Abstract
Background
Gastric cancer is a heterogeneous disease associated to deregulated gastric epithelia tight junction barrier function and di novo expression of claudin-6; these changes are associated with epithelial-mesenchymal transition, enhanced invasiveness, metastatic progression, resistance to chemotherapy, and poor prognosis. Gastric cancer stem cells represent a rare population of cells within the tumor implicated in tumor growth and higher tumorigenic capacity. The possible relation between claudin-6 expression and the expression of some markers associated to epithelial mesenchymal transition and cancer stem cells in gastric cancer cells have never been explored.
Methods and results
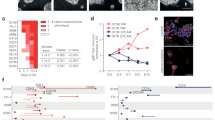
CD44, CD24, Twist, Villin, DCLK1, claudin-6, NANOG, E-Cadherin, SOX2, and SNAI1 expression was evaluated by immunofluorescence and cytofluorometry in wild type and Claudin-6 transfected AGS cells. Cell migration assays were also performed. Differentially expressed genes and biological processes analysis was performed to determine gene preponderance. The results showed that claudin-6 overexpression enriched the CD44 + /CD24- subpopulation with an overall increase in the expression and the number of CD44 + cells. A significant increase in NANOG, SOX2 and SNAI1 expression and enhanced cell migration was observed in claudin-6 transfected cells. Transcriptome analysis revealed 271 genes involved in enhanced biological processes with only 31 with a significantly p value; thirteen of those genes are closely associated to epithelial mesenchymal transition processes and folding and unfolding processes of proteins in the endoplasmic reticulum.
Conclusions
The pro-tumorigenic effect of claudin-6 in gastric cancer could be associated to dedifferentiation of epithelial cells and an increase in di novo cancer stem cell genesis.





Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M et al (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136(5):E359–E386
Gao JP, Xu W, Liu WT, Yan M, Zhu ZG (2018) Tumor heterogeneity of gastric cancer: from the perspective of tumor-initiating cell. World J Gastroenterol 24(24):2567–2581
Wang H, Yang X (2015) The expression patterns of tight junction protein claudin-1, -3, and -4 in human gastric neoplasms and adjacent non-neoplastic tissues. Int J Clin Exp Pathol 8(1):881–887
Suzuki H, Tani K, Tamura A, Tsukita S, Fujiyoshi Y (2015) Model for the architecture of claudin-based paracellular ion channels through tight junctions. J Mol Biol 427(2):291–297
Iravani O, Tay BW, Chua PJ, Yip GW, Bay BH (2013) Claudins and gastric carcinogenesis. Exp Biol Med (Maywood) 238(4):344–349
Corsini M, Ravaggi A, Odicino F, Santin AD, Ravelli C, Presta M et al (2018) Claudin3 is localized outside the tight junctions in human carcinomas. Oncotarget 9(26):18446–18453
Jung H, Jun KH, Jung JH, Chin HM, Park WB (2011) The expression of claudin-1, claudin-2, claudin-3, and claudin-4 in gastric cancer tissue. J Surg Res 167(2):e185–e191
Ahmad R, Kumar B, Pan K, Dhawan P, Singh AB (2017) HDAC-4 regulates claudin-2 expression in EGFR-ERK1/2 dependent manner to regulate colonic epithelial cell differentiation. Oncotarget 8(50):87718–87736
Hwang TL, Changchien TT, Wang CC, Wu CM (2014) Claudin-4 expression in gastric cancer cells enhances the invasion and is associated with the increased level of matrix metalloproteinase-2 and -9 expression. Oncol Lett 8(3):1367–1371
Son HJ, An CH, Yoo NJ, Lee SH (2020) Tight junction-related CLDN5 and CLDN6 genes, and gap junction-related GJB6 and GJB7 genes are somatically mutated in gastric and colorectal cancers. Pathol Oncol Res 26(3):1983–1987
Hashimoto I, Oshima T (2020) Claudins and gastric cancer: an overview. Cancers (Basel) 14(2):290
Stadler CR, Bähr-Mahmud H, Plum LM, Schmoldt K, Kölsch AC, Türeci Ö et al (2016) Characterization of the first-in-class T-cell-engaging bispecific single-chain antibody for targeted immunotherapy of solid tumors expressing the oncofetal protein claudin 6. Oncoimmunology 5(3):e1091555
Ushiku T, Shinozaki-Ushiku A, Maeda D, Morita S, Fukayama M (2012) Distinct expression pattern of claudin-6, a primitive phenotypic tight junction molecule, in germ cell tumours and visceral carcinomas. Histopathology 61(6):1043–1056
Manku G, Hueso A, Brimo F, Chan P, Gonzalez-Peramato P, Jabado N et al (2016) Changes in the expression profiles of claudins during gonocyte differentiation and in seminomas. Andrology 4(1):95–110
