Abstract
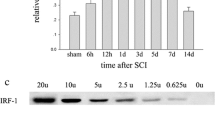
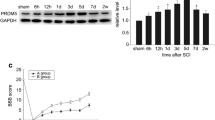
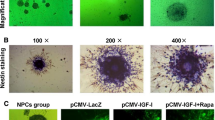
IGFBP6, a member of the insulin-like growth factor-binding proteins family that contains six high affinity IGFBPs, modulates insulin-like growth factor (IGF) activity and also showed an independent effect of IGF, such as growth inhibition and apoptosis. However, the role of IGFBP6 in spinal cord injury (SCI) remains largely elusive. In this study, we have performed an acute SCI model in adult rats and investigated the dynamic changes of IGFBP6 expression in the spinal cord. Our results showed that IGFBP6 was upregulated significantly after SCI, which was paralleled with the levels of apoptotic proteins p53 and active caspase-3. Immunofluorescent labeling showed that IGFBP6 was co-localizated with active caspase-3 and p53 in neurons. To further investigate the function of IGFBP6, an apoptosis model was established in primary neuronal cells. When IGFBP6 was knocked down by specific short interfering RNA (siRNA), the protein levels of active caspase-3 and Bax as well as the number of apoptotic primary neurons were significantly decreased in our study. Taken together, our findings suggest that the change of IGFBP6 protein expression plays a key role in neuronal apoptosis after SCI.







Similar content being viewed by others
References
Zhang J, Feng G, Bao G, Xu G, Sun Y, Li W, Wang L, Chen J, Jin H, Cui Z (2015) Nuclear translocation of PKM2 modulates astrocyte proliferation via p27 and -catenin pathway after spinal cord injury. Cell cycle 14(16):2609–2618. doi:10.1080/15384101.2015.1064203
Khalatbary AR, Zarrinjoei GR (2012) Anti-inflammatory effect of oleuropein in experimental rat spinal cord trauma. Iranian Red Crescent Med J 14 (4):229–234
Saadoun S, Bell BA, Verkman AS, Papadopoulos MC (2008) Greatly improved neurological outcome after spinal cord compression injury in AQP4-deficient mice. Brain 131 (Pt 4):1087–1098. doi:10.1093/brain/awn014
DePaul MA, Palmer M, Lang BT, Cutrone R, Tran AP, Madalena KM, Bogaerts A, Hamilton JA, Deans RJ, Mays RW, Busch SA, Silver J (2015) Intravenous multipotent adult progenitor cell treatment decreases inflammation leading to functional recovery following spinal cord injury. Scientific reports 5:16795. doi:10.1038/srep16795
Wells JE, Rice TK, Nuttall RK, Edwards DR, Zekki H, Rivest S, Yong VW (2003) An adverse role for matrix metalloproteinase 12 after spinal cord injury in mice. J Neurosci 23(31):10107–10115
Alkabie S, Boileau AJ (2016) The role of therapeutic hypothermia after traumatic spinal cord injury-a systematic review. World Neurosurg 86:432–449. doi:10.1016/j.wneu.2015.09.079
Bareiss SK, Dugan E, Brewer KL (2015) PI3K mediated activation of GSK-3beta reduces at-level primary afferent growth responses associated with excitotoxic spinal cord injury dysesthesias. Mol Pain 11:35. doi:10.1186/s12990-015-0041-2
Wu MF, Zhang SQ, Liu JB, Li Y, Zhu QS, Gu R (2015) Neuroprotective effects of electroacupuncture on early- and late-stage spinal cord injury. Neural Regen Res 10(10):1628–1634. doi:10.4103/1673-5374.167762
Casha S, Zygun D, McGowan MD, Bains I, Yong VW, Hurlbert RJ (2012) Results of a phase II placebo-controlled randomized trial of minocycline in acute spinal cord injury. Brain 135 (Pt 4):1224–1236. doi:10.1093/brain/aws072
