Abstract
Background
Myricetin, a type of flavonol commonly found in fruits and herbs, has demonstrated anticancer properties by triggering the process of apoptosis or programmed cell death in tumor cells. Despite the absence of mitochondria and nuclei, erythrocytes can undergo programmed cell death, also known as eryptosis.This process is characterized by cell shrinkage, externalization of phosphatidylserine (PS) on the cell membrane, and the formation of membrane blebs. The signaling of eryptosis involves Ca2+ influx, the formation of reactive oxygen species (ROS), and the accumulation of cell surface ceramide. The present study explored the effects of myricetin on eryptosis.
Methods and results
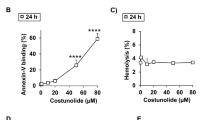
Human erythrocytes were exposed to various concentrations of myricetin (2–8 µM) for 24 h. Flow cytometry was used to assess the markers of eryptosis, including PS exposure, cellular volume, cytosolic Ca2+ concentration, and ceramide accumulation. In addition, the levels of intracellular ROS were measured using the 2’,7’-dichlorofluorescin diacetate (DCFDA) assay. The myricetin-treated (8 µM) erythrocytes significantly increased Annexin-positive cells, Fluo-3 fluorescence intensity, DCF fluorescence intensity, and the accumulation of ceramide. The impact of myricetin on the binding of annexin-V was significantly reduced, but not completely eliminated, by the nominal removal of extracellular Ca2+.
Conclusion
Myricetin triggers eryptosis, which is accompanied and, at least in part, caused by Ca2+ influx, oxidative stress and increase of ceramide abundance.





Similar content being viewed by others
Abbreviations
- PS:
-
Phosphatidylserine
- [Ca2+]i :
-
Cytosolic Ca2+ activity
- ROS:
-
Reactive oxygen species
- FSC:
-
Forward scatter
- DCFDA:
-
2′,7′-dichlorodihydrofluorescein diacetate
- DMSO:
-
Dimethyl sulfoxide
- FITC:
-
Fluorescein isothiocyanate
- EGTA:
-
Ethylenebis (oxyethylenenitrilo) tetraacetic acid
- BSA:
-
Bovine serum albumin
References
Jiang M, Zhu M, Wang L, Yu S (2019) Anti-tumor effects and associated molecular mechanisms of myricetin. Biomed Pharmacother 120:109506. https://doi.org/10.1016/j.biopha.2019.109506
Felice MR, Maugeri A, De Sarro G, Navarra M, Barreca D (2022) Molecular pathways involved in the anti-cancer activity of flavonols: a focus on myricetin and kaempferol. Int J Mol Sci 23(8):4411
Javed Z, Khan K, Herrera-Bravo J, Naeem S, Iqbal MJ, Raza Q, Sadia H, Raza S, Bhinder M, Calina D (2022) Myricetin: targeting signaling networks in cancer and its implication in chemotherapy. Cancer Cell Int 22(1):239
Chen Y-C, He X-L, Qi L, Shi W, Yuan L-W, Huang M-Y, Xu Y-L, Chen X, Gu L, Zhang L-L (2022) Myricetin inhibits interferon-γ-induced PD-L1 and IDO1 expression in lung cancer cells. Biochem Pharmacol 197:114940
Knickle A, Rasmussen A, Hoskin DW (2020) Myricetin induces apoptosis mediated by oxidative stress in 4T1 and E0771 mammary cancer cells. Mol Cell Toxicol 16:283–289
Ji A, Hu L, Ma D, Qiang G, Yan D, Zhang G, Jiang C (2022) Myricetin induces apoptosis and protective autophagy through endoplasmic reticulum stress in hepatocellular carcinoma. Evidence-Based Complementary and Alternative Medicine 2022
Zhou H, Xu L, Shi Y, Gu S, Wu N, Liu F, Huang Y, Qian Z, Xue W, Wang X (2023) A Novel Myricetin Derivative with Anti-cancer Properties induces cell cycle arrest and apoptosis in A549 cells. Biol Pharm Bull 46(1):42–51
Xie Y, Wang Y, Xiang W, Wang Q, Cao Y (2020) Molecular mechanisms of the action of myricetin in cancer. Mini Rev Med Chem 20(2):123–133
