Abstract
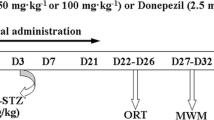
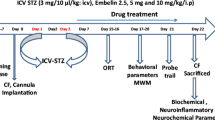
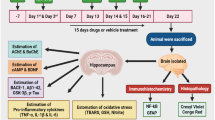
Alzheimer’s disease (AD) is the most common neurodegenerative disorder among the elderly. In the light of increasing AD prevalence and lack of effective treatment, new strategies to prevent or reverse this condition are needed. Levetiracetam (LEV) is a newer antiepileptic drug that is commonly used to treat certain types of seizures. Researches indicated that LEV has several other pharmacological activities, including improvement of cognitive function. In this study, the recovery effects of chronic (28 days) administration of LEV (50, 100, and 150 mg/kg, ip) on cognitive deficits caused by the intracerebroventricular (icv) injection of streptozotocin (STZ), as a model for sporadic AD, were evaluated in rats. We also considered the protective effects of LEV against hippocampal cell loss, oxidative damage, acetylcholinesterase (AChE) activity, neuroinflammation, and tauopathy caused by STZ. LEV (100 and 150 mg/kg) significantly attenuated the STZ-induced learning and memory impairments in the passive avoidance and Morris water maze (MWM) tasks. In addition, LEV suppressed STZ-induced hippocampal neuronal loss, while restored alterations in the redox status (lipid peroxides and glutathione), AChE activity, proinflammatory cytokines (IL-1β, IL-6, TNF-α), and hyperphosphorylation of tau linked to STZ administration. In conclusion, our study demonstrated that LEV alleviated hippocampal cell death and memory deficits in STZ-AD rats, through mitigating oxidative damage, suppression of proinflammatory cytokines expression, and inhibition of abnormal tau hyperphosphorylation.







Similar content being viewed by others
Data availability
Derived data supporting the findings of this study are available from the corresponding author [HRS] on request.
References
Abed El-Gaphar OAM, Abo-Youssef AM, Halal GK (2018) Levetiracetam mitigates lipopolysaccharide-induced JAK2/STAT3 and TLR4/MAPK signaling pathways activation in a rat model of adjuvant- induced arthritis. Eur J Pharmacol 826:85–95
Agostinho P, Cunha RA, Oliveira C (2010) Neuroinflammation, oxidative stress and the pathogenesis of Alzheimer’s disease. Curr Pharm Des 16:2766–2778
Alam Q, Alam MZ, Mushtaq G, Damanhouri GA, Rasool M, Kamal MA, Haque A (2016) Inflammatory Process in Alzheimer’s and Parkinson’s Diseases: Central Role of Cytokines. Curr Pharm Des 22:541–548
Alrabiah H (2019) Levetiracetam. Profiles Drug Subst Excip Relat Methodol 44:167–204
Amani M, Zolghadrnasab M, Salari AA (2019) NMDA receptor in the hippocampus alters neurobehavioral phenotypes through inflammatory cytokines in rats with sporadic Alzheimer-like disease. Physiol Behav 202:52–61
Barilar JO, Knezovic A, Grünblatt E, Riederer P, Salkovic-Petrisic M (2015) Nine-month follow-up of the insulin receptor signalling cascade in the brain of streptozotocin rat model of sporadic Alzheimer’s disease. J Neural Transm (Vienna) 122:565–576
Barrientos RM, Kitt MM, Watkins LR, Maier SF (2015) Neuroinflammation in the normal aging hippocampus. Neuroscience 309:84–99
Celikyurt IK, Ulak G, Mutlu O, Akar FY, Mulayim S, Erden F, Komsuoglu SS (2012) Positive impact of levetiracetam on emotional learning and memory in naive mice. Life Sci 90:185–189
Chen XQ, Mobley WC (2019) Alzheimer disease pathogenesis: insights from molecular and cellular biology studies of oligomeric Aβ and tau species. Front Neurosci 13:659
Chung HY, Sung B, Jung KJ, Zou Y, Yu BP (2006) The molecular inflammatory process in aging. Antioxid Redox Sign 8:572–581
