Abstract
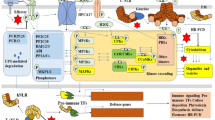
Plants live in constantly changing and often unfavorable or stressful environments. Environmental changes induce biotic and abiotic stress, which, in turn, may cause genomic DNA damage. Hence, plants simultaneously suffer abiotic/biotic stress and DNA damage. However, little information is available on the signaling crosstalk that occurs between DNA damage and abiotic/biotic stresses. Arabidopsis thaliana SUPPRESSOR OF GAMMA RESPONSE1 (SOG1) is a pivotal transcription factor that regulates thousands of genes in response to DNA double-strand break (DSB), and we recently reported that SOG1 has a role in immune responses. In the present study, the effects of SOG1 overexpression on the DNA damage and immune responses were examined. Results found that SOG1 overexpression enhances the regulation of numerous downstream genes. Relative to the wild type plants, then, DNA damage responses were observed to be strongly induced. SOG1 overexpression also upregulates chitin (a major components of fungal cell walls) responsive genes in the presence of DSBs, implying that pathogen defense response is activated by DNA damage via SOG1. Further, SOG1 overexpression enhances fungal resistance. These results suggest that SOG1 regulates crosstalk between DNA damage response and the immune response and that plants have evolved a sophisticated defense network to contend with environmental stress.
Key message
Overexpression of SOG1 enhances various DNA damage responses and fungal resistance, suggesting that SOG1 regulates the crosstalk between the DNA damage response and immune responses.











Similar content being viewed by others
References
Adachi S, Minamisawa K, Okushima Y, Inagaki S, Yoshiyama K, Kondou Y, Kaminuma E, Kawashima M, Toyoda T, Matsui M, Kurihara D, Matsunaga S, Umeda M (2011) Programmed induction of endoreduplication by DNA double-strand breaks in Arabidopsis. Proc Natl Acad Sci USA 108:10004–10009
Aklilu BB, Culligan KM (2016) Molecular evolution and functional diversification of replication protein A1 in plants. Front Plant Sci 7:33
Atkinson NJ, Lilley CJ, Urwin PE (2013) Identification of genes involved in the response of Arabidopsis to simultaneous biotic and abiotic stresses. Plant Physiol 162:2028–2041
Bourbousse C, Vegesna N, Law JA (2018) SOG1 activator and MYB3R repressors regulate a complex DNA damage network in Arabidopsis. Proc Natl Acad Sci USA 115:E12453–E12462
Brogue K, Chet I, Holliday M, Cressman R, Biddle P, Knowlton S, Mauvais CJ, Broglie R (1991) Transgenic plants with enhanced resistance to the fungal pathogen Rhizoctonia solani. Science 254:1194–1197
Chatterjee N, Walker GC (2017) Mechanisms of DNA damage, repair, and mutagenesis. Environ Mol Mutagen 58:235–263
Chen YL, Lee CY, Cheng KT, Chang WH, Huang RN, Nam HG, Chen YR (2014) Quantitative peptidomics study reveals that a wound-induced peptide from PR-1 regulates immune signaling in tomato. Plant Cell 26:4135–4148
Chen P, Takatsuka H, Takahashi N, Kurata R, Fukao Y, Kobayashi K, Ito M, Umeda M (2017) Arabidopsis R1R2R3-Myb proteins are essential for inhibiting cell division in response to DNA damage. Nat Commun 8:635
Crooks GE, Hon G, Chandonia JM, Brenner SE (2004) WebLogo: a sequence logo generator. Genome Res 14:1188–1190
Culligan KM, Robertson CE, Foreman J, Doerner P, Britt AB (2006) ATR and ATM play both distinct and additive roles in response to ionizing radiation. Plant J 48:947–961
Durrant WE, Wang S, Dong X (2007) Arabidopsis SNI1 and RAD51D regulate both gene transcription and DNA recombination during the defense response. Proc Natl Acad Sci USA 104:4223–4227
Fulcher N, Sablowski R (2009) Hypersensitivity to DNA damage in plant stem cell niches. Proc Natl Acad Sci USA 106:20984–20988
Furukawa T, Curtis MJ, Tominey CM, Duong YH, Wilcox BW, Aggoune D, Hays JB, Britt AB (2010) A shared DNA-damage-response pathway for induction of stem-cell death by UVB and by gamma irradiation. DNA Repair (Amst) 9:940–948
Gamir J, Darwiche R, Van't Hof P, Choudhary V, Stumpe M, Schneiter R, Mauch F (2017) The sterol-binding activity of PATHOGENESIS-RELATED PROTEIN 1 reveals the mode of action of an antimicrobial protein. Plant J 89:502–509
Gupta A, Dixit SK, Senthil-Kumar M (2016) Drought stress predominantly endures Arabidopsis thaliana to pseudomonas syringae infection. Front Plant Sci 7:808