Zavala-Zendejas VE, Torres-Martinez AC, Salas-Morales B, Fortoul TI, Montano LF, Rendon-Huerta EP (2011) Claudin-6, 7, or 9 overexpression in the human gastric adenocarcinoma cell line AGS increases its invasiveness, migration, and proliferation rate. Cancer Invest 29(1):1–11
Torres-Martinez AC, Gallardo-Vera JF, Lara-Holguin AN, Montano LF, Rendon-Huerta EP (2017) Claudin-6 enhances cell invasiveness through claudin-1 in AGS human adenocarcinoma gastric cancer cells. Exp Cell Res 350(1):226–235
Brabletz S, Schuhwerk H, Brabletz T, Stemmler MP (2021) Dynamic EMT: a multi-tool for tumor progression. EMBO J 40(18):e108647
Chen T, You Y, Jiang H, Wang ZZ (2017) Epithelial-mesenchymal transition (EMT): a biological process in the development, stem cell differentiation, and tumorigenesis. J Cell Physiol 232(12):3261–3272
Gowrikumar S, Singh AB, Dhawan P (2019) Role of claudin proteins in regulating cancer stem cells and chemoresistance-potential implication in disease prognosis and therapy. Int J Mol Sci 21(1):53
Akhavan MM, Karimi M, Ghodrati M, Falahtpishe H (2011) AT1 receptors activation enhances the expression of MMP-2, MMP-13 and VEGF but not MMP-9 in B16F10 melanoma cells. Pak J Biol Sci 14(17):821–830
Sakuma K, Aoki M, Kannagi R (2012) Transcription factors c-Myc and CDX2 mediate E-selectin ligand expression in colon cancer cells undergoing EGF/bFGF-induced epithelial-mesenchymal transition. Proc Natl Acad Sci USA 109(20):7776–7781
Mazzoni A, Maggi L, Siracusa F, Ramazzotti M, Rossi MC, Santarlasci V et al (2019) Eomes controls the development of Th17-derived (non-classic) Th1 cells during chronic inflammation. Eur J Immunol 49(1):79–95
Carvalho BS, Irizarry RA (2010) A framework for oligonucleotide microarray preprocessing. Bioinformatics 26(19):2363–2367
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W et al (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43(7):e47
Falcon S, Gentleman R (2007) Using GOstats to test gene lists for GO term association. Bioinformatics 23(2):257–258
Fu Y, Li H, Hao X (2017) The self-renewal signaling pathways utilized by gastric cancer stem cells. Tumour Biol 39(4):1010428317697577
Zhang C, Li C, He F, Cai Y, Yang H (2011) Identification of CD44+CD24+ gastric cancer stem cells. J Cancer Res Clin Oncol 137(11):1679–1686
Chang JC (2016) Cancer stem cells: role in tumor growth, recurrence, metastasis, and treatment resistance. Medicine (Baltimore) 95(1 Suppl 1):S20–S25
Rawla P, Barsouk A (2019) Epidemiology of gastric cancer: global trends, risk factors and prevention. Prz Gastroenterol 14(1):26–38
Nassar D, Blanpain C (2016) Cancer stem cells: basic concepts and therapeutic implications. Annu Rev Pathol 11:47–76
Najafi M, Mortezaee K, Majidpoor J (2019) Cancer stem cell (CSC) resistance drivers. Life Sci 234:116781
Vlashi E, Pajonk F (2015) Cancer stem cells, cancer cell plasticity and radiation therapy. Semin Cancer Biol 31:28–35
Ishizawa K, Rasheed ZA, Karisch R, Wang Q, Kowalski J, Susky E et al (2010) Tumor-initiating cells are rare in many human tumors. Cell Stem Cell 7(3):279–282
Rasheed ZA, Yang J, Wang Q, Kowalski J, Freed I, Murter C et al (2010) Prognostic significance of tumorigenic cells with mesenchymal features in pancreatic adenocarcinoma. J Natl Cancer Inst 102(5):340–351
Yang ZF, Ho DW, Ng MN, Lau CK, Yu WC, Ngai P et al (2008) Significance of CD90+ cancer stem cells in human liver cancer. Cancer Cell 13(2):153–166
Dalerba P, Dylla SJ, Park IK, Liu R, Wang X, Cho RW et al (2007) Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci USA 104(24):10158–10163
Nguyen PH, Giraud J, Chambonnier L, Dubus P, Wittkop L, Belleannee G et al (2017) Characterization of biomarkers of tumorigenic and chemoresistant cancer stem cells in human gastric carcinoma. Clin Cancer Res 23(6):1586–1597
Nguyen PH, Giraud J, Staedel C, Chambonnier L, Dubus P, Chevret E et al (2016) All-trans retinoic acid targets gastric cancer stem cells and inhibits patient-derived gastric carcinoma tumor growth. Oncogene 35(43):5619–5628
Ayob AZ, Ramasamy TS (2018) Cancer stem cells as key drivers of tumour progression. J Biomed Sci 25(1):20
Tabaries S, Siegel PM (2017) The role of claudins in cancer metastasis. Oncogene 36(9):1176–1190