Byrnes KR, Stoica BA, Fricke S, Di Giovanni S, Faden AI (2007) Cell cycle activation contributes to post-mitotic cell death and secondary damage after spinal cord injury. Brain 130 (Pt 11):2977–2992. doi:10.1093/brain/awm179
Niu C, Yip HK (2011) Neuroprotective signaling mechanisms of telomerase are regulated by brain-derived neurotrophic factor in rat spinal cord motor neurons. J Neuropathol Exp Neurol 70(7):634–652. doi:10.1097/NEN.0b013e318222b97b
Choo AM, Liu J, Dvorak M, Tetzlaff W, Oxland TR (2008) Secondary pathology following contusion, dislocation, and distraction spinal cord injuries. Exp Neurol 212(2):490–506. doi:10.1016/j.expneurol.2008.04.038
Xu Y, Zhang L, Sun SK, Zhang X (2014) CC chemokine ligand 18 and IGF-binding protein 6 as potential serum biomarkers for prostate cancer. Tohoku J Exp Med 233(1):25–31
Hwa V, Oh Y, Rosenfeld RG (1999) The insulin-like growth factor-binding protein (IGFBP) superfamily. Endocr Rev 20(6):761–787. doi:10.1210/edrv.20.6.0382
Cui J, Ma C, Qiu J, Ma X, Wang X, Chen H, Huang B (2011) A novel interaction between insulin-like growth factor binding protein-6 and the vitamin D receptor inhibits the role of vitamin D3 in osteoblast differentiation. Mol Cell Endocrinol 338(1–2):84–92. doi:10.1016/j.mce.2011.03.011
Sueoka N, Lee HY, Wiehle S, Cristiano RJ, Fang B, Ji L, Roth JA, Hong WK, Cohen P, Kurie JM (2000) Insulin-like growth factor binding protein-6 activates programmed cell death in non-small cell lung cancer cells. Oncogene 19(38):4432–4436. doi:10.1038/sj.onc.1203813
Grellier P, Berrebi D, Peuchmaur M, Babajko S (2002) The IGF system in neuroblastoma xenografts: focus on IGF-binding protein-6. J Endocrinol 172(3):467–476
Iosef C, Gkourasas T, Jia CY, Li SS, Han VK (2008) A functional nuclear localization signal in insulin-like growth factor binding protein-6 mediates its nuclear import. Endocrinology 149(3):1214–1226. doi:10.1210/en.2007-0959
Gruner JA (1992) A monitored contusion model of spinal cord injury in the rat. J Neurotrauma 9 (2):123–126; discussion 126–128. doi:10.1089/neu.1992.9.123
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12(1):1–21
Bethea JR, Dietrich WD (2002) Targeting the host inflammatory response in traumatic spinal cord injury. Curr Opin Neurol 15(3):355–360
Keane RW, Davis AR, Dietrich WD (2006) Inflammatory and apoptotic signaling after spinal cord injury. J Neurotrauma 23(3–4):335–344. doi:10.1089/neu.2006.23.335
Oyinbo CA (2011) Secondary injury mechanisms in traumatic spinal cord injury: a nugget of this multiply cascade. Acta Neurobiol Exp 71(2):281–299
Zhang J, Cui Z, Feng G, Bao G, Xu G, Sun Y, Wang L, Chen J, Jin H, Liu J, Yang L, Li W (2015) RBM5 and p53 expression after rat spinal cord injury: implications for neuronal apoptosis. Int J Biochem Cell Biol 60:43–52. doi:10.1016/j.biocel.2014.12.020
Morrison RS, Kinoshita Y, Johnson MD, Guo W, Garden GA (2003) p53-dependent cell death signaling in neurons. Neurochem Res 28(1):15–27
Miscusi M, Ebner F, Ceccariglia S, Menegazzi M, Mariotto S, Berra L, Del Fa A, Gangitano C, Lauretti L, Missori P, Delfini R, Suzuki H (2006) Early nuclear factor-kappaB activation and inducible nitric oxide synthase expression in injured spinal cord neurons correlating with a diffuse reduction of constitutive nitric oxide synthase activity. J Neurosurg Spine 4(6):485–493. doi:10.3171/spi.2006.4.6.485
