Abstract
Purpose
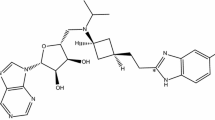
Exploration of the chemical, analytical and pharmacokinetic properties of the API, RO7304898, an allosteric EGFR inhibitor, intended to be developed as a mixture of two rapidly interconverting diastereoisomers with composition ratio of approximately 1:1.
Methods
Assessment of diastereoisomer stereochemistry, interconversion rates, binding to EGFR protein, metabolic stability and in vivo PK in Wistar-Han rats was conducted.
Results
The two diastereoisomers of the API undergo fast interconversion at physiologically relevant pH and direct EGFR binding studies revealed diastereoisomer B to be the active moiety. Pharmacokinetic studies in rat revealed a low-moderate total plasma clearance of the API along with similar plasma concentration-time profiles for diastereoisomers A and B, and the diastereoisomeric ratio reached stable equilibrium favoring formation of the potent diastereoisomer B. In in vitro incubations, the API was metabolically stable in plasma and hepatocyte suspension incubations in all species tested except that of rat hepatocytes. Additionally, only small species differences in the A:B composition were observed in vitro with the potent diastereoisomer B being the predominant form.
Conclusions
We demonstrated that the API, a mixture of two diastereoisomers; A (impotent) and B (potent), undergoes rapid interconversion which is faster than the apparent distribution and elimination rates of the individual diastereoisomers in vivo in rat, serving to diminish concerns that separate diastereoisomer effects may occur in subsequent pharmacologic and pivotal toxicological studies. Whilst vigilant monitoring of the diastereoisomeric ratio will need to be continued, this data adds confidence on the development pathway for this API to the clinic.









Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this published article.
Abbreviations
- A:B:
-
Ratio of diastereoisomer A to B
- API:
-
Active pharmaceutical agent
- CLint:
-
Intrinsic clearance
- D20:
-
Deuterated water
- D6-DMSO:
-
Deuterated dimethyl sulfoxide
- DMSO:
-
Dimethyl sulfoxide
- d.r.:
-
Diastereoisomeric ratio
- EGFR:
-
Epidermal growth factor receptor
- EMA:
-
European Medicines Agency
- FDA:
-
Food and Drug Agency
- H/D:
-
Hydrogen/Deuterium
- HPLC-MS:
-
High performance liquid chromatography coupled mass spectrometer
- HPLC-UV:
-
High-performance liquid chromatography coupled ultra-violet (UV) spectroscopy
- IS:
-
Analytical internal standard
- i.v.:
-
Intravenous
- K3-EDTA:
-
Tripotassium ethylenediaminetetraacetic acid
- KD:
-
Equilibrium dissociation constant
- LC-MS/MS:
-
Liquid chromatography coupled tandem mass spectrometer
- LLE:
-
Liquid liquid extraction
- LLOQ:
-
Lower limit of quantitation
- min:
-
Minutes
- MRM:
-
Multiple reaction monitoring
- NMR:
-
Nuclear magnetic resonance
- NOE:
-
Nuclear Overhauser Effect
- PK:
-
Pharmacokinetic
- p.o. :
-
Per os, oral
- PP:
-
Protein precipitation
- PPM:
-
Parts per million
- QC:
-
Quality control
- RT:
-
Room temperature
- s:
-
Second
- TKI:
-
Tyrosine kinase inhibitor
References
Yu HA, Pao W. Targeted therapies: Afatinib--new therapy option for EGFR-mutant lung cancer. Nat Rev Clin Oncol. 2013;10(10):551–2.
Thress KS, Paweletz CP, Felip E, Cho BC, Stetson D, Dougherty B, et al. Acquired EGFR C797S mutation mediates resistance to AZD9291 in non-small cell lung cancer harboring EGFR T790M. Nat Med. 2015;21(6):560–2.
US FDA. Development of New Stereoisomeric Drugs 1992 [Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/development-new-stereoisomeric-drugs.
EMA. Investigation of chiral active substances 1993 [Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/investigation-chiral-active-substances_en.pdf.
Moilanen A-M, Riikonen R, Oksala R, Ravanti L, Aho E, Wohlfahrt G, et al. Discovery of ODM-201, a new-generation androgen receptor inhibitor targeting resistance mechanisms to androgen signaling-directed prostate cancer therapies. Sci Rep. 2015;5(1):12007.
Taavitsainen P, Gieschen H, Korjamo T, Kähkönen M, Malmström C, Prien O, et al. Absorption, distribution, metabolism and excretion of darolutamide (a novel non-steroidal androgen receptor antagonist) in rats. Xenobiotica. 2020;50(8):967–79.
Nykänen P, Korjamo T, Gieschen H, Zurth C, Koskinen M. Pharmacokinetics of Darolutamide, its Diastereomers and Active Metabolite in the Mouse: Response to Saini NK et al. (2020). Drug Metab Lett. 2020.
Lon H-K, Mendonca N, Goss S, Othman AA, Locke C, Jin Z, et al. Pharmacokinetics, safety, tolerability, and pharmacodynamics of Alicapistat, a selective inhibitor of human Calpains 1 and 2 for the treatment of Alzheimer disease: an overview of phase 1 studies. Clin Pharmacol Drug Dev. 2019;8(3):290–303.