Yao A, Shen Y, Zhang Z, Zou Z, Wang A, Chen S, Zhang H, Chen F, Zhao J, Chen Z, Shan Y, Zhang X (2018) Sulforaphane and myricetin act synergistically to induce apoptosis in 3T3L1 adipocytes. Mol Med Rep 17(2):2945–2951. https://doi.org/10.3892/mmr.2017.8235
Kim W, Yang HJ, Youn H, Yun YJ, Seong KM, Youn B (2010) Myricetin inhibits akt survival signaling and induces bad-mediated apoptosis in a low dose ultraviolet (UV)-B-irradiated HaCaT human immortalized keratinocytes. J Radiat Res 51(3):285–296. https://doi.org/10.1269/jrr.09141
Kim GD (2017) Myricetin inhibits angiogenesis by inducing apoptosis and suppressing PI3K/Akt/mTOR signaling in endothelial cells. J cancer Prev 22(4):219
Yao X, Zhang J, Lu Y, Deng Y, Zhao R, Xiao S (2022) Myricetin restores Aβ-Induced mitochondrial impairments in N2a-SW cells. ACS Chem Neurosci 13(4):454–463
Sun J, Sun J, Zhou X (2019) Protective functions of myricetin in LPS-induced cardiomyocytes H9c2 cells injury by regulation of MALAT1. Eur J Med Res 24:1–9
Dubey R, Kulkarni SH, Dantu SC, Panigrahi R, Sardesai DM, Malik N, Acharya JD, Chugh J, Sharma S, Kumar A (2021) Myricetin protects pancreatic β-cells from human islet amyloid polypeptide (hIAPP) induced cytotoxicity and restores islet function. Biol Chem 402(2):179–194
Aminzadeh A, Bashiri H (2019) Myricetin ameliorates high glucose-induced endothelial dysfunction in human umbilical vein endothelial cells. Cell Biochem Funct. https://doi.org/10.1002/cbf.3442
Tan G, Uson-Lopez RA, Rahman MM, Hosokawa T, Saito T, Kurasaki M (2018) Myricetin enhances on apoptosis induced by serum deprivation in PC12 cells mediated by mitochondrial signaling pathway. Environ Toxicol Pharmacol 57:175–180. https://doi.org/10.1016/j.etap.2017.12.016
Rajendran P, Maheshwari U, Muthukrishnan A, Muthuswamy R, Anand K, Ravindran B, Dhanaraj P, Balamuralikrishnan B, Chang SW, Chung WJ (2021) Myricetin: versatile plant based flavonoid for cancer treatment by inducing cell cycle arrest and ROS–reliant mitochondria-facilitated apoptosis in A549 lung cancer cells and in silico prediction. Mol Cell Biochem 476:57–68
Kimura AM, Tsuji M, Yasumoto T, Mori Y, Oguchi T, Tsuji Y, Umino M, Umino A, Nishikawa T, Nakamura S (2021) Myricetin prevents high molecular weight Aβ1–42 oligomer-induced neurotoxicity through antioxidant effects in cell membranes and mitochondria. Free Radic Biol Med 171:232–244
Lang E, Lang F (2015) Mechanisms and pathophysiological significance of eryptosis, the suicidal erythrocyte death. Semin Cell Dev Biol 39:35–42. https://doi.org/10.1016/j.semcdb.2015.01.009
Repsold L, Joubert AM (2018) Eryptosis: An Erythrocyte’s Suicidal Type of Cell Death. Biomed Res Int 2018:9405617. https://doi.org/10.1155/2018/9405617
Föller M, Lang F (2020) Ion transport in eryptosis, the suicidal death of erythrocytes. Front Cell Dev Biology 8:597
Lang PA, Kaiser S, Myssina S, Wieder T, Lang F, Huber SM (2003) Role of Ca2+-activated K + channels in human erythrocyte apoptosis. Am J Physiol Cell Physiol 285(6):C1553–C1560
Lang E, Lang F (2015) Triggers, inhibitors, mechanisms, and significance of eryptosis: the suicidal erythrocyte death. Biomed Res Int 2015:513518. https://doi.org/10.1155/2015/513518
Lang F, Jilani K, Lang E (2015) Therapeutic potential of manipulating suicidal erythrocyte death. Expert Opin Ther Targets 19(9):1219–1227. https://doi.org/10.1517/14728222.2015.1051306
Dreischer P, Duszenko M, Stein J, Wieder T (2022) Eryptosis: programmed death of Nucleus-Free, Iron-Filled blood cells. Cells 11(3):503
McCaig WD, Hodges AL, Deragon MA, Haluska RJ Jr, Bandyopadhyay S, Ratner AJ, Spitalnik SL, Hod EA, LaRocca TJ (2019) Storage Primes Erythrocytes for Necroptosis and Clearance. Cell Physiol Biochem 53(3):496–507. https://doi.org/10.33594/000000153