Conway ME (2020) Alzheimer’s disease: targeting the glutamatergic system. Biogerontology 21:257–274
Correia SC, Santos RX, Perry G, Zhu X, Moreira PI, Smith MA (2011) Insulin-resistant brain state: the culprit in sporadic Alzheimer’s disease? Ageing Res Rev 10:264–273
Detrait E, Leclercq K, Löscher W, Potschka H, Niespodziany I, Hanon E, Kaminski R, Matagne A, Lamberty Y (2010) Brivaracetam does not alter spatial learning and memory in both normal and amygdala-kindled rats. Epilepsy Res 91:74–83
Devi L, Ohno M (2013) Effects of levetiracetam, an antiepileptic drug, on memory impairments associated with aging and Alzheimer’s disease in mice. Neurobiol Learn Mem 102:7–11
Ebada MA, Alkanj S, Ebada M, Abdelkarim AH, Diab A, Aziz MAE, Soliman AM, Fayed N, Bahbah EI, Negida A (2019) Safety and efficacy of levetiracetam for the management of levodopa- induced dyskinesia in patients with Parkinson’s disease: a systematic review. CNS Neurol Disord Drug Targets 18:317–325
Ebrahimi K, Majdi A, Baghaiee B, Hosseini SH, Sadigh-Eteghad S (2017) Physical activity and beta-amyloid pathology in Alzheimer’s disease: A sound mind in a sound body. Excli J 16:959–972
El Sayed NS, Ghoneum MH (2020) Antia, a natural antioxidant product, attenuates cognitive dysfunction in Streptozotocin-induced mouse model of sporadic Alzheimer’s disease by targeting the amyloidogenic, inflammatory, autophagy, and oxidative stress pathways. Oxid Med Cell Longev 2020:4386562
Elçioğlu H, Kabasakal L, Alan S, Salva E, Tufan F, Karan M (2013) Thalidomide attenuates learning and memory deficits induced by intracerebroventricular administration of streptozotocin in rats. Biotech Histochem 88:145–152
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Erbaş O, Yılmaz M, Taşkıran D (2016) Levetiracetam attenuates rotenone-induced toxicity: A rat model of Parkinson’s disease. Environ Toxicol Pharmacol 42:226–230
Fanoudi S, Hosseini M, Alavi MS, Boroushaki MT, Hosseini A, Sadeghnia HR (2018) Everolimus, a mammalian target of rapamycin inhibitor, ameliorated streptozotocin-induced learning and memory deficits via neurochemical alterations in male rats. Excli J 17:999–1017
Fanoudi S, Alavi MS, Hosseini M, Sadeghnia HR (2019) Nigella sativa and thymoquinone attenuate oxidative stress and cognitive impairment following cerebral hypoperfusion in rats. Metab Brain Dis 34:1001–1010
Grieb P (2016) Intracerebroventricular streptozotocin injections as a model of Alzheimer’s disease: in search of a relevant mechanism. Mol Neurobiol 53:1741–1752
Haghikia A, Ladage K, Hinkerohe D, Vollmar P, Heupel K, Dermietzel R, Faustmann PM (2008) Implications of antiinflammatory properties of the anticonvulsant drug levetiracetam in astrocytes. J Neurosci Res 86:1781–1788
Hanon E, Klitgaard H (2001) Neuroprotective properties of the novel antiepileptic drug levetiracetam in the rat middle cerebral artery occlusion model of focal cerebral ischemia. Seizure 10:287–293
Imai T, Sugiyama T, Iwata S, Nakamura S, Shimazawa M, Hara H (2020) Levetiracetam, an antiepileptic drug has neuroprotective effects on intracranial hemorrhage injury. Neurosci 431:25–33
Inaba T, Miyamoto N, Hira K, Ueno Y, Yamashiro K, Watanabe M, Shimada Y, Hattori N, Urabe T (2019) Protective role of levetiracetam against cognitive impairment and brain white matter damage in mouse prolonged cerebral Hypoperfusion. Neuroscience 414:255–264
Itoh K, Ishihara Y, Komori R, Nochi H, Taniguchi R, Chiba Y, Ueno M, Takata-Tsuji F, Dohgu S, Kataoka Y (2016) Levetiracetam treatment influences blood-brain barrier failure associated with angiogenesis and inflammatory responses in the acute phase of epileptogenesis in post-status epilepticus mice. Brain Res 1652:1–13
