T, Nakanishi H, Nishizawa NK (2014c) Rice nicotianamine synthase localizes to particular vesicles for proper function. Plant Signal Behav 9:e28660
Oki H, Yamaguchi H, Nakanishi H, Mori S (1999) Introduction of the reconstructed yeast ferric reductase gene, refre1, into tobacco. Plant Soil 215:211–220
Oki H, Kim S, Nakanishi H, Takahashi M, Yamaguchi H, Mori S, Nishizawa NK (2004) Directed evolution of yeast ferric reductase to produce plants with tolerance to iron deficiency in alkaline soils. Soil Sci Plant Nutr 50:1159–1165
Otani M, Shimada T, Kimura T, Saito A (1998) Transgenic plant production from embryogenic callus of sweet potato (Ipomoea batatas (L.) lam.) using agrobacterium tumefaciens. Plant Biotechnol 15:11–16
Robinson NJ, Procter CM, Connolly EL, Guerinot ML (1999) A ferric-chelate reductase for iron uptake from soils. Nature 397:694–697
Rockstrom J, Steffen W, Noone K, Persson A, Chapin FS 3rd, Lambin EF, Lenton TM, Scheffer M, Folke C, Schellnhuber HJ et al (2009) A safe operating space for humanity. Nature 461:472–475
Schroeder JI, Delhaize E, Frommer WB, Guerinot ML, Harrison MJ, Herrera-Estrella L, Horie T, Kochian LV, Munns R, Nishizawa NK et al (2013) Using membrane transporters to improve crops for sustainable food production. Nature 497:60–66
Schuler M, Rellán-Álvarez R, Fink-Straube C, Abadía J, Bauer P (2012) Nicotianamine functions in the phloem-based transport of iron to sink organs, in pollen development and pollen tube growth in Arabidopsis. Plant Cell 24:2380–2400
Schwartz L (2008) China fuels ethanol industry with yams, sweet potatoes and cassava. Available from: http://www.renewableenergyworldcom/rea/news/story?id.52450. Accessed 28 Aug 2008
Shojima S, Nishizawa NK, Mori S (1989) Establishment of a cell-free system for the biosynthesis of nicotianamine. Plant Cell Physiol 30:673–677
Shojima S, Nishizawa NK, Fushiya S, Nozoe S, Irifune T, Mori S (1990) Biosynthesis of phytosiderophores. In vitro biosynthesis of 2′-deoxymugineic acid from L-methionine and nicotianamine. Plant Physiol 93:1497–1503
Suzuki K, Higuchi K, Nakanishi H, Nishizawa NK, Mori S (1999) Cloning of nicotianamine synthase genes from Arabidopsis thaliana. Soil Sci Plant Nutr 45:993–1002
Suzuki M, Takahashi M, Tsukamoto T, Watanabe S, Matsuhashi S, Yazaki J, Kishimoto N, Kikuchi S, Nakanishi H, Mori S et al (2006) Biosynthesis and secretion of mugineic acid family phytosiderophores in zinc-deficient barley. Plant J 48:85–97
Suzuki M, Morikawa KC, Nakanishi H, Takahashi M, Saigusa M, Mori S, Nishizawa NK (2008) Transgenic rice lines that include barley genes have increased tolerance to low iron availability in a calcareous paddy soil. Soil Sci Plant Nutr 54:77–85
Takagi S (1976) Naturally occurring iron-chelating compounds in oat-and rice-root washings: I. Activity measurement and preliminary characterization. Soil Sci Plant Nutr 22:423–433
Takahashi M, Terada Y, Nakai I, Nakanishi H, Yoshimura E, Mori S, Nishizawa NK (2003) Role of nicotianamine in the intracellular delivery of metals and plant reproductive development. Plant Cell 15:1263–1280
United Nations Framework Conventionon Climate Change (1992) http://unfccc.int/resource/docs/convkp/conveng.pdf
Usuda K, Wada Y, Ishimaru Y, Kobayashi T, Takahashi M, Nakanishi H, Nagato Y, Mori S, Nishizawa NK (2009) Genetically engineered rice containing larger amounts of nicotianamine to enhance the antihypertensive effect. Plant Biotechnol J 7:87–95
Vasconcelos M, Eckert H, Arahana V, Graef G, Grusak M-A, Clemente T (2006) Molecular and phenotypic characterization of transgenic soybean expressing the Arabidopsis ferric chelate reductase gene, FRO2. Planta 224:1116–1128
Vert G, Grotz N, Dédaldéchamp F, Gaymard F, Guerinot ML, Briat JF, Curie C (2002) IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 14:1223–1233
von Wiren N, Klair S, Bansal S, Briat JF, Khodr H, Shioiri T, Leigh RA, Hider RC (1999) Nicotianamine chelates both FeIII and FeII. Implications for metal transport in plants. Plant Physiol 119:1107–1114
Wallace A, Lunt O-R (1960) Iron chlorosis in horticultural plants, a review. J Am Soc Hortic Sci 75:819–840
Welch RM, Graham RD (2004) Breeding for micronutrients in staple food crops from a human nutrition perspective. J Exp Bot 55:353–364
Woolfe JA (1992) Sweet potato: an untapped food resource. Cambridge University Press, Cambridge, p. 643
Ziska LH, Runion GB, Tomecek M, Prior SA, Torbet HA, Sicher R (2009) An evaluation of cassava, sweet potato and field corn as potential carbohydrate sources for bioethanol production in Alabama and Maryland. Biomass & Bioenergy 33:1503e8
Acknowledgements
This work was supported by an advanced low carbon technology research and development program (ALCA) from the Japan Science and Technology Agency (to N. K. N.), by a grant-in-aid for Young Scientists (B) (grant number 15 K18658) from JSPS KAKENHI (to T.N.) and by a grant from Uragami-zaidan (to T.N.). We are grateful to Dr. Khurram Bashir (RIKEN CSRS) for reading the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Eric Paterson.
The amino acid sequences used in this paper are available in the RAP-DB/DDBJ/GenBank/TAIR/SoyKb nucleotide sequence databases under the accession numbers OsNAS1 (Os03g0307300), OsNAS2 (Os03g0307200), OsNAS3 (Os07g0689600), ZmNAS1 (BAB87846.1), ZmNAS2 (BAB87847.2), ZmNAS3 (BAB91326.1), HvNAS1 (BAB17827.1), HvNAS2 (BAA74582.1), HvNAS3 (BAA74581.1), HvNAS4 (BAA74583.1), HvNAS5 (Q9ZQV5), HvNAS6 (BAA74586.1), HvNAS7 (BAA74587.1), NASHOR1 (Q9XFB6.1), NASHOR2 (Q9XFB7.1), AtNAS1 (AT5G04950), AtNAS2 (AT5G56080), AtNAS3 (AT1G09240), AtNAS4 (AT1G56430), TcNAS1 (AJ300446.1), SlNAS (NP_001296307.1), TaNAS1 (CAC82913.1),Glyma19g41630.1, Glyma03g39050.1, Glyma09g04100.1, Glyma15g40180.1 and Glyma08g18710.1.
Rights and permissions
About this article
Cite this article
Nozoye, T., Otani, M., Senoura, T. et al. Overexpression of barley nicotianamine synthase 1 confers tolerance in the sweet potato to iron deficiency in calcareous soil. Plant Soil 418, 75–88 (2017). https://doi.org/10.1007/s11104-016-3134-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-3134-4