Ben-David U, Nudel N, Benvenisty N (2013) Immunologic and chemical targeting of the tight-junction protein claudin-6 eliminates tumorigenic human pluripotent stem cells. Nat Commun 4:1992
Sugimoto K, Ichikawa-Tomikawa N, Satohisa S, Akashi Y, Kanai R, Saito T et al (2013) The tight-junction protein claudin-6 induces epithelial differentiation from mouse F9 and embryonic stem cells. PLoS ONE 8(10):e75106
Wang L, Xue Y, Shen Y, Li W, Cheng Y, Yan X et al (2012) Claudin 6: a novel surface marker for characterizing mouse pluripotent stem cells. Cell Res 22(6):1082–1085
Li J (2021) Dysregulated expression of claudins in cancer. Oncol Lett 22(3):641
Rendon-Huerta E, Teresa F, Teresa GM, Xochitl GS, Georgina AF, Veronica ZZ et al (2010) Distribution and expression pattern of claudins 6, 7, and 9 in diffuse- and intestinal-type gastric adenocarcinomas. J Gastrointest Cancer 41(1):52–59
Yang M, Li Y, Ruan Y, Lu Y, Lin D, Xie Y et al (2018) CLDN6 enhances chemoresistance to ADM via AF-6/ERKs pathway in TNBC cell line MDAMB231. Mol Cell Biochem 443(1–2):169–180
Kohmoto T, Masuda K, Shoda K, Takahashi R, Ujiro S, Tange S et al (2020) Claudin-6 is a single prognostic marker and functions as a tumor-promoting gene in a subgroup of intestinal type gastric cancer. Gastric Cancer 23(3):403–417
Chen C, Zhao S, Karnad A, Freeman JW (2018) The biology and role of CD44 in cancer progression: therapeutic implications. J Hematol Oncol 11(1):64
Jaggupilli A, Elkord E (2012) Significance of CD44 and CD24 as cancer stem cell markers: an enduring ambiguity. Clin Dev Immunol 2012:708036
Hui D, Chen J, Jiang Y, Pan Y, Zhang Z, Dong M et al (2020) CD44+CD24-/low sphere-forming cells of EBV-associated gastric carcinomas show immunosuppressive effects and induce tregs partially through production of PGE2. Exp Cell Res 390(2):111968
Lizarraga-Verdugo E, Avendano-Felix M, Bermudez M, Ramos-Payan R, Perez-Plasencia C, Aguilar-Medina M (2020) Cancer stem cells and its role in angiogenesis and vasculogenic mimicry in gastrointestinal cancers. Front Oncol 10:413
Cao X, Cao D, Jin M, Jia Z, Kong F, Ma H et al (2014) CD44 but not CD24 expression is related to poor prognosis in non-cardia adenocarcinoma of the stomach. BMC Gastroenterol 14:157
Zhu Y, Wang C, Becker SA, Hurst K, Nogueira LM, Findlay VJ et al (2018) miR-145 Antagonizes SNAI1-mediated stemness and radiation resistance in colorectal cancer. Mol Ther 26(3):744–754
van Schaijik B, Davis PF, Wickremesekera AC, Tan ST, Itinteang T (2018) Subcellular localisation of the stem cell markers OCT4, SOX2, NANOG, KLF4 and c-MYC in cancer: a review. J Clin Pathol 71(1):88–91
Arndt GM, Dossey L, Cullen LM, Lai A, Druker R, Eisbacher M et al (2009) Characterization of global microRNA expression reveals oncogenic potential of miR-145 in metastatic colorectal cancer. BMC Cancer 9:374
Das PK, Islam F, Lam AK (2020) The roles of cancer stem cells and therapy resistance in colorectal carcinoma. Cells 9(6):1392
Bakir B, Chiarella AM, Pitarresi JR, Rustgi AK (2020) EMT, MET, plasticity, and tumor metastasis. Trends Cell Biol 30(10):764–776
Zhan M, Wang H, Chen T, Chen W, Yang L, He M et al (2015) NOX1 mediates chemoresistance via HIF1α/MDR1 pathway in gallbladder cancer. Biochem Biophys Res Commun 468(1–2):79–85
Mercurio AM (2019) VEGF/neuropilin signaling in cancer stem cells. Int J Mol Sci 20(3):490
Voog J, Jones DL (2010) Stem cells and the niche: a dynamic duo. Cell Stem Cell 6(2):103–115
Barbato L, Bocchetti M, Di Biase A, Regad T (2019) Cancer stem cells and targeting strategies. Cells 8(8):926
Acknowledgements
Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica (PAPIIT) grants IN218019 and IN221519, and Programa de Becas Posdoctorales DGAPA-UNAM, Universidad Nacional Autónoma de México, México, supported this work.
Funding
DGAPA-PAPIIT,IN218019,Erika Patricia Rendón-Huerta,IN221519,Luis F Montaño
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by PAM-G, FC-V, and AAdC. The first draft of the manuscript was written by LFM and EPRH and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no conflict of interest exists.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Medrano-Gonzálezl, P.A., Cruz-Villegas, F., Alarcón del Carmen, A. et al. Claudin-6 increases SNAI1, NANOG and SOX2 gene expression in human gastric adenocarcinoma AGS cells. Mol Biol Rep 49, 11663–11674 (2022). https://doi.org/10.1007/s11033-022-07976-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07976-z