Itoh K, Taniguchi R, Matsuo T, Oguro A, Vogel CFA, Yamazaki T, Ishihara Y (2019) Suppressive effects of levetiracetam on neuroinflammation and phagocytic microglia: A comparative study of levetiracetam, valproate and carbamazepine. Neurosci Lett 708:134363
Kim JE, Choi HC, Song HK, Jo SM, Kim DS, Choi SY, Kim YI, Kang TC (2010) Levetiracetam inhibits interleukin-1 beta inflammatory responses in the hippocampus and piriform cortex of epileptic rats. Neurosci Lett 471:94–99
Lamberty Y, Margineanu DG, Klitgaard H (2000) Absence of negative impact of levetiracetam on cognitive function and memory in normal and amygdala-kindled rats. Epilepsy Behav 1:333–342
Lamtai M, Azirar S, Zghari O, Ouakki S, El Hessni A, Mesfioui A, Ouichou A (2021) Melatonin Ameliorates Cadmium-Induced Affective and Cognitive Impairments and Hippocampal Oxidative Stress in Rat. Biol Trace Elem Res 199:1445–1455
Lasierra-Cirujeda J, Coronel P, Aza M, Gimeno M (2013) Beta-amyloidolysis and glutathione in Alzheimer’s disease. J Blood Med 4:31–38
Lee Y, Kim YH, Park SJ, Huh JW, Kim SH, Kim SU, Kim JS, Jeong KJ, Lee KM, Hong Y (2014) Insulin/IGF signaling-related gene expression in the brain of a sporadic Alzheimer’s disease monkey model induced by intracerebroventricular injection of streptozotocin. J Alzheimer’s Dis 38:251–267
Lyseng-Williamson KA (2011) Levetiracetam: a review of its use in epilepsy. Drugs 71:489–514
Marini H, Costa C, Passaniti M, Esposito M, Campo GM, Ientile R, Adamo EB, Marini R, Calabresi P, Altavilla D, Minutoli L, Pisani F, Squadrito F (2004) Levetiracetam protects against kainic acid-induced toxicity. Life Sci 74:1253–1264
Masters CL, Bateman R, Blennow K, Rowe CC, Sperling RA, Cummings JL (2015) Alzheimer’s disease. Nat Rev Dis Primers 1:15056
Miyazaki I, Murakami S, Torigoe N, Kitamura Y, Asanuma M (2016) Neuroprotective effects of levetiracetam target xCT in astrocytes in parkinsonian mice. J Neurochem 136:194–204
Mohammad HMF, Sami MM, Makary S, Toraih EA, Mohamed AO, El-Ghaiesh SH (2019) Neuroprotective effect of levetiracetam in mouse diabetic retinopathy: Effect on glucose transporter-1 and GAP43 expression. Life Sci 232:116588
Moran SP, Dickerson JW, Cho HP, Xiang Z, Maksymetz J, Remke DH, Lv X, Doyle CA, Rajan DH, Niswender CM, Engers DW, Lindsley CW, Rook JM, Conn PJ (2018) M(1)-positive allosteric modulators lacking agonist activity provide the optimal profile for enhancing cognition. Neuropsychopharmacol 43:1763–1771
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim Biophys Acta 582:67–78
Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods 11:47–60
Muller AP, Ferreira GK, Pires AJ, de Bem Silveira G, de Souza DL, de Abreu Brandolfi J, de Souza CT, Paula MM, Silveira PCL (2017) Gold nanoparticles prevent cognitive deficits, oxidative stress and inflammation in a rat model of sporadic dementia of Alzheimer’s type. Mater Sci Eng C 77:476–483
Niu HX, Wang JZ, Wang DL, Miao JJ, Li H, Liu ZG, Yuan X, Liu W, Zhou JR (2018) The Orally Active Noncompetitive AMPAR Antagonist Perampanel Attenuates Focal Cerebral Ischemia Injury in Rats. Cell Mol Neurobiol 38:459–466
Oliveira AA, Nogueira CR, Nascimento VS, Aguiar LM, Freitas RM, Sousa FC, Viana GS, Fonteles MM (2005) Evaluation of levetiracetam effects on pilocarpine-induced seizures: cholinergic muscarinic system involvement. Neurosci Lett 385:184–188
Paula-Lima AC, De Felice FG, Brito-Moreira J, Ferreira ST (2005) Activation of GABAA receptors by taurine and muscimol blocks the neurotoxicity of β-amyloid in rat hippocampal and cortical neurons. Neuropharmacol 49:1140–1148
Paxinos G, Watson C (1998) A stereotaxic atlas of the rat brain. Academic, New York
