Abstract
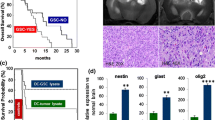
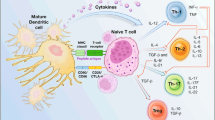
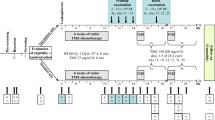
Establishment of a detection platform for glioblastoma-dendritic cell (DC) vaccine preparation and to determine the efficacy of the vaccine in a clinical trial. Autologous glioblastoma-DC vaccine was prepared from a glioblast specimen procured from surgical resection. The specimen was used to enrich the vaccine with peripherally blood-derived DCs after heat-shock induced, glioblastoma apoptosis. The control group received conventional treatment of surgery and radio-chemotherapy post-operation. The therapeutic group received a combination of glioblastoma-DC vaccine and conventional therapy. A comparison of the functional immune parameters, including tumor control, rate live time, Karnofsky scores, and complications occurring in each group were observed and recorded. The proportions of peripheral CD3+, CD3+CD4+, CD4+/CD8+, and NK cells were significantly higher after DC vaccination than the control group (P < 0.05). Serum levels of IL-2, IL-12, and IFN-γwere significantly higher after DC vaccination than in the control group (P < 0.05). Nine months after vaccination, tumor control rate is significantly improved in the DC group compared with the control group (P < 0.05); survival rate was significantly higher in DC group than in control group (P < 0.05) and the time to relapse was significantly longer in DC group than that in control group (P < 0.05). Karnofsky scores were better in DC vaccination group 6 and 9 months post-treatment compared with the control group (P < 0.05). The combination of glioma DC vaccine and radiotherapy/chemotherapy post-operatively enhances the immune function of patients, increases the tumor control rate, prolongs the survival time and relapse duration, improves the quality of life, and therefore provides a more effective intervention of treating glioblastoma.


Similar content being viewed by others
References
Winn R, Kliot M, and Brem H (2009) Youmans Neurological Surgery., Fifth ed., Philadelphia.
Preston-Martin S, Davis F, and McKean-Cowdin R. (2001) Epidemiology of primary brain tumors., In Brain tumor immunotherapy. (Liau, L. M., D.P., B., T.F., C., and D.D., B., Eds.), pp 47-72, Human Press.
Okada, H., Attanucci, J., Giezeman-Smits, K. M., Brissette-Storkus, C., Fellows, W. K., Gambotto, A., et al. (2001). Immunization with an antigen identified by cytokine tumor vaccine-assisted SEREX (CAS) suppressed growth of the rat 9L glioma in vivo. Cancer Research, 61, 2625–2631.
Schneider, T., Gerhards, R., Kirches, E., & Firsching, R. (2001). Preliminary results of active specific immunization with modified tumor cell vaccine in glioblastoma multiforme. Journal of Neuro-oncology, 53, 39–46.
Plautz, G. E., Miller, D. W., Barnett, G. H., Stevens, G. H., Maffett, S., Kim, J., et al. (2000). T cell adoptive immunotherapy of newly diagnosed gliomas. Clinical Cancer Research, 6, 2209–2218.
Plautz, G. E., Mukai, S., Cohen, P. A., & Shu, S. (2000). Cross-presentation of tumor antigens to effector T cells is sufficient to mediate effective immunotherapy of established intracranial tumors. Journal of Immunology, 165, 3656–3662.
Liau, L. M., Black, K. L., Prins, R. M., Sykes, S. N., DiPatre, P. L., Cloughesy, T. F., et al. (1999). Treatment of intracranial gliomas with bone marrow-derived dendritic cells pulsed with tumor antigens. Journal of Neurosurgery, 90, 1115–1124.
Caruso, D. A., Orme, L. M., Neale, A. M., Radcliff, F. J., Amor, G. M., Maixner, W., et al. (2004). Results of a phase 1 study utilizing monocyte-derived dendritic cells pulsed with tumor RNA in children and young adults with brain cancer. Neuro Oncol., 6, 236–246.
Wheeler, C. J. (2010). Dendritic cell vaccines to combat glioblastoma. Expert Rev Neurother., 10, 483–486.
Karnofsky DA, and Burchenal JH. (1949) The Clinical Evaluation of Chemotherapeutic Agents in Cancer., In Evaluation of Chemotherapeutic Agents (CM, M., Ed.), p 196, Columbia Univ Press.
Therasse, P., Arbuck, S. G., Eisenhauer, E. A., Wanders, J., Kaplan, R. S., Rubinstein, L., et al. (2000). New guidelines to evaluate the response to treatment in solid tumors European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. Journal of the National Cancer Institute, 92, 205–216.
Hildenbrand, B., Sauer, B., Kalis, O., Stoll, C., Freudenberg, M. A., Niedermann, G., et al. (2007). Immunotherapy of patients with hormone-refractory prostate carcinoma pre-treated with interferon-gamma and vaccinated with autologous PSA-peptide loaded dendritic cells–a pilot study. Prostate., 67, 500–508.