Al Mamun Bhuyan A, Lang F (2018) Inhibition of Erythrocyte Cell membrane scrambling following Energy Depletion and hyperosmotic shock by Alectinib. Cell Physiol Biochem 51(5):1996–2009. https://doi.org/10.1159/000495777
Gajecki D, Gawryś J, Szahidewicz-Krupska E, Doroszko A (2022) Role of erythrocytes in nitric oxide metabolism and paracrine regulation of endothelial function. Antioxidants 11(5):943
Byrnes JR, Wolberg AS (2017) Red blood cells in thrombosis. Blood The Journal of the American Society of Hematology 130(16):1795–1799
Bissinger R, Bhuyan AAM, Qadri SM, Lang F (2019) Oxidative stress, eryptosis and anemia: a pivotal mechanistic nexus in systemic diseases. FEBS J 286(5):826–854
Abed M, Towhid ST, Mia S, Pakladok T, Alesutan I, Borst O, Gawaz M, Gulbins E, Lang F (2012) Sphingomyelinase-induced adhesion of eryptotic erythrocytes to endothelial cells. Am J Physiology-Cell Physiol 303(9):C991–C999
Alfhili MA, Basudan AM, Aljaser FS, Dera A, Alsughayyir J (2021) Bioymifi, a novel mimetic of TNF-related apoptosis-induced ligand (TRAIL), stimulates eryptosis. Med Oncol 38:1–10
Alamri HS, Alsughayyir J, Akiel M, Al-Sheikh YA, Basudan AM, Dera A, Barhoumi T, Basuwdan AM, Alfhili MA (2021) Stimulation of calcium influx and CK1α by NF‐κB antagonist [6]‐Gingerol reprograms red blood cell longevity. J Food Biochem 45(1):e13545
Fink M, Bhuyan AAM, Nürnberg B, Faggio C, Lang F (2019) Triggering of eryptosis, the suicidal erythrocyte death, by phenoxodiol. Naunyn Schmiedebergs Arch Pharmacol 392:1311–1318
Alfhili MA, Alsughayyir J, Basudan AB (2021) Epidemic dropsy toxin, sanguinarine chloride, stimulates sucrose-sensitive hemolysis and breakdown of membrane phospholipid asymmetry in human erythrocytes. Toxicon 199:41–48
Bissinger R, Bhuyan AAM, Qadri SM, Lang F (2019) Oxidative stress, eryptosis and anemia: a pivotal mechanistic nexus in systemic diseases. FEBS J 286(5):826–854. https://doi.org/10.1111/febs.14606
Lang F, Bissinger R, Abed M, Artunc F (2017) Eryptosis - the neglected cause of Anemia in End Stage Renal Disease. Kidney Blood Press Res 42(4):749–760. https://doi.org/10.1159/000484215
Abed M, Artunc F, Alzoubi K, Honisch S, Baumann D, Foller M, Lang F (2014) Suicidal erythrocyte death in end-stage renal disease. J Mol Med 92(8):871–879. https://doi.org/10.1007/s00109-014-1151-4
Bissinger R, Artunc F, Qadri SM, Lang F (2016) Reduced erythrocyte survival in uremic patients under hemodialysis or peritoneal Dialysis. Kidney Blood Press Res 41(6):966–977. https://doi.org/10.1159/000452600
Tozoni SS, Dias GF, Bohnen G, Grobe N, Pecoits-Filho R, Kotanko P, Moreno-Amaral AN (2019) Uremia and Hypoxia independently induce eryptosis and erythrocyte Redox Imbalance. Cell Physiol Biochem 53:794–804. https://doi.org/10.33594/000000173
Virzi GM, Milan Manani S, Clementi A, Castegnaro S, Brocca A, Riello C, de Cal M, Giuliani A, Battaglia GG, Crepaldi C, Ronco C (2019) Eryptosis is altered in peritoneal Dialysis patients. Blood Purif 1–7. https://doi.org/10.1159/000501541
Lang PA, Beringer O, Nicolay JP, Amon O, Kempe DS, Hermle T, Attanasio P, Akel A, Schafer R, Friedrich B, Risler T, Baur M, Olbricht CJ, Zimmerhackl LB, Zipfel PF, Wieder T, Lang F (2006) Suicidal death of erythrocytes in recurrent hemolytic uremic syndrome. J Mol Med 84(5):378–388. https://doi.org/10.1007/s00109-006-0058-0
Balola AHA, Mayer B, Bartolmas T, Salama A (2019) Sublytic Terminal Complement Components Induce Eryptosis in Autoimmune Haemolytic Anaemia related to IgM Autoantibodies. Cell Physiol Biochem 53(3):453–464. https://doi.org/10.33594/000000150
Voelkl J, Alzoubi K, Mamar AK, Ahmed MS, Abed M, Lang F (2013) Stimulation of suicidal erythrocyte death by increased extracellular phosphate concentrations. Kidney Blood Press Res 38(1):42–51. https://doi.org/10.1159/000355752