Hain R, Reif HJ, Krause E, Langebartels R, Kindl H, Vornam B, Wiese W, Schmelzer E, Schreier PH, Stocker RH et al (1993) Disease resistance results from foreign phytoalexin expression in a novel plant. Nature 361:153–156
Hong JH, Savina M, Du J, Devendran A, Kannivadi Ramakanth K, Tian X, Sim WS, Mironova VV, Xu J (2017) A Sacrifice-for-Survival Mechanism Protects Root Stem Cell Niche from Chilling Stress. Cell 170:102–113e14
Huang CH, Mirabelli CK, Jan Y, Crooke ST (1981) Single-strand and double-strand deoxyribonucleic acid breaks produced by several bleomycin analogues. Biochemistry 20:233–238
Jirage D, Tootle TL, Reuber TL, Frost LN, Feys BJ, Parker JE, Ausubel FM, Glazebrook J (1999) Arabidopsis thaliana PAD4 encodes a lipase-like gene that is important for salicylic acid signaling. Proc Natl Acad Sci USA 96:13583–13588
Kaku H, Nishizawa Y, Ishii-Minami N, Akimoto-Tomiyama C, Dohmae N, Takio K, Minami E, Shibuya N (2006) Plant cells recognize chitin fragments for defense signaling through a plasma membrane receptor. Proc Natl Acad Sci USA 103:11086–11091
Kiba A, Nishihara M, Nakatsuka T, Yamamura S (2007) Pathogenesis-related protein 1 homologue is an antifungal protein in Wasabia japonica leaves and confers resistance to Botrytis cinerea in transgenic tobacco. Plant Biotechnol 24:247–253
Kohonen T (1984) Self-organized formation of topologically correct feature maps. Biol Cybern 43:59–69
Koroleva OA, Tomlinson M, Parinyapong P, Sakvarelidze L, Leader D, Shaw P, Doonan JH (2004) CycD1, a putative G1 cyclin from Antirrhinum majus, accelerates the cell cycle in cultured tobacco BY-2 cells by enhancing both G1/S entry and progression through S and G2 phases. Plant Cell 16:2364–2379
Kovalchuk I, Kovalchuk O, Kalck V, Boyko V, Filkowski J, Heinlein M, Hohn B (2003) Pathogen-induced systemic plant signal triggers DNA rearrangements. Nature 423:760–762
Lageix S, Catrice O, Deragon JM, Gronenborn B, Pelissier T, Ramirez BC (2007) The nanovirus-encoded Clink protein affects plant cell cycle regulation through interaction with the retinoblastoma-related protein. J Virol 81:4177–4185
Libault M, Wan J, Czechowski T, Udvardi M, Stacey G (2007) Identification of 118 Arabidopsis transcription factor and 30 ubiquitin-ligase genes responding to chitin, a plant-defense elicitor. Mol Plant Microbe Interact 20:900–911
Liu D, Raghothama KG, Hasegawa PM, Bressan RA (1994) Osmotin overexpression in potato delays development of disease symptoms. Proc Natl Acad Sci USA 91:1888–1892
Ma B, Pan Y, Zheng J, Levine AJ, Nussinov R (2007) Sequence analysis of p53 response-elements suggests multiple binding modes of the p53 tetramer to DNA targets. Nucleic Acids Res 35:2986–3001
Maere S, Heymans K, Kuiper M (2005) BiNGO: a Cytoscape plugin to assess overrepresentation of gene ontology categories in biological networks. Bioinformatics 21:3448–3449
Mittler R (2006) Abiotic stress, the field environment and stress combination. Trends Plant Sci 11:15–19
Ogita N, Okushima Y, Tokizawa M, Yamamoto YY, Tanaka M, Seki M, Makita Y, Matsui M, Okamoto-Yoshiyama K, Sakamoto T, Kurata T, Hiruma K, Saijo Y, Takahashi N, Umeda M (2018) Identifying the target genes of SUPPRESSOR OF GAMMA RESPONSE 1, a master transcription factor controlling DNA damage response in Arabidopsis. Plant J 94:439–453
Prasch CM, Sonnewald U (2013) Simultaneous application of heat, drought, and virus to Arabidopsis plants reveals significant shifts in signaling networks. Plant Physiol 162:1849–1866
Reczek EE, Flores ER, Tsay AS, Attardi LD, Jacks T (2003) Multiple response elements and differential p53 binding control Perp expression during apoptosis. Mol Cancer Res 1:1048–1057
Ricaud L, Proux C, Renou JP, Pichon O, Fochesato S, Ortet P, Montane MH (2007) ATM-mediated transcriptional and developmental responses to gamma-rays in Arabidopsis. PLoS ONE 2:e430
Riet KB, Ndlovu N, Piater LA, Dubery IA (2016) Simultaneous analysis of defense-related phytohormones in Arabidopsis thaliana responding to fungal infection. Appl Plant Sci 4:1600013
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140
Sarowar S, Kim YJ, Kim EN, Kim KD, Hwang BK, Islam R, Shin JS (2005) Overexpression of a pepper basic pathogenesis-related protein 1 gene in tobacco plants enhances resistance to heavy metal and pathogen stresses. Plant Cell Rep 24:216–224