Kling A, Jantos K, Mack H, Hornberger W, Drescher K, Nimmrich V, et al. Discovery of novel and highly selective inhibitors of Calpain for the treatment of Alzheimer’s disease: 2-(3-phenyl-1H-pyrazol-1-yl)-nicotinamides. J Med Chem. 2017;60(16):7123–38.
Jantos K, Kling A, Mack H, Hornberger W, Moeller A, Nimmrich V, et al. Discovery of ABT-957: 1-Benzyl-5-oxopyrrolidine-2-carboxamides as selective calpain inhibitors with enhanced metabolic stability. Bioorg Med Chem Lett. 2019;29(15):1968–73.
Wang H, Edom RW, Kumar S, Vincent S, Shen Z. Separation and quantification of two diastereomers of a drug candidate in rat plasma by ultra-high pressure liquid chromatography/mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2007;854(1–2):26–34.
Du J, Ma Z, Zhang Y, Wang T, Chen X, Zhong D. Simultaneous determination of ornidazole and its main metabolites in human plasma by LC-MS/MS: application to a pharmacokinetic study. Bioanalysis. 2014;6(18):2343–56.
Bao D, Ross BS, Sofia MJ. A liquid chromatography-tandem mass spectrometry method for the quantitative determination of diastereomers of a phosphoramidate nucleotide prodrug (PSI-7851) in human plasma. Biomed Chromatogr. 2012;26(5):583–8.
Ledvina AR, Dayton B, Hoffmann M, Steege T, Cape S, Holmes V, et al. Development and validation of bioanalytical methods to support investigations of AZD9496 in the clinic. Bioanalysis. 2020;12(5):305–17.
Briscoe CJ, Hage DS. Factors affecting the stability of drugs and drug metabolites in biological matrices. Bioanalysis. 2009;1(1):205–20.
Li W, Zhang J, Tse FL. Strategies in quantitative LC-MS/MS analysis of unstable small molecules in biological matrices. Biomed Chromatogr. 2011;25(1–2):258–77.
Liu M, Ma JY, Zhang Y, Wang X, Zhao H, Du A, et al. An LC-MS/MS method for simultaneous determination of cefprozil diastereomers in human plasma and its application for the bioequivalence study of two cefprozil tablets in healthy Chinese volunteers. Biomed Chromatogr. 2016;30(3):288–93.
US FDA. Bioanalytical Method Validation Guidance for Industry 2018 [Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry.
EMA. Guideline on bioanalytical method development 2012 [Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-bioanalytical-method-validation_en.pdf.
Acknowledgements and Disclosures
Thomas Wirz, Monique Wittig-Kieffer, Fred Avenel and Pawel Dzygiel for bioanalytical method development and sample analysis. Annie Blondé, Claudia Senn and Veronique Dall’Asen for formulation and conduct of the rat in vivo single dose PK study and Laurent Gand for evaluation of the the rat pharmacokinetic parameters. Kenichi Umehara, Florian Klammers, Isabelle Walter and Vincent Monin for the conduct and evaluation of the in vitro plasma and hepatocyte studies. Dominique Burger expressed and purified the EGFR LR protein for the direct binding assay. Martin Binder for acquisition and interpretation of NMR data. Stefania Beato, Daniela Fraier, Luca Ferrari and Dietrich Türck for scientific discussions. The authors have no conflicts of interest to declare that are relevant to the content of this article.
Author information
Authors and Affiliations
Contributions
Conception and study design: Caroline Rynn, Katja Heinig, Filippo Sadojevich, Alfred Ross, Philipp Koldewey and Saša Miladinovic.
Acquisition and analysis of data: Caroline Rynn, Katja Heinig, Filippo Sadojevich, Alfred Ross, Philipp Koldewey and Saša Miladinovic.
Drafting and revision of the manuscript: Caroline Rynn, Katja Heinig, Filippo Sadojevich, Jeannine-Petrig Schaffland, Georg Jaeschke, Alfred Ross, Philipp Koldewey, Saša Miladinovic and Jin Wang.
Critical review of the manuscript: Daniela Fraier, Luca Ferrari and Stafania Beato.
Final review and approval: Caroline Rynn, Katja Heinig, Filippo Sadojevich, Jeannine Petrig Schaffland, Georg Jaeschke, Alfred Ross, Philipp Koldewey, Saša Miladinovic and Jin Wang.
Corresponding author
Ethics declarations
Research Involving Animals
The protocol was reviewed and approved by the Institutional Animal Care and Use Committee of the Cantonal Veterinary Office Basel, Switzerland. The study was performed in accordance with the ‘Guide For The Care and Use of Laboratory Animals’ as adopted and promulgated by the U.S. National Institutes of Health.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 493 kb)
Rights and permissions
About this article
Cite this article
Heinig, K., Sladojevich, F., Petrig Schaffland, J. et al. Chemical, Analytical and Pharmacokinetic Characterisation of RO7304898, an API Consisting of Two Rapidly Interconverting Diastereoisomers. Pharm Res 39, 653–667 (2022). https://doi.org/10.1007/s11095-022-03234-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-022-03234-w