Abed M, Feger M, Alzoubi K, Pakladok T, Frauenfeld L, Geiger C, Towhid ST, Lang F (2013) Sensitization of erythrocytes to suicidal erythrocyte death following water deprivation. Kidney Blood Press Res 37(6):567–578. https://doi.org/10.1159/000355737
Kempe-Teufel DS, Bissinger R, Qadri SM, Wagner R, Peter A, Lang F (2018) Cellular markers of eryptosis are altered in type 2 diabetes. Clin Chem Lab Med 56(7):e177–e180. https://doi.org/10.1515/cclm-2017-1058
Lang E, Gatidis S, Freise NF, Bock H, Kubitz R, Lauermann C, Orth HM, Klindt C, Schuier M, Keitel V, Reich M, Liu G, Schmidt S, Xu HC, Qadri SM, Herebian D, Pandyra AA, Mayatepek E, Gulbins E, Lang F, Haussinger D, Lang KS, Foller M, Lang PA (2015) Conjugated bilirubin triggers anemia by inducing erythrocyte death. Hepatology (Baltimore MD) 61(1):275–284. https://doi.org/10.1002/hep.27338
Xu W, Peng F, Deng Y, Fan X, Li N (2019) The emerging roles of eryptosis in liver diseases. Transfus Clin Biol. https://doi.org/10.1016/j.tracli.2019.05.004
Lang PA, Schenck M, Nicolay JP, Becker JU, Kempe DS, Lupescu A, Koka S, Eisele K, Klarl BA, Rubben H, Schmid KW, Mann K, Hildenbrand S, Hefter H, Huber SM, Wieder T, Erhardt A, Haussinger D, Gulbins E, Lang F (2007) Liver cell death and anemia in Wilson disease involve acid sphingomyelinase and ceramide. Nat Med 13(2):164–170. https://doi.org/10.1038/nm1539
du Plooy JN, Bester J, Pretorius E (2018) Eryptosis in haemochromatosis: implications for rheology. Clin Hemorheol Microcirc 69(4):457–469. https://doi.org/10.3233/CH-170325
Pretorius E (2018) Erythrocyte deformability and eryptosis during inflammation, and impaired blood rheology. Clin Hemorheol Microcirc 69(4):545–550. https://doi.org/10.3233/CH-189205
Bester J, Swanepoel AC, Windberger U (2022) Pathological changes in Erythrocytes during inflammation and infection, vol 13. Frontiers Media SA
Borst O, Abed M, Alesutan I, Towhid ST, Qadri SM, Foller M, Gawaz M, Lang F (2012) Dynamic adhesion of eryptotic erythrocytes to endothelial cells via CXCL16/SR-PSOX. Am J Physiol Cell Physiol 302(4):C644–C651
Andrews DA, Low PS (1999) Role of red blood cells in thrombosis. Curr Opin Hematol 6(2):76–82
Chung SM, Bae ON, Lim KM, Noh JY, Lee MY, Jung YS, Chung JH (2007) Lysophosphatidic acid induces thrombogenic activity through phosphatidylserine exposure and procoagulant microvesicle generation in human erythrocytes. Arterioscler Thromb Vasc Biol 27(2):414–421
Li D, Zheng X, Zhang Y, Li X, Chen X, Yin Y, Hu J, Li J, Guo M, Wang X (2022) What should be responsible for eryptosis in chronic kidney disease? Kidney and Blood Pressure Research 47(6):375–390
Scovino AM, Totino PRR, Morrot A (2022) Eryptosis as a new insight in malaria pathogenesis.Frontiers in Immunology13
Acknowledgements
The authors acknowledge the meticulous preparation of the manuscript by Lejla Subasic.
Funding
This study was funded by China Postdoctoral Science Foundation (2021MD703799).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. JL, AAMB and FL conceived and designed research. Material preparation, data collection and analysis were performed by JL, AAMB, KM, XZ and KZ. The first draft of the manuscript was written by FL and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Ethics approval
The study is approved by the ethics committee of Eberhard-Karls-University of Tuebingen (184/2003 V).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, J., Mamun Bhuyan, A.A., Ma, K. et al. Myricetin-induced suicidal erythrocyte death. Mol Biol Rep 50, 4253–4260 (2023). https://doi.org/10.1007/s11033-023-08350-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08350-3