Pilipenko V, Pupure J, Rumaks J, Beitnere U, Dzirkale Z, Skumbins R, Klusa V (2015) GABAA agonist muscimol ameliorates learning/memory deficits in streptozocin-induced Alzheimer’s disease non-transgenic rat model. SpringerPlus 4:P36
Rai S, Kamat PK, Nath C, Shukla R (2013) A study on neuroinflammation and NMDA receptor function in STZ (ICV) induced memory impaired rats. J Neuroimmunol 254:1–9
Rajabian A, Sadeghnia HR, Hosseini A, Mousavi SH, Boroushaki MT (2020) 3-Acetyl-11-keto-β-boswellic acid attenuated oxidative glutamate toxicity in neuron-like cell lines by apoptosis inhibition. J Cell Biochem 121:1778–1789
Rajasekar N, Nath C, Hanif K, Shukla R (2017) Intranasal insulin administration ameliorates streptozotocin (ICV)-induced insulin receptor dysfunction, neuroinflammation, amyloidogenesis, and memory impairment in rats. Mol Neurobiol 54:6507–6522
Ravelli KG, dos Anjos Rosário B, Camarini R, Hernandes MS, Britto LR (2017) Intracerebroventricular streptozotocin as a model of Alzheimer’s disease: neurochemical and behavioral characterization in mice. Neurotoxicity Res 31:327–333
Reeta KH, Mehla J, Gupta YK (2009) Curcumin is protective against phenytoin-induced cognitive impairment and oxidative stress in rats. Brain Res 1301:52–60
Sanchez PE, Zhu L, Verret L, Vossel KA, Orr AG, Cirrito JR, Devidze N, Ho K, Yu G-Q, Palop JJ (2012) Levetiracetam suppresses neuronal network dysfunction and reverses synaptic and cognitive deficits in an Alzheimer’s disease model. Proc Natl Acad Sci 109:E2895–E2903
Schliebs R, Arendt T (2011) The cholinergic system in aging and neuronal degeneration. Behav Brain Res 221:555–563
Schoenberg MR, Rum RS, Osborn KE, Werz MA (2017) A randomized, double-blind, placebo‐controlled crossover study of the effects of levetiracetam on cognition, mood, and balance in healthy older adults. Epilepsia 58:1566–1574
Shannon HE, Love PL (2004) Effects of antiepileptic drugs on working memory as assessed by spatial alternation performance in rats. Epilepsy Behav 5:857–865
Sharma Y, Garabadu D (2020) Intracerebroventricular streptozotocin administration impairs mitochondrial calcium homeostasis and bioenergetics in memory-sensitive rat brain regions. Exp Brain Res 238:2293–2306
Shi JQ, Shen W, Chen J, Wang BR, Zhong LL, Zhu YW, Zhu HQ, Zhang QQ, Zhang YD, Xu J (2011) Anti-TNF-α reduces amyloid plaques and tau phosphorylation and induces CD11c-positive dendritic-like cell in the APP/PS1 transgenic mouse brains. Brain Res 1368:239–247
Shi JQ, Wang BR, Tian YY, Xu J, Gao L, Zhao SL, Jiang T, Xie HG, Zhang YD (2013) Antiepileptics topiramate and levetiracetam alleviate behavioral deficits and reduce neuropathology in APPswe/PS1dE9 transgenic mice. CNS Neurosci Ther 19:871–881
Sola I, Aso E, Frattini D, López-González I, Espargaró A, Sabaté R, Di Pietro O, Luque FJ, Clos MV, Ferrer I, Muñoz-Torrero D (2015) Novel levetiracetam derivatives that are effective against the Alzheimer-like phenotype in mice: synthesis, in vitro, ex vivo, and in vivo efficacy studies. J Med Chem 58:6018–6032
Stienen MN, Haghikia A, Dambach H, Thöne J, Wiemann M, Gold R, Chan A, Dermietzel R, Faustmann PM, Hinkerohe D, Prochnow N (2011) Anti-inflammatory effects of the anticonvulsant drug levetiracetam on electrophysiological properties of astroglia are mediated via TGFβ1 regulation. Br J Pharmacol 162:491–507
Stuchbury G, Münch G (2005) Alzheimer’s associated inflammation, potential drug targets and future therapies. J Neural Transm (Vienna) 112:429–453
Tan J, Paquette V, Levine M, Ensom MHH (2017) Levetiracetam clinical pharmacokinetic monitoring in pediatric patients with epilepsy. Clin Pharmacokinet 56:1267–1285