Liau, L. M., Prins, R. M., Kiertscher, S. M., Odesa, S. K., Kremen, T. J., Giovannone, A. J., et al. (2005). Dendritic cell vaccination in glioblastoma patients induces systemic and intracranial T-cell responses modulated by the local central nervous system tumor microenvironment. Clinical Cancer Research, 11, 5515–5525.
Yu, J. S., Liu, G., Ying, H., Yong, W. H., Black, K. L., & Wheeler, C. J. (2004). Vaccination with tumor lysate-pulsed dendritic cells elicits antigen-specific, cytotoxic T-cells in patients with malignant glioma. Cancer Research, 64, 4973–4979.
Yu, J. S., Wheeler, C. J., Zeltzer, P. M., Ying, H., Finger, D. N., Lee, P. K., et al. (2001). Vaccination of malignant glioma patients with peptide-pulsed dendritic cells elicits systemic cytotoxicity and intracranial T-cell infiltration. Cancer Research, 61, 842–847.
De Vleeschouwer, S., Fieuws, S., Rutkowski, S., Van Calenbergh, F., Van Loon, J., Goffin, J., et al. (2008). Postoperative adjuvant dendritic cell-based immunotherapy in patients with relapsed glioblastoma multiforme. Clinical Cancer Research, 14, 3098–3104.
Yamanaka, R., Homma, J., Yajima, N., Tsuchiya, N., Sano, M., Kobayashi, T., et al. (2005). Clinical evaluation of dendritic cell vaccination for patients with recurrent glioma: results of a clinical phase I/II trial. Clinical Cancer Research, 11, 4160–4167.
Sikorski, C. W., & Lesniak, M. S. (2005). Immunotherapy for malignant glioma: current approaches and future directions. Neurological Research, 27, 703–716.
Broder, H., Anderson, A., Kremen, T. J., Odesa, S. K., & Liau, L. M. (2003). MART-1 adenovirus-transduced dendritic cell immunization in a murine model of metastatic central nervous system tumor. Journal of Neuro-oncology, 64, 21–30.
Heimberger, A. B., Archer, G. E., Crotty, L. E., McLendon, R. E., Friedman, A. H., Friedman, H. S., et al. (2002). Dendritic cells pulsed with a tumor-specific peptide induce long-lasting immunity and are effective against murine intracerebral melanoma. Neurosurgery., 50, 158–164. discussion 164-156.
Jiang, N., Wang, G. S., Li, H., Zhang, J., Zhang, J. F., Yi, S. H., et al. (2009). Immunization with dendritic cells infected with mTERT adenovirus vector effectively elicits immunity against mouse H22 hepatoma in vivo. Zhonghua Zhong Liu Za Zhi., 31, 405–409.
Berntsen, A., Trepiakas, R., Wenandy, L., Geertsen, P. F., thor Straten, P., Andersen, M. H., et al. (2008). Therapeutic dendritic cell vaccination of patients with metastatic renal cell carcinoma: a clinical phase 1/2 trial. J Immunother., 31, 771–780.
Ribas, A., Comin-Anduix, B., Chmielowski, B., Jalil, J., de la Rocha, P., McCannel, T. A., et al. (2009). Dendritic cell vaccination combined with CTLA4 blockade in patients with metastatic melanoma. Clinical Cancer Research, 15, 6267–6276.
Soleimani, A., Berntsen, A., Svane, I. M., & Pedersen, A. E. (2009). Immune responses in patients with metastatic renal cell carcinoma treated with dendritic cells pulsed with tumor lysate. Scandinavian Journal of Immunology, 70, 481–489.
Niu, H. (2004). Experimental and clinical research of dendritic cell and syngeneic immunotherapy of brain glioma. The Chinese-German Journal of Clinical Oncology., 3, 147–150.
Wheeler, C. J., Black, K. L., Liu, G., Ying, H., Yu, J. S., Zhang, W., et al. (2003). Thymic CD8 + T cell production strongly influences tumor antigen recognition and age-dependent glioma mortality. Journal of Immunology, 171, 4927–4933.
Liu, G., Black, K. L., & Yu, J. S. (2006). Sensitization of malignant glioma to chemotherapy through dendritic cell vaccination. Expert Rev Vaccines., 5, 233–247.
Oken, M. M., Creech, R. H., Tormey, D. C., Horton, J., Davis, T. E., McFadden, E. T., et al. (1982). Toxicity and response criteria of the Eastern Cooperative Oncology Group. American Journal of Clinical Oncology, 5, 649–655.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jie, X., Hua, L., Jiang, W. et al. Clinical Application of a Dendritic Cell Vaccine Raised Against Heat-Shocked Glioblastoma. Cell Biochem Biophys 62, 91–99 (2012). https://doi.org/10.1007/s12013-011-9265-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-011-9265-6