Kotipatruni RR, Dasari VR, Veeravalli KK, Dinh DH, Fassett D, Rao JS (2011) p53- and Bax-mediated apoptosis in injured rat spinal cord. Neurochem Res 36(11):2063–2074. doi:10.1007/s11064-011-0530-2
Yuan B, Liu D, Liu X (2014) Spinal cord stimulation exerts analgesia effects in chronic constriction injury rats via suppression of the TLR4/NF-kappaB pathway. Neurosci Lett 581:63–68. doi:10.1016/j.neulet.2014.08.023
Miller FD, Pozniak CD, Walsh GS (2000) Neuronal life and death: an essential role for the p53 family. Cell Death Differ 7(10):880–888. doi:10.1038/sj.cdd.4400736
Alshatwi AA, Subash-Babu P, Antonisamy P (2016) Violacein induces apoptosis in human breast cancer cells through up regulation of BAX, p53 and down regulation of MDM2. Exp Toxicol Pathol 68(1):89–97. doi:10.1016/j.etp.2015.10.002
Follis AV, Llambi F, Merritt P, Chipuk JE, Green DR, Kriwacki RW (2015) Pin1-induced proline isomerization in cytosolic p53 mediates BAX activation and apoptosis. Mol Cell 59(4):677–684. doi:10.1016/j.molcel.2015.06.029
Martinou JC, Youle RJ (2011) Mitochondria in apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev Cell 21(1):92–101. doi:10.1016/j.devcel.2011.06.017
Bleicken S, Hofhaus G, Ugarte-Uribe B, Schroder R, Garcia-Saez AJ (2016) cBid, Bax and Bcl-xL exhibit opposite membrane remodeling activities. Cell Death Dis 7:e2121. doi:10.1038/cddis.2016.34
Zhang C, Lu L, Li Y, Wang X, Zhou J, Liu Y, Fu P, Gallicchio MA, Bach LA, Duan C (2012) IGF binding protein-6 expression in vascular endothelial cells is induced by hypoxia and plays a negative role in tumor angiogenesis. Int J Cancer 130(9):2003–2012. doi:10.1002/ijc.26201
Messmer-Blust A, An X, Li J (2009) Hypoxia-regulated angiogenic inhibitors. Trends Cardiovasc Med 19(8):252–256. doi:10.1016/j.tcm.2010.02.006
Koyama N, Zhang J, Huqun, Miyazawa H, Tanaka T, Su X, Hagiwara K (2008) Identification of IGFBP-6 as an effector of the tumor suppressor activity of SEMA3B. Oncogene 27(51):6581–6589
Kannan K, Amariglio N, Rechavi G, Jakob-Hirsch J, Kela I, Kaminski N, Getz G, Domany E, Givol D (2001) DNA microarrays identification of primary and secondary target genes regulated by p53. Oncogene 20(18):2225–2234. doi:10.1038/sj.onc.1204319
Han JJ, Huang BR, Wang X, Ma XL, Chen H (2009) Nuclear localization of insulin-like growth factor binding protein-6. Zhongguo yi xue ke xue yuan xue bao Acta Academiae Medicinae Sinicae 31 (6):735–739. doi:10.3881/j.issn.1000-503X.2009.06.017
Acknowledgements
This work was supported by the following funding, including the Health Research of the Jiangsu Province (Z201316); the Science and Technology of construction and people’s livelihood Foundation of Jiangsu Province (BL2014061); the ‘Top Six Types of Talents’ Financial Assistance of Jiangsu Province Grant (WSW-007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work, there is no professional or other personal interest of any nature or kind in any product, service and company that could be construed as influencing the position presented in, or the review of, the manuscript entitled.
Additional information
Song Wang and Zhiming Cui are contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, S., Liu, Y., Wu, C. et al. The Expression of IGFBP6 after Spinal Cord Injury: Implications for Neuronal Apoptosis. Neurochem Res 42, 455–467 (2017). https://doi.org/10.1007/s11064-016-2092-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-2092-9