Trikash I, Kasatkina L, Lykhmus O, Skok M (2020) Nicotinic acetylcholine receptors regulate clustering, fusion and acidification of the rat brain synaptic vesicles. Neurochem Int 138:104779
Ueda Y, Doi T, Nagatomo K, Tokumaru J, Takaki M, Willmore LJ (2007) Effect of levetiracetam on molecular regulation of hippocampal glutamate and GABA transporters in rats with chronic seizures induced by amygdalar FeCl3 injection. Brain Res 1151:55–61
Vafaee F, Hosseini M, Hassanzadeh Z, Edalatmanesh MA, Sadeghnia HR, Seghatoleslam M, Mousavi SM, Amani A, Shafei MN (2015) The effects of nigella sativa hydro-alcoholic extract on memory and brain tissues oxidative damage after repeated seizures in rats. Iran J Pharm Res 14:547–557
Varoglu AO, Yildirim A, Aygul R, Gundogdu OL, Sahin YN (2010) Effects of valproate, carbamazepine, and levetiracetam on the antioxidant and oxidant systems in epileptic patients and their clinical importance. Clin Neuropharmacol 33:155–157
Verloes R, Scotto A-M, Gobert J, Wülfert E (1988) Effects of nootropic drugs in a scopolamine-induced amnesia model in mice. Psychopharmacology 95:226–230
Vogl C, Mochida S, Wolff C, Whalley BJ, Stephens GJ (2012) The synaptic vesicle glycoprotein 2A ligand levetiracetam inhibits presynaptic Ca2+ channels through an intracellular pathway. Mol Pharmacol 82:199–208
Wang MJ, Jiang L, Chen HS, Cheng L (2019) Levetiracetam protects against cognitive impairment of subthreshold convulsant discharge model rats by activating Protein Kinase C (PKC)-Growth-Associated Protein 43 (GAP-43)-Calmodulin-Dependent Protein Kinase (CaMK) signal transduction pathway. Med Sci Monit 25:4627–4638
Xiao R (2016) Levetiracetam might act as an efficacious drug to attenuate cognitive deficits of Alzheimer’s disease. Curr Top Med Chem 16:565–573
Xu J, Gao H, Zhang L, Rong S, Yang W, Ma C, Chen M, Huang Q, Deng Q, Huang F (2019) Melatonin alleviates cognition impairment by antagonizing brain insulin resistance in aged rats fed a high-fat diet. J pineal Res 67:e12584
Yeo HG, Lee Y, Jeon C-Y, Jeong KJ, Jin YB, Kang P, Kim SU, Kim JS, Huh JW, Kim YH (2015) Characterization of cerebral damage in a monkey model of Alzheimer’s disease induced by intracerebroventricular injection of streptozotocin. J Alzheimer’s Dis 46:989–1005
Zou H, Brayer SW, Hurwitz M, Niyonkuru C, Fowler LE, Wagner AK (2013) Neuroprotective, neuroplastic, and neurobehavioral effects of daily treatment with levetiracetam in experimental traumatic brain injury. Neurorehabil Neural Repair 27:878–888
Funding
This work was supported by the National Institute for Medical Research Development (NIMAD) of Islamic Republic of Iran [grant number 963629] and Vice Chancellery for Research and Technology, Mashhad University of Medical Sciences [grant number 4000621].
Author information
Authors and Affiliations
Contributions
HRS conceived and designed the study. MSA and SF collected the data and performed the data analysis under the supervision of HRS and MH. MSA and SF wrote the manuscript. HRS and MH revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
All animal experiments were conducted according to the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals, and the protocol was approved by the Institutional Animal Ethics Committee of Mashhad University of Medical Sciences, Mashhad, Iran.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alavi, M.S., Fanoudi, S., Hosseini, M. et al. Beneficial effects of levetiracetam in streptozotocin-induced rat model of Alzheimer’s disease. Metab Brain Dis 37, 689–700 (2022). https://doi.org/10.1007/s11011-021-00888-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00888-0