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Shin SH, Pak JH, Kim MJ, Kim HJ, Oh JS, Choi HK, Jung HW, Chung YS (2014) An acidic PATHOGENESIS-RELATED1 gene of Oryza grandiglumis is involved in disease resistance response against bacterial infection. Plant Pathol J 30:208–214
Sjogren CA, Bolaris SC, Larsen PB (2015) Aluminum-dependent terminal differentiation of the Arabidopsis root tip is mediated through an ATR-, ALT2-, and SOG1-regulated transcriptional response. Plant Cell 27:2501–2515
Song J, Bent AF (2014) Microbial pathogens trigger host DNA double-strand breaks whose abundance is reduced by plant defense responses. PLoS Pathog 10:e1004030
Suzuki N, Rivero RM, Shulaev V, Blumwald E, Mittler R (2014) Abiotic and biotic stress combinations. New Phytol 203:32–43
Takahashi I, Kojima S, Sakaguchi N, Umeda-Hara C, Umeda M (2010) Two Arabidopsis cyclin A3s possess G1 cyclin-like features. Plant Cell Rep 29:307–315
Uknes S, Mauch-Mani B, Moyer M, Potter S, Williams S, Dincher S, Chandler D, Slusarenko A, Ward E, Ryals J (1992) Acquired resistance in Arabidopsis. Plant Cell 4:645–656
van Helden J, Rios AF, Collado-Vides J (2000) Discovering regulatory elements in non-coding sequences by analysis of spaced dyads. Nucleic Acids Res 28:1808–1818
Wehrens R, Buydens LM (2007) Self- and super-organizing maps in R: the kohonen package. J Stat Softw 21:1–19
Xing DH, Lai ZB, Zheng ZY, Vinod KM, Fan BF, Chen ZX (2008) Stress- and pathogen-induced Arabidopsis WRKY48 is a transcriptional activator that represses plant basal defense. Mol Plant 1:459–470
Yalpani N, Silverman P, Wilson TM, Kleier DA, Raskin I (1991) Salicylic acid is a systemic signal and an inducer of pathogenesis-related proteins in virus-infected tobacco. Plant Cell 3:809–818
Yan S, Wang W, Marques J, Mohan R, Saleh A, Durrant WE, Song J, Dong X (2013) Salicylic acid activates DNA damage responses to potentiate plant immunity. Mol Cell 52:602–610
Yoshiyama K, Conklin PA, Huefner ND, Britt AB (2009) Suppressor of gamma response 1 (SOG1) encodes a putative transcription factor governing multiple responses to DNA damage. Proc Natl Acad Sci USA 106:12843–12848
Yoshiyama KO, Kobayashi J, Ogita N, Ueda M, Kimura S, Maki H, Umeda M (2013) ATM-mediated phosphorylation of SOG1 is essential for the DNA damage response in Arabidopsis. EMBO Rep 14:817–822
Yoshiyama KO, Kimura S, Maki H, Britt AB, Umeda M (2014) The role of SOG1, a plant-specific transcriptional regulator, in the DNA damage response. Plant Signal Behav 9:e28889
Yoshiyama KO, Kaminoyama K, Sakamoto T, Kimura S (2017) Increased phosphorylation of ser-gln sites on SUPPRESSOR OF GAMMA RESPONSE1 strengthens the DNA damage response in Arabidopsis thaliana. Plant Cell 29:3255–3268
Acknowledgements
The authors thank Drs. Atsushi Higashitani and Shusei Sato for their support in the present study. We also thank Drs. Mika Teranishi and Yasukazu Daigaku for their helpful discussion throughout the study. This work was supported by JSPS KAKENHI (Grant No. 17K07455 to K.O.Y.; Grant No. 19K06708 to N. T.; Grant No. 18H04822 to K. H.; Grant No. 16KT0031 and 18H02467 to Y. S.; Grant No. 15H05945 and 17H01872 to J. H.; Grant No. 17H06470, 17H06477 and 17H03965 to M. U.; Grant No. 16H01472, 16K07408, 18H04787, and 18H04844 to S. K.), by JST (Grant No. JPMJPR16Q7 to K. H.), by the MEXT Supported Program for the Strategic Research Foundation at Private Universities from the Ministry of Education, Culture, Sports, Science & Technology of Japan, (Grant No. S1511023 to S.K.), and by the Sumitomo foundation (Grant No. 181147 to K. O. Y).
Author information
Authors and Affiliations
Contributions
KOY planned the experiments and performed most of the experiments. TS performed analysis of SOG1-binding sequence. SK performed data RNA-seq data analysis. NA, NT, KH, YS, and MU, performed pathogen inoculating assay. KOY, JH, and SK conceived the project and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoshiyama, K.O., Aoshima, N., Takahashi, N. et al. SUPPRESSOR OF GAMMA RESPONSE 1 acts as a regulator coordinating crosstalk between DNA damage response and immune response in Arabidopsis thaliana. Plant Mol Biol 103, 321–340 (2020). https://doi.org/10.1007/s11103-020-00994